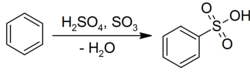From Wikipedia, the free encyclopedia
|
|||
 |
|||
 |
|||
| Names | |||
|---|---|---|---|
| IUPAC name
Sulfuric acid
|
|||
| Other names
Oil of vitriol
|
|||
| Identifiers | |||
| 7664-93-9 |
|||
| ChEBI | CHEBI:26836 |
||
| ChEMBL | ChEMBL572964 |
||
| ChemSpider | 1086 |
||
| EC number | 231-639-5 | ||
| Jmol-3D images | Image | ||
| KEGG | D05963 |
||
| RTECS number | WS5600000 | ||
| UNII | O40UQP6WCF |
||
| UN number | 1830 | ||
| Properties | |||
| H 2SO 4 |
|||
| Molar mass | 98.079 g/mol | ||
| Appearance | Clear, colorless, odorless liquid | ||
| Density | 1.84 g/cm3, liquid | ||
| Melting point | 10 °C (50 °F; 283 K) | ||
| Boiling point | 337 °C (639 °F; 610 K) When sulfuric acid is above 300 °C (572 °F), it will decompose slowly | ||
| miscible | |||
| Acidity (pKa) | −3, 1.99 | ||
| Viscosity | 26.7 cP (20 °C) | ||
| Thermochemistry | |||
Std molar
entropy (S |
157 J·mol−1·K−1[1] | ||
Std enthalpy of
formation (ΔfH |
−814 kJ·mol−1[1] | ||
| Hazards | |||
| MSDS | ICSC 0362 | ||
| GHS pictograms |  |
||
| GHS signal word | Danger | ||
| H314 | |||
| P260, P264, P280, P301+330+331, P303+361+353, P363, P304+340, P305+351+338, P310, P321, P310, P405, P501 | |||
| EU Index | 016-020-00-8 | ||
| EU classification | |||
| R-phrases | R35 | ||
| S-phrases | (S1/2) S26 S30 S45 | ||
| NFPA 704 | |||
| Flash point | Non-flammable | ||
| 15 mg/m3 (IDLH), 1 mg/m3 (TWA), 2 mg/m3 (STEL) | |||
LD50 (Lethal dose)
|
2140 mg/kg (oral, rat), LC50 = 25 mg/m3 (inhalation, rat) | ||
| US health exposure limits (NIOSH): | |||
PEL (Permissible)
|
TWA 1 mg/m3[4] | ||
| Related compounds | |||
Related strong acids
|
Selenic acid Hydrochloric acid Nitric acid Chromic acid |
||
Related compounds
|
Sulfurous acid Peroxymonosulfuric acid Sulfur trioxide Oleum |
||
Except where noted otherwise, data is given for materials in their standard state (at 25 °C (77 °F), 100 kPa)
|
|||
Sulfuric acid is a diprotic acid and shows different properties depending upon its concentration. Its corrosiveness on other materials, like metals, living tissues or even stones, can be mainly ascribed to its strong acidic nature and, if concentrated, strong dehydrating and oxidizing properties. Sulfuric acid at a high concentration can cause very serious damage upon contact, since not only does it cause chemical burns via hydrolysis, but also secondary thermal burns through dehydration.[8][9] It can lead to permanent blindness if splashed onto eyes and irreversible damage if swallowed.[8] Accordingly, safety precautions should be strictly observed when handling it. Moreover, it is hygroscopic, readily absorbing water vapour from the air.[5]
Sulfuric acid has a wide range of applications including domestic acidic drain cleaner,[10] electrolyte in lead-acid batteries and various cleaning agents. It is also a central substance in the chemical industry. Principal uses include mineral processing, fertilizer manufacturing, oil refining, wastewater processing, and chemical synthesis. It is widely produced with different methods, such as contact process, wet sulfuric acid process and some other methods.
