Model of the antioxidant
metabolite glutathione. The yellow sphere is the
redox-active sulfur atom that provides antioxidant activity, while the red, blue, white, and dark grey spheres represent oxygen, nitrogen, hydrogen, and carbon atoms, respectively.
An
antioxidant is a
molecule that inhibits the
oxidation of other molecules. Oxidation is a
chemical reaction involving the loss of electrons or an increase in oxidation state. Oxidation reactions can produce
free radicals. In turn, these radicals can start
chain reactions. When the chain reaction occurs in a
cell, it can cause damage or death to the cell. Antioxidants terminate these chain reactions by removing free radical intermediates, and inhibit other oxidation reactions. They do this by being oxidized themselves, so antioxidants are often
reducing agents such as
thiols,
ascorbic acid (vitamin C), or
polyphenols.
[1]

Substituted
phenols and derivatives of
phenylenediamine are common antioxidants used to inhibit gum formation in gasoline (petrol).
Although oxidation reactions are crucial for life, they can also be damaging; plants and animals maintain complex systems of multiple types of antioxidants, such as
glutathione,
vitamin C,
vitamin A, and
vitamin E as well as
enzymes such as
catalase,
superoxide dismutase and various
peroxidases. Insufficient levels of antioxidants, or
inhibition of the antioxidant enzymes, cause
oxidative stress and may damage or kill cells. Oxidative stress is damage to cell structure and cell function by overly reactive oxygen-containing molecules and chronic excessive inflammation. Oxidative stress seems to play a significant role in many human diseases, including cancers. The use of antioxidants in
pharmacology is intensively studied, particularly as treatments for stroke and
neurodegenerative diseases. For these reasons, oxidative stress can be considered to be both the cause and the consequence of some diseases.
Antioxidants are widely used in
dietary supplements and have been investigated for the prevention of diseases such as cancer,
coronary heart disease and
altitude sickness.
[2] Although initial studies suggested that antioxidant supplements might promote health, later large
trials including of beta-carotene, vitamin A, and vitamin E singly or in different combinations suggest that supplementation has no effect on mortality or possibly increases it.
[3][4][5] Randomized trials of taking antioxidants including beta carotene, vitamin E, vitamin C and selenium have shown no effect on cancer risk or have increased cancer risk.
[6][7][8][9][10][11][12] Supplementation with selenium or vitamin E does not reduce the risk of cardiovascular disease.
[13][14]
Antioxidants also have many industrial uses, such as
preservatives in food and cosmetics and to prevent the degradation of rubber and gasoline.
[15]
Health effects
Pharmaceuticals
Tirilazad mesylate is an anti-oxidant steroid derivative that inhibits the lipid peroxidation that is believed to play a key in neuronal death in stroke and head injury. It demonstrated activity in animal models of stroke.
[16] Human trials demonstrated no effect on mortality or other outcomes in subarachnoid haemorrhage,
[17] and worsened results in ischemic stroke.
[18]
Similarly, the designed antioxidant NXY-059 exhibited efficacy in animal models, but failed to improve stroke outcomes in a clinical trial.
[19] As of November 2014, other antioxidants are being studied as potential neuroprotectants.
[20]
Common pharmaceuticals (and supplements) with antioxidant properties may interfere with the efficacy of certain anticancer medication and radiation.
[21][22]
Relation to diet

Structure of
resveratrol under study for its potential as a dietary antioxidant
Although some levels of antioxidant vitamins in the diet are required for good health, there is considerable doubt as to whether antioxidant supplements have anti-disease activity; and if they are actually beneficial, which antioxidant(s) are needed and in what amounts.
[23][24][25] Indeed, some authors argue that the hypothesis that antioxidants could prevent chronic diseases
[23][26] has now been disproved and that the idea was misguided from the beginning.
[27] Rather, dietary
polyphenols may have non-antioxidant roles in minute concentrations that affect cell-to-cell signaling,
receptor sensitivity, inflammatory
enzyme activity or
gene regulation.
[28][29]
For overall
life expectancy, it has even been suggested that moderate levels of oxidative stress may increase lifespan in the worm
Caenorhabditis elegans, by inducing a protective response to increased levels of reactive oxygen species.
[30] The suggestion that increased life expectancy comes from increased oxidative stress conflicts with results seen in the yeast
Saccharomyces cerevisiae,
[31] and the situation in mammals is even less clear.
[32][33][34] Nevertheless, antioxidant supplements do not appear to increase life expectancy in humans.
[35]
Although antioxidants have been investigated for potential effects on
neurodegenerative diseases such as
Alzheimer's disease,
Parkinson's disease, and
amyotrophic lateral sclerosis,
[36][37] these studies have been inconclusive.
[38][39][40]
Physical exercise
During exercise, oxygen consumption can increase by a factor of more than 10.
[41] This leads to a large increase in the production of oxidants and results in damage that contributes to muscular fatigue during and after exercise. The
inflammatory response that occurs after strenuous exercise is also associated with oxidative stress, especially in the 24 hours after an exercise session. The immune system response to the damage done by exercise peaks 2 to 7 days after exercise, which is the period during which most of the adaptation that leads to greater fitness occurs. During this process, free radicals are produced by
neutrophils to remove damaged tissue. As a result, excessive antioxidant levels may inhibit recovery and adaptation mechanisms.
[42]
The evidence for benefits from antioxidant supplementation in vigorous exercise is mixed. There is strong evidence that one of the adaptations resulting from exercise is a strengthening of the body's antioxidant defenses, particularly the glutathione system, to regulate the increased oxidative stress.
[43] This effect may be to some extent protective against diseases which are associated with oxidative stress, which would provide a partial explanation for the lower incidence of major diseases and better health of those who undertake regular exercise.
[44]
No benefits for physical performance to athletes are seen with vitamin E supplementation.
[45] Indeed, despite its key role in preventing
lipid membrane peroxidation, 6 weeks of vitamin E supplementation had no effect on muscle damage in ultramarathon runners.
[46] Although there appears to be no increased requirement for vitamin C in athletes, there is some evidence that vitamin C supplementation increased the amount of intense exercise that can be done and vitamin C supplementation before strenuous exercise may reduce the amount of muscle damage.
[47][48] Other studies found no such effects, and some research suggests that supplementation with amounts as high as
1000 mg inhibits recovery.
[49]
A review published in Sports Medicine looked at 150 studies on antioxidant supplementation during exercise. The review found that even studies that found a reduction in oxidative stress failed to demonstrate benefits to performance or prevention of muscle damage. Some studies indicated that antioxidant supplementation could work against the cardiovascular benefits of exercise.
[50]
Adverse effects
Relatively strong reducing acids can have
antinutrient effects by binding to
dietary minerals such as iron and zinc in the
gastrointestinal tract and preventing them from being absorbed.
[51] Notable examples are
oxalic acid,
tannins and
phytic acid, which are high in plant-based diets.
[52] Calcium and iron deficiencies are not uncommon in diets in
developing countries where less meat is eaten and there is high consumption of phytic acid from beans and unleavened
whole grain bread.
[53]
Nonpolar antioxidants such as
eugenol—a major component of
oil of cloves—have toxicity limits that can be exceeded with the misuse of undiluted
essential oils.
[57] Toxicity associated with high doses of water-soluble antioxidants such as ascorbic acid are less of a concern, as these compounds can be excreted rapidly in
urine.
[58] More seriously, very high doses of some antioxidants may have harmful long-term effects. The beta-Carotene and Retinol Efficacy Trial (CARET) study of lung cancer patients found that smokers given supplements containing beta-carotene and vitamin A had increased rates of lung cancer.
[59] Subsequent studies confirmed these adverse effects.
[60]
These harmful effects may also be seen in non-smokers, as a recent
meta-analysis including data from approximately 230,000 patients showed that β-carotene, vitamin A or vitamin E supplementation is associated with increased mortality but saw no significant effect from vitamin C.
[61] No health risk was seen when all the randomized controlled studies were examined together, but an increase in mortality was detected when only high-quality and low-bias risk trials were examined separately.
[62] As the majority of these low-bias trials dealt with either
elderly people, or people with disease, these results may not apply to the general population.
[63] This meta-analysis was later repeated and extended by the same authors, with the new analysis published by the
Cochrane Collaboration; confirming the previous results.
[62] These two publications are consistent with some previous meta-analyzes that also suggested that Vitamin E supplementation increased mortality,
[64] and that antioxidant supplements increased the risk of
colon cancer.
[65] Beta-carotene may also increase lung cancer.
[65][66] Overall, the large number of clinical trials carried out on antioxidant supplements suggest that either these products have no effect on health, or that they cause a small increase in mortality in elderly or vulnerable populations.
[23][24][61]
While antioxidant supplementation is widely used in attempts to prevent the development of cancer, antioxidants may interfere with cancer treatments,
[67] since the environment of cancer cells causes high levels of oxidative stress, making these cells more susceptible to the further oxidative stress induced by treatments. As a result, by reducing the redox stress in cancer cells, antioxidant supplements (and pharmaceuