From Wikipedia, the free encyclopedia
In 1897, Thomson showed that cathode rays were composed of previously unknown negatively charged particles, which he calculated must have bodies much smaller than atoms and a very large value for their charge-to-mass ratio.[3] Thus he is credited with the discovery and identification of the electron; and with the discovery of the first subatomic particle. Thomson is also credited with finding the first evidence for isotopes of a stable (non-radioactive) element in 1913, as part of his exploration into the composition of canal rays (positive ions). His experiments to determine the nature of positively charged particles, with Francis William Aston, were the first use of mass spectrometry and led to the development of the mass spectrograph.[3]
Thomson was awarded the 1906 Nobel Prize in Physics for the discovery of the electron and for his work on the conduction of electricity in gases.[4] Seven of his students, and his son George Paget Thomson, also became Nobel Prize winners.
Biography
Joseph John Thomson was born 18 December 1856 in Cheetham Hill, Manchester, Lancashire, England. His mother, Emma Swindells, came from a local textile family. His father, Joseph James Thomson, ran an antiquarian bookshop founded by a great-grandfather. He had a brother two years younger than he was, Frederick Vernon Thomson.[5]His early education was in small private schools where he demonstrated outstanding talent and interest in science. In 1870 he was admitted to Owens College at the unusually young age of 14. His parents planned to enroll him as an apprentice engineer to Sharp-Stewart & Co, a locomotive manufacturer, but these plans were cut short when his father died in 1873.[5]
He moved on to Trinity College, Cambridge in 1876. In 1880, he obtained his BA in mathematics (Second Wrangler in the Tripos[6] and 2nd Smith's Prize).[7] He applied for and became a Fellow of Trinity College as of 1881.[8] Thomson received his MA (with Adams Prize) in 1883.[7]
Thomson was elected a Fellow of the Royal Society[1] on 12 June 1884 and served as President of the Royal Society from 1915 to 1920.
On 22 December 1884 Thomson was chosen to become Cavendish Professor of Physics at the University of Cambridge.[3] The appointment caused considerable surprise, given that candidates such as Richard Glazebrook were older and more experienced in laboratory work. Thomson was known for his work as a mathematician, where he was recognized as an exceptional talent.[9]
In 1890, Thomson married Rose Elisabeth Paget, daughter of Sir George Edward Paget, KCB, a physician and then Regius Professor of Physic at Cambridge. They had one son, George Paget Thomson, and one daughter, Joan Paget Thomson.
He was awarded a Nobel Prize in 1906, "in recognition of the great merits of his theoretical and experimental investigations on the conduction of electricity by gases." He was knighted in 1908 and appointed to the Order of Merit in 1912. In 1914 he gave the Romanes Lecture in Oxford on "The atomic theory". In 1918 he became Master of Trinity College, Cambridge, where he remained until his death. Joseph John Thomson died on 30 August 1940 and was buried in Westminster Abbey, close to Sir Isaac Newton.
One of Thomson's greatest contributions to modern science was in his role as a highly gifted teacher. One of his students was Ernest Rutherford, who later succeeded him as Cavendish Professor of Physics. In addition to Thomson himself, seven of his research assistants and his son won Nobel Prizes in physics. His son won the Nobel Prize in 1937 for proving the wavelike properties of electrons.
Career
Early work
Thomson's prize-winning master's work, Treatise on the motion of vortex rings, shows his early interest in atomic structure.[4] In it, Thomson mathematically described the motions of William Thomson's vortex theory of atoms.[9]Thomson published a number of papers addressing both mathematical and experimental issues of electromagnetism. He examined the electromagnetic theories of light of James Clerk Maxwell, introduced the concept of electromagnetic mass of a charged particle, and demonstrated that a moving charged body would apparently increase in mass.[9]
Much of his work in mathematical modelling of chemical processes can be thought of as early computational chemistry.[3] In further work, published in book form as Applications of dynamics to physics and chemistry (1888), Thomson addressed the transformation of energy in mathematical and theoretical terms, suggesting that all energy might be kinetic.[9] His next book, Notes on recent researches in electricity and magnetism (1893), built upon Maxwell's Treatise upon electricity and magnetism, and was sometimes referred to as "the third volume of Maxwell".[4] In it, Thomson emphasized physical methods and experimentation and included extensive figures and diagrams of apparatus, including a number for the passage of electricity through gases.[9] His third book, Elements of Mathematical theory of electricity and magnetism (1895) was a readable introduction to a wide variety of subjects, and achieved considerable popularity as a textbook.[9]
A series of four lectures, given by Thomson on a visit to Princeton University in 1896, were subsequently published as Discharge of electricity through gases (1897). Thomson also presented a series of six lectures at Yale University in 1904.[4]
Discovery of the electron
Several scientists, such as William Prout and Norman Lockyer, had suggested that atoms were built up from a more fundamental unit, but they envisioned this unit to be the size of the smallest atom, hydrogen. Thomson, in 1897, was the first to suggest that the fundamental unit was more than 1,000 times smaller than an atom, suggesting the subatomic particle now known as the electron. Thomson discovered this through his explorations on the properties of cathode rays. Thomson made his suggestion on 30 April 1897 following his discovery that Lenard rays could travel much further through air than expected for an atom-sized particle.[10] He estimated the mass of cathode rays by measuring the heat generated when the rays hit a thermal junction and comparing this with the magnetic deflection of the rays. His experiments suggested not only that cathode rays were over 1,000 times lighter than the hydrogen atom, but also that their mass was the same in whichever type of atom they came from. He concluded that the rays were composed of very light, negatively charged particles which were a universal building block of atoms. He called the particles "corpuscles", but later scientists preferred the name electron which had been suggested by George Johnstone Stoney in 1891, prior to Thomson's actual discovery.[11]In April 1897, Thomson had only early indications that the cathode rays could be deflected electrically (previous investigators such as Heinrich Hertz had thought they could not be). A month after Thomson's announcement of the corpuscle he found that he could reliably deflect the rays by an electric field if he evacuated the discharge tube to a very low pressure. By comparing the deflection of a beam of cathode rays by electric and magnetic fields he obtained more robust measurements of the mass to charge ratio that confirmed his previous estimates.[12] This became the classic means of measuring the charge and mass of the electron.
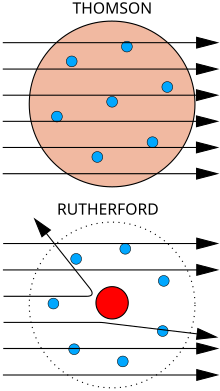
Thomson believed that the corpuscles emerged from the atoms of the trace gas inside his cathode ray tubes. He thus concluded that atoms were divisible, and that the corpuscles were their building blocks. In 1904 Thomson suggested a model of the atom, hypothesizing that it was a sphere of positive matter within which electrostatic forces determined the positioning of the corpuscles.[3] To explain the overall neutral charge of the atom, he proposed that the corpuscles were distributed in a uniform sea of positive charge. In this "plum pudding" model the electrons were seen as embedded in the positive charge like plums in a plum pudding (although in Thomson's model they were not stationary, but orbiting rapidly).[13][14]
Isotopes and mass spectrometry
In 1912, as part of his exploration into the composition of canal rays, Thomson and his research assistant F. W. Aston channelled a stream of neon ions through a magnetic and an electric field and measured its deflection by placing a photographic plate in its path.[5] They observed two patches of light on the photographic plate (see image on right), which suggested two different parabolas of deflection, and concluded that neon is composed of atoms of two different atomic masses (neon-20 and neon-22), that is to say of two isotopes.[15] This was the first evidence for isotopes of a stable element; Frederick Soddy had previously proposed the existence of isotopes to explain the decay of certain radioactive elements.
J.J. Thomson's separation of neon isotopes by their mass was the first example of mass spectrometry, which was subsequently improved and developed into a general method by F. W. Aston and by A. J. Dempster.[3]
Other work
In 1905, Thomson discovered the natural radioactivity of potassium.[16]In 1906, Thomson demonstrated that hydrogen had only a single electron per atom. Previous theories allowed various numbers of electrons.[17][18]
Experiments with cathode rays
Earlier, physicists debated whether cathode rays were immaterial like light ("some process in the aether") or were "in fact wholly material, and ... mark the paths of particles of matter charged with negative electricity", quoting Thomson.[12] The aetherial hypothesis was vague,[12] but the particle hypothesis was definite enough for Thomson to test.Experiments on the magnetic deflection of cathode rays
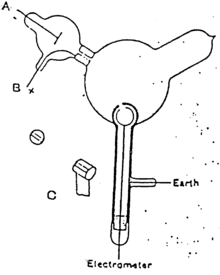
Thomson first investigated the magnetic deflection of cathode rays. Cathode rays were produced in the side tube on the left of the apparatus and passed through the anode into the main bell jar, where they were deflected by a magnet. Thomson detected their path by the fluorescence on a squared screen in the jar. He found that whatever the material of the anode and the gas in the jar, the deflection of the rays was the same, suggesting that the rays were of the same form whatever their origin.[19]Experiment to show that cathode rays were electrically charged
While supporters of the aetherial theory accepted the possibility that negatively charged particles are produced in Crookes tubes[citation needed], they believed that they are a mere by-product and that the cathode rays themselves are immaterial[citation needed]. Thomson set out to investigate whether or not he could actually separate the charge from the rays.
Thomson constructed a Crookes tube with an electrometer set to one side, out of the direct path of the cathode rays. Thomson could trace the path of the ray by observing the phosphorescent patch it created where it hit the surface of the tube. Thomson observed that the electrometer registered a charge only when he deflected the cathode ray to it with a magnet. He concluded that the negative charge and the rays were one and the same.[10]
Experiment to show that cathode rays could be deflected electrically
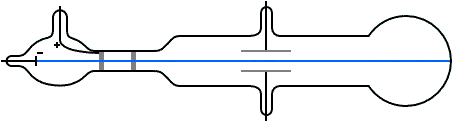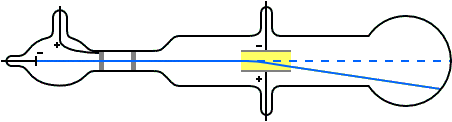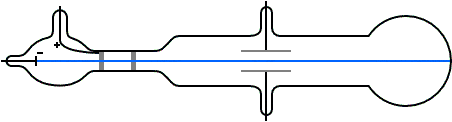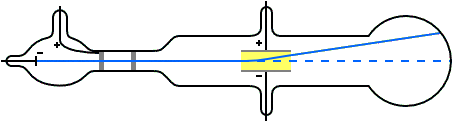
Thomson's illustration of the Crookes tube by which he observed the deflection of cathode rays by an electric field (and later measured their mass to charge ratio). Cathode rays were emitted from the cathode C, passed through slits A (the anode) and B (grounded), then through the electric field generated between plates D and E, finally impacting the surface at the far end.
In May–June 1897, Thomson investigated whether or not the rays could be deflected by an electric field.[5] Previous experimenters had failed to observe this, but Thomson believed their experiments were flawed because their tubes contained too much gas.
Thomson constructed a Crookes tube with a better vacuum. At the start of the tube was the cathode from which the rays projected. The rays were sharpened to a beam by two metal slits – the first of these slits doubled as the anode, the second was connected to the earth. The beam then passed between two parallel aluminium plates, which produced an electric field between them when they were connected to a battery. The end of the tube was a large sphere where the beam would impact on the glass, created a glowing patch. Thomson pasted a scale to the surface of this sphere to measure the deflection of the beam. Note that any electron beam would collide with some residual gas atoms within the Crookes tube, thereby ionizing them and producing electrons and ions in the tube (space charge); in previous experiments this space charge electrically screened the externally applied electric field. However, in Thomson's Crookes tube the density of residual atoms was so low that the space charge from the electrons and ions was insufficient to electrically screen the externally applied electric field, which permitted Thomson to successfully observe electrical deflection.
When the upper plate was connected to the negative pole of the battery and the lower plate to the positive pole, the glowing patch moved downwards, and when the polarity was reversed, the patch moved upwards.
Experiment to measure the mass to charge ratio of cathode rays
In his classic experiment, Thomson measured the mass-to-charge ratio of the cathode rays by measuring how much they were deflected by a magnetic field and comparing this with the electric deflection. He used the same apparatus as in his previous experiment, but placed the discharge tube between the poles of a large electromagnet. He found that the mass to charge ratio was over a thousand times lower than that of a hydrogen ion (H+), suggesting either that the particles were very light and/or very highly charged.[12] Significantly, the rays from every cathode yielded the same mass-to-charge ratio. This is in contrast to anode rays (now known to arise from positive ions emitted by the anode), where the mass-to-charge ratio varies from anode-to-anode. Thomson himself remained critical of what his work established, in his Nobel Prize acceptance speech referring to "corpuscles" rather than "electrons".
Thomson's calculations can be summarised as follows (notice that we reproduce here Thomson's original notations, using F instead of E for the Electric field and H instead of B for the magnetic field):
The electric deflection is given by Θ = Fel/mv2 where Θ is the angular electric deflection, F is applied electric intensity, e is the charge of the cathode ray particles, l is the length of the electric plates, m is the mass of the cathode ray particles and v is the velocity of the cathode ray particles.
The magnetic deflection is given by φ = Hel/mv where φ is the angular magnetic deflection and H is the applied magnetic field intensity.
The magnetic field was varied until the magnetic and electric deflections were the same, when Θ = φ and Fel/mv2= Hel/mv. This can be simplified to give m/e = H2l/FΘ. The electric deflection was measured separately to give Θ and H, F and l were known, so m/e could be calculated.
Conclusions
As the cathode rays carry a charge of negative electricity, are deflected by an electrostatic force as if they were negatively electrified, and are acted on by a magnetic force in just the way in which this force would act on a negatively electrified body moving along the path of these rays, I can see no escape from the conclusion that they are charges of negative electricity carried by particles of matter.As to the source of these particles, Thomson believed they emerged from the molecules of gas in the vicinity of the cathode.
—J. J. Thomson[12]
If, in the very intense electric field in the neighbourhood of the cathode, the molecules of the gas are dissociated and are split up, not into the ordinary chemical atoms, but into these primordial atoms, which we shall for brevity call corpuscles; and if these corpuscles are charged with electricity and projected from the cathode by the electric field, they would behave exactly like the cathode rays.Thomson imagined the atom as being made up of these corpuscles orbiting in a sea of positive charge; this was his plum pudding model. This model was later proved incorrect when his student Ernest Rutherford showed that the positive charge is concentrated in the nucleus of the atom.
—J. J. Thomson[20]
Awards and recognition
- Adams Prize (1882)
- Royal Medal (1894)
- Hughes Medal (1902)
- Nobel Prize for Physics (1906)
- Elliott Cresson Medal (1910)
- Copley Medal (1914)
- Franklin Medal (1922)
In November 1927, J.J. Thomson opened the self-titled Thomson building in the Leys School, Cambridge.[23]