History

John Dalton's 1808 sulfuric acid molecule shows a central sulfur atom bonded to three oxygen atoms, or sulfur trioxide, the anhydride of sulfuric acid.
The study of vitriol, a category of glassy minerals from which the acid can be derived, began in ancient times. Sumerians had a list of types of vitriol that they classified according to the substances' color. Some of the earliest discussions on the origin and properties of vitriol is in the works of the Greek physician Dioscorides (first century AD) and the Roman naturalist Pliny the Elder (23–79 AD). Galen also discussed its medical use. Metallurgical uses for vitriolic substances were recorded in the Hellenistic alchemical works of Zosimos of Panopolis, in the treatise Phisica et Mystica, and the Leyden papyrus X.[11]
Persian alchemists Jābir ibn Hayyān (c. 721 – c. 815 AD), Razi (865 – 925 AD), and Jamal Din al-Watwat (d. 1318, wrote the book Mabāhij al-fikar wa-manāhij al-'ibar), included vitriol in their mineral classification lists. Ibn Sina focused on its medical uses and different varieties of vitriol.[11]
Sulfuric acid was called "oil of vitriol" by medieval European alchemists because it was prepared by roasting "green vitriol" (iron (II) sulfate) in an iron retort. There are references to it in the works of Vincent of Beauvais and in the Compositum de Compositis ascribed to Saint Albertus Magnus. A passage from Pseudo-Geber´s Summa Perfectionis was long considered to be the first recipe for sulfuric acid, but this was a misinterpretation.[11]
In the seventeenth century, the German-Dutch chemist Johann Glauber prepared sulfuric acid by burning sulfur together with saltpeter (potassium nitrate, KNO
3), in the presence of steam. As saltpeter decomposes, it oxidizes the sulfur to SO
3, which combines with water to produce sulfuric acid. In 1736, Joshua Ward, a London pharmacist, used this method to begin the first large-scale production of sulfuric acid.
In 1746 in Birmingham, John Roebuck adapted this method to produce sulfuric acid in lead-lined chambers, which were stronger, less expensive, and could be made larger than the previously used glass containers. This process allowed the effective industrialization of sulfuric acid production. After several refinements, this method, called the lead chamber process or "chamber process", remained the standard for sulfuric acid production for almost two centuries.[1]
Sulfuric acid created by John Roebuck's process approached a 65% concentration. Later refinements to the lead chamber process by French chemist Joseph Louis Gay-Lussac and British chemist John Glover improved concentration to 78%. However, the manufacture of some dyes and other chemical processes require a more concentrated product. Throughout the 18th century, this could only be made by dry distilling minerals in a technique similar to the original alchemical processes. Pyrite (iron disulfide, FeS
2) was heated in air to yield iron(II) sulfate, FeSO
4, which was oxidized by further heating in air to form iron(III) sulfate, Fe2(SO4)3, which, when heated to 480 °C, decomposed to iron(III) oxide and sulfur trioxide, which could be passed through water to yield sulfuric acid in any concentration. However, the expense of this process prevented the large-scale use of concentrated sulfuric acid.[1]
In 1831, British vinegar merchant Peregrine Phillips patented the contact process, which was a far more economical process for producing sulfur trioxide and concentrated sulfuric acid. Today, nearly all of the world's sulfuric acid is produced using this method.[1]
Physical properties
Grades of sulfuric acid
Although nearly 99% sulfuric acid can be made, the subsequent loss of SO3 at the boiling point brings the concentration to 98.3% acid. The 98% grade is more stable in storage, and is the usual form of what is described as "concentrated sulfuric acid." Other concentrations are used for different purposes. Some common concentrations are:[12][13]
| Mass fraction H2SO4 |
Density (kg/L) |
Concentration (mol/L) |
Common name |
|---|---|---|---|
| 10% | 1.07 | ~1 | dilute sulfuric acid |
| 29–32% | 1.25–1.28 | 4.2–5 | battery acid (used in lead–acid batteries) |
| 62–70% | 1.52–1.60 | 9.6–11.5 | chamber acid fertilizer acid |
| 78–80% | 1.70–1.73 | 13.5–14 | tower acid Glover acid |
| 98% | 1.83 | ~18 | concentrated sulfuric acid |
"Chamber acid" and "tower acid" were the two concentrations of sulfuric acid produced by the lead chamber process, chamber acid being the acid produced in lead chamber itself (<70% to avoid contamination with nitrosylsulfuric acid) and tower acid being the acid recovered from the bottom of the Glover tower.[12][13] They are now obsolete as commercial concentrations of sulfuric acid, although they may be prepared in the laboratory from concentrated sulfuric acid if needed. In particular, "10M" sulfuric acid (the modern equivalent of chamber acid, used in many titrations) is prepared by slowly adding 98% sulfuric acid to an equal volume of water, with good stirring: the temperature of the mixture can rise to 80 °C (176 °F) or higher.[13]
Sulfuric acid reacts with its anhydride, SO
3, to form H
2S
2O
7, called pyrosulfuric acid, fuming sulfuric acid, Disulfuric acid or oleum or, less commonly, Nordhausen acid. Concentrations of oleum are either expressed in terms of % SO
3 (called % oleum) or as % H
2SO
4 (the amount made if H
2O were added); common concentrations are 40% oleum (109% H
2SO
4) and 65% oleum (114.6% H
2SO
4). Pure H
2S
2O
7 is a solid with melting point of 36 °C.
Pure sulfuric acid has a vapor pressure of <0 .001="" 145.8="" 1="" 25="" and="" at="" class="reference" id="cite_ref-OEHHA_14-0" nbsp="" sup="" torr="">[14]
Pure sulfuric acid is a viscous clear liquid, like oil, and this explains the old name of the acid ('oil of vitriol').
Commercial sulfuric acid is sold in several different purity grades. Technical grade H
2SO
4 is impure and often colored, but is suitable for making fertilizer. Pure grades, such as United States Pharmacopeia (USP) grade, are used for making pharmaceuticals and dyestuffs. Analytical grades are also available.
There are nine hydrates known, but three of them were confirmed to be tetrahydrate (H2SO4·4H2O), hemihexahydrate (H2SO4·6 1⁄2H2O) and octahydrate (H2SO4·8H2O).
Polarity and conductivity
| Species | mMol/kg |
|---|---|
| HSO− 4 |
15.0 |
| H 3SO+ 4 |
11.3 |
| H 3O+ |
8.0 |
| HS 2O− 7 |
4.4 |
| H 2S 2O 7 |
3.6 |
| H 2O |
0.1 |
2SO
4 is a very polar liquid, having a dielectric constant of around 100. It has a high electrical conductivity, caused by dissociation through protonating itself, a process known as autoprotolysis.[16]
- 2 H
2SO
4 H
H
3SO+
4 + HSO−
4
- Kap(25 °C)= [H
3SO+
4] [HSO−
4] = 2.7×10−4
In spite of the viscosity of the acid, the effective conductivities of the H
3SO+
4 and HSO−
4 ions are high due to an intra-molecular proton-switch mechanism (analogous to the Grotthuss mechanism in water), making sulfuric acid a good conductor of electricity. It is also an excellent solvent for many reactions.
Chemical properties
Reaction with water and dehydrating property
Because the hydration reaction of sulfuric acid is highly exothermic, dilution should always be performed by adding the acid to the water rather than the water to the acid.[17] Because the reaction is in an equilibrium that favors the rapid protonation of water, addition of acid to the water ensures that the acid is the limiting reagent. This reaction is best thought of as the formation of hydronium ions:
- H
2SO
4 + H
2O → H
3O+ + HSO4− K1 = 2.4×106 (strong acid)
- HSO−
4 + H
2O → H
3O+ + SO2−
4 K
2 = 1.0×10−2 [18]
4 is the bisulfate anion and SO2−
4 is the sulfate anion. K1 and K2 are the acid dissociation constants.
Because the hydration of sulfuric acid is thermodynamically favorable and the affinity of it for water is sufficiently strong, sulfuric acid is an excellent dehydrating agent. Concentrated sulfuric acid has a very powerful dehydrating property, removing water (H2O) from other compounds including sugar and other carbohydrates and producing carbon, heat, steam, and a more dilute acid containing increased amounts of hydronium and bisulfate ions.
In laboratory, this is often demonstrated by mixing table sugar (sucrose) into sulfuric acid. The sugar changes from white to dark brown and then to black as carbon is formed. A rigid column of black, porous carbon will emerge as well. The carbon will smell strongly of caramel due to the heat generated.[19]
- C12H22O11 (white sucrose) + sulfuric acid → 12 C(black graphitic foam) + 11 H2O (steam) + sulfuric acid/water mixture
- (C
6H
10O
5)n + sulfuric acid → 6n C + 5n H
2O
- CuSO4·5H2O (blue crystal) + sulfuric acid → CuSO4 (white powder) + 5 H2O
Acid-base properties
As an acid, sulfuric acid reacts with most bases to give the corresponding sulfate. For example, the blue copper salt copper(II) sulfate, commonly used for electroplating and as a fungicide, is prepared by the reaction of copper(II) oxide with sulfuric acid:- CuO (s) + H
2SO
4 (aq) → CuSO
4 (aq) + H
2O (l)
3COOH, and forms sodium bisulfate:
- H
2SO
4 + CH
3COONa → NaHSO
4 + CH
3COOH
2, which is important in nitration reactions involving electrophilic aromatic substitution. This type of reaction, where protonation occurs on an oxygen atom, is important in many organic chemistry reactions, such as Fischer esterification and dehydration of alcohols.

Solid state structure of the [D3SO4]+ ion present in [D3SO4]+[SbF6]−, synthesized by using DF in place of HF. (see text)
When allowed to react with superacids, sulfuric acid can act as a base and be protonated, forming the [H3SO4]+ ion. Salt of [H3SO4]+ have been prepared using the following reaction in liquid HF:
- ((CH3)3SiO)2SO2 + 3 HF + SbF5 → [H3SO4]+[SbF6]− + 2 (CH3)3SiF
- 2 H2SO4
 [H3O]+ + [HS2O7]−
[H3O]+ + [HS2O7]−
Reactions with metals and strong oxidizing property
Dilute sulfuric acid reacts with metals via a single displacement reaction as with other typical acids, producing hydrogen gas and salts (the metal sulfate). It attacks reactive metals (metals at positions above copper in the reactivity series) such as iron, aluminium, zinc, manganese, magnesium and nickel.- Fe (s) + H
2SO
4 (aq) → H
2 (g) + FeSO
4 (aq)
- 2 H2SO4 + 2 e− → SO2 + 2 H2O + SO42−
- Cu + 2 H2SO4 → SO2 + 2 H2O + SO42− + Cu2+
Reactions with non-metals
Hot concentrated sulfuric acid oxidizes non-metals such as carbon[21] and sulfur.- C + 2 H2SO4 → CO2 + 2 SO2 + 2 H2O
- S + 2 H2SO4 → 3 SO2 + 2 H2O
Reaction with sodium chloride
It reacts with sodium chloride, and gives hydrogen chloride gas and sodium bisulfate:- NaCl + H2SO4 → NaHSO4 + HCl
Electrophilic aromatic substitution
Benzene undergoes electrophilic aromatic substitution with sulfuric acid to give the corresponding sulfonic acids:[22]Occurrence
Pure sulfuric acid is not encountered naturally on Earth in anhydrous form, due to its great affinity for water. Dilute sulfuric acid is a constituent of acid rain, which is formed by atmospheric oxidation of sulfur dioxide in the presence of water – i.e., oxidation of sulfurous acid. Sulfur dioxide is the main byproduct produced when sulfur-containing fuels such as coal or oil are burned.
Sulfuric acid is formed naturally by the oxidation of sulfide minerals, such as iron sulfide. The resulting water can be highly acidic and is called acid mine drainage (AMD) or acid rock drainage (ARD). This acidic water is capable of dissolving metals present in sulfide ores, which results in brightly colored, toxic streams. The oxidation of pyrite (iron sulfide) by molecular oxygen produces iron(II), or Fe2+:
- 2 FeS
2 (s) + 7 O
2 + 2 H
2O → 2 Fe2+ (aq) + 4 SO2−
4 (aq) + 4 H+
- 4 Fe2+ + O
2 + 4 H+ → 4 Fe3+ + 2 H
2O
- Fe3+ (aq) + 3 H
2O → Fe(OH)
3 (s) + 3 H+
- FeS
2 (s) + 14 Fe3+ + 8 H
2O → 15 Fe2+ (aq) + 2 SO2−
4 (aq) + 16 H+
ARD can also produce sulfuric acid at a slower rate, so that the acid neutralizing capacity (ANC) of the aquifer can neutralize the produced acid. In such cases, the total dissolved solids (TDS) concentration of the water can be increased from the dissolution of minerals from the acid-neutralization reaction with the minerals.
Sulfuric acid is used as a defence by certain marine species, for example, the phaeophyte alga Desmarestia munda (order Desmarestiales) concentrates sulfuric acid in cell vacuoles.[23]
Extraterrestrial sulfuric acid
Venus
Sulfuric acid is produced in the upper atmosphere of Venus by the Sun's photochemical action on carbon dioxide, sulfur dioxide, and water vapor. Ultraviolet photons of wavelengths less than 169 nm can photodissociate carbon dioxide into carbon monoxide and atomic oxygen. Atomic oxygen is highly reactive. When it reacts with sulfur dioxide, a trace component of the Venusian atmosphere, the result is sulfur trioxide, which can combine with water vapor, another trace component of Venus's atmosphere, to yield sulfuric acid. In the upper, cooler portions of Venus's atmosphere, sulfuric acid exists as a liquid, and thick sulfuric acid clouds completely obscure the planet's surface when viewed from above. The main cloud layer extends from 45–70 km above the planet's surface, with thinner hazes extending as low as 30 km and as high as 90 km above the surface. The permanent Venusian clouds produce a concentrated acid rain, as the clouds in the atmosphere of Earth produce water rain.The atmosphere exhibits a sulfuric acid cycle. As sulfuric acid rain droplets fall down through the hotter layers of the atmosphere's temperature gradient, they are heated up and release water vapor, becoming more and more concentrated. When they reach temperatures above 300 °C, sulfuric acid begins to decompose into sulfur trioxide and water, both in the gas phase. Sulfur trioxide is highly reactive and dissociates into sulfur dioxide and atomic oxygen, which oxidizes traces of carbon monoxide to form carbon dioxide. Sulfur dioxide and water vapor rise on convection currents from the mid-level atmospheric layers to higher altitudes, where they will be transformed again into sulfuric acid, and the cycle repeats.
Europa
Infrared spectra from NASA's Galileo mission show distinct absorptions on Jupiter's moon Europa that have been attributed to one or more sulfuric acid hydrates. Sulfuric acid in solution with water causes significant freezing-point depression of water's melting point, down to 210 K (−63 °C), and this would make more likely the existence of liquid solutions beneath Europa's icy crust.The interpretation of the spectra is somewhat controversial.Some planetary scientists prefer to assign the spectral features to the sulfate ion, perhaps as part of one or more minerals on Europa's surface.[24]
Manufacture
Sulfuric acid is produced from sulfur, oxygen and water via the conventional contact process (DCDA) or the wet sulfuric acid process (WSA).Contact process
In the first step, sulfur is burned to produce sulfur dioxide.- S (s) + O
2 (g) → SO
2 (g)
- 2 SO
2 (g) + O
2 (g) 2 SO
2 SO
3 (g) (in presence of V
2O
5)
2SO
4 to form oleum (H
2S
2O
7), also known as fuming sulfuric acid.
The oleum is then diluted with water to form concentrated sulfuric acid.
- H
2SO
4 (l) + SO
3 (g)→ H
2S
2O
7 (l)
- H
2S
2O
7 (l) + H
2O (l) → 2 H
2SO
4 (l)
3 in water is not practical due to the highly exothermic nature of the reaction between sulfur trioxide and water. The reaction forms a corrosive aerosol that is very difficult to separate, instead of a liquid.
- SO
3 (g) + H
2O (l) → H
2SO
4 (l)
Wet sulfuric acid process
In the first step, sulfur is burned to produce sulfur dioxide:- S(s) + O
2(g) → SO
2(g)
2S) gas is incinerated to SO
2 gas:
- 2 H
2S + 3 O
2 → 2 H
2O + 2 SO
2 (−518 kJ/mol)
- 2 SO
2 + O
2 → 2 SO
3 (−99 kJ/mol) (reaction is reversible)
2SO
4:
- SO
3 + H
2O → H
2SO
4(g) (−101 kJ/mol)
2SO
4:
- H
2SO
4(g) → H
2SO
4(l) (−69 kJ/mol)
Other methods
Another method is the less well-known metabisulfite method, in which metabisulfite is placed at the bottom of a beaker, and 12.6 molar concentration hydrochloric acid is added. The resulting gas is bubbled through nitric acid, which will release brown/red vapors. The completion of the reaction is indicated by the ceasing of the fumes. This method does not produce an inseparable mist, which is quite convenient.Sulfuric acid can be produced in the laboratory by burning sulfur in air and dissolving the gas produced in a hydrogen peroxide solution.
- SO2 + H2O2 → H2SO4
In early to mid nineteenth century "vitriol" plants existed, among other places, in Prestonpans in Scotland, Shropshire and the Lagan Valley in County Antrim Ireland where it was used as a bleach for linen. Early bleaching of linen was done using milk but this was a slow process and the use of vitriol sped up the bleaching process.
Uses
Sulfuric acid is a very important commodity chemical, and indeed, a nation's sulfuric acid production is a good indicator of its industrial strength.[26] World production in 2004 was about 180 million tonnes, with the following geographic distribution: Asia 35%, North America (including Mexico) 24%, Africa 11%, Western Europe 10%, Eastern Europe and Russia 10%, Australia and Oceania 7%, South America 7%.[27] Most of this amount (~60%) is consumed for fertilizers, particularly superphosphates, ammonium phosphate and ammonium sulfates. About 20% is used in chemical industry for production of detergents, synthetic resins, dyestuffs, pharmaceuticals, petroleum catalysts, insecticides and antifreeze, as well as in various processes such as oil well acidicizing, aluminium reduction, paper sizing, water treatment. About 6% of uses are related to pigments and include paints, enamels, printing inks, coated fabrics and paper, and the rest is dispersed into a multitude of applications such as production of explosives, cellophane, acetate and viscose textiles, lubricants, non-ferrous metals and batteries.[28]
Industrial production of chemicals
The major use for sulfuric acid is in the "wet method" for the production of phosphoric acid, used for manufacture of phosphate fertilizers. In this method, phosphate rock is used, and more than 100 million tonnes are processed annually. This raw material is shown below as fluorapatite, though the exact composition may vary. This is treated with 93% sulfuric acid to produce calcium sulfate, hydrogen fluoride (HF) and phosphoric acid. The HF is removed as hydrofluoric acid. The overall process can be represented as:- Ca
5F(PO
4)
3 + 5 H
2SO
4 + 10 H
2O → 5 CaSO
4·2 H
2O + HF + 3 H
3PO
4
Another important use for sulfuric acid is for the manufacture of aluminium sulfate, also known as paper maker's alum. This can react with small amounts of soap on paper pulp fibers to give gelatinous aluminium carboxylates, which help to coagulate the pulp fibers into a hard paper surface. It is also used for making aluminium hydroxide, which is used at water treatment plants to filter out impurities, as well as to improve the taste of the water. Aluminium sulfate is made by reacting bauxite with sulfuric acid:
- 2 AlO(OH) + 3 H
2SO
4 → Al
2(SO
4)
3 + 4 H
2O
Sulfur-iodine cycle
The sulfur-iodine cycle is a series of thermo-chemical processes used to obtain hydrogen. It consists of three chemical reactions whose net reactant is water and whose net products are hydrogen and oxygen.2 H
2SO
4 → 2 SO
2 + 2 H
2O + O
2(830 °C) I
2 + SO
2 + 2 H
2O → 2 HI + H
2SO
4(120 °C) 2 HI → I
2 + H
2(320 °C)
The sulfur-iodine cycle has been proposed as a way to supply hydrogen for a hydrogen-based economy. It does not require hydrocarbons like current methods of steam reforming. But note that all of the available energy in the hydrogen so produced is supplied by the heat used to make it.
The sulfur-iodine cycle is currently being researched as a feasible method of obtaining hydrogen, but the concentrated, corrosive acid at high temperatures poses currently insurmountable safety hazards if the process were built on a large scale.
Industrial cleaning agent
Sulfuric acid is used in large quantities by the iron and steelmaking industry to remove oxidation, rust and scaling from rolled sheet and billets prior to sale to the automobile and major appliances industry.[citation needed] Used acid is often recycled using a spent acid regeneration (SAR) plant. These plants combust spent acid[clarification needed] with natural gas, refinery gas, fuel oil or other fuel sources. This combustion process produces gaseous sulfur dioxide (SO2) and sulfur trioxide (SO3) which are then used to manufacture "new" sulfuric acid. SAR plants are common additions to metal smelting plants, oil refineries, and other industries where sulfuric acid is consumed in bulk, as operating a SAR plant is much cheaper than the recurring costs of spent acid disposal and new acid purchases.Catalyst
Sulfuric acid is used for a variety of other purposes in the chemical industry. For example, it is the usual acid catalyst for the conversion of cyclohexanone oxime to caprolactam, used for making nylon. It is used for making hydrochloric acid from salt via the Mannheim process. Much H2SO
4 is used in petroleum refining, for example as a catalyst for the reaction of isobutane with isobutylene to give isooctane, a compound that raises the octane rating of gasoline (petrol).
Electrolyte

Acidic drain cleaners usually contain sulfuric acid at a high concentration which turns a piece of pH paper red and chars it instantly, demonstrating both the strong acidic nature and dehydrating property.
Sulfuric acid acts as the electrolyte in lead-acid (car) batteries (lead-acid accumulator):
At anode:
- Pb + SO
42− ⇌ PbSO
4 + 2 e−
- PbO
2 + 4 H+ + SO
42− + 2 e− ⇌ PbSO
4 + 2 H2O

An acidic drain cleaner can be used to dissolve grease, hair and even tissue paper inside water pipes.
Overall:
- Pb + PbO
2 + 4 H+ + 2 SO
42− ⇌ 2 PbSO
4 + 2 H2O
Domestic uses
Sulfuric acid at high concentrations is frequently the major ingredient in acidic drain cleaners[10] which are used to remove grease, hair, tissue paper, etc. Similar to their alkaline versions, such drain openers can dissolve fats and proteins via hydrolysis. Moreover, as concentrated sulfuric acid has a strong dehydrating property, it can remove tissue paper via dehydrating process as well. Since the acid may react with water vigorously, such acidic drain openers should be added slowly into the pipe to be cleaned.Health
Sulfuric acid and sulfonated phenolics are the primary ingredients in Debacterol, a liquid topical agent that is used in the treatment of recurrent aphthous stomatitis (canker sores/mouth ulcers) or for any procedures in the oral cavity which require controlled, focal debridement of necrotic tissues.[29]Safety
Laboratory hazards
Sulfuric acid is capable of causing very severe burns, especially when it is at high concentrations. In common with other corrosive acids and alkali, it readily decomposes proteins and lipids through amide and ester hydrolysis upon contact with living tissues, such as skin and flesh. In addition, it exhibits a strong dehydrating property on carbohydrates, liberating extra heat and causing secondary thermal burns.[9][8] Accordingly, it rapidly attacks the cornea and can induce permanent blindness if splashed onto eyes. If ingested, it damages internal organs irreversibly and may even be fatal.[5] Protective equipment should hence always be used when handling it. Moreover, its strong oxidizing property makes it highly corrosive to many metals and may extend its destruction on other materials.[8] Because of such reasons, damage posed by sulfuric acid is potentially more severe than that by other comparable strong acids, such as hydrochloric acid and nitric acid.
Sulfuric acid must be stored carefully in containers made of nonreactive material (such as glass). Solutions equal to or stronger than 1.5 M are labeled "CORROSIVE", while solutions greater than 0.5 M but less than 1.5 M are labeled "IRRITANT". However, even the normal laboratory "dilute" grade (approximately 1 M, 10%) will char paper if left in contact for a sufficient time.
The standard first aid treatment for acid spills on the skin is, as for other corrosive agents, irrigation with large quantities of water. Washing is continued for at least ten to fifteen minutes to cool the tissue surrounding the acid burn and to prevent secondary damage. Contaminated clothing is removed immediately and the underlying skin washed thoroughly.
Preparation of the diluted acid can also be dangerous due to the heat released in the dilution process. The concentrated acid is always added to water and not the other way around, to take advantage of the relatively high heat capacity of water. Addition of water to concentrated sulfuric acid leads to the dispersal of a sulfuric acid aerosol or worse, an explosion. Preparation of solutions greater than 6 M (35%) in concentration is most dangerous, as the heat produced may be sufficient to boil the diluted acid: efficient mechanical stirring and external cooling (such as an ice bath) are essential.
On a laboratory scale, sulfuric acid can be diluted by pouring concentrated acid onto crushed ice made from de-ionized water. The ice melts in an endothermic process while dissolving the acid. The amount of heat needed to melt the ice in this process is greater than the amount of heat evolved by dissolving the acid so the solution remains cold. After all the ice has melted, further dilution can take place using water.
Industrial hazards
Although sulfuric acid is non-flammable, contact with metals in the event of a spillage can lead to the liberation of hydrogen gas. The dispersal of acid aerosols and gaseous sulfur dioxide is an additional hazard of fires involving sulfuric acid.The main occupational risks posed by this acid are skin contact leading to burns (see above) and the inhalation of aerosols. Exposure to aerosols at high concentrations leads to immediate and severe irritation of the eyes, respiratory tract and mucous membranes: this ceases rapidly after exposure, although there is a risk of subsequent pulmonary edema if tissue damage has been more severe. At lower concentrations, the most commonly reported symptom of chronic exposure to sulfuric acid aerosols is erosion of the teeth, found in virtually all studies: indications of possible chronic damage to the respiratory tract are inconclusive as of 1997. In the United States, the permissible exposure limit (PEL) for sulfuric acid is fixed at 1 mg/m3: limits in other countries are similar. There have been reports of sulfuric acid ingestion leading to vitamin B12 deficiency with subacute combined degeneration. The spinal cord is most often affected in such cases, but the optic nerves may show demyelination, loss of axons and gliosis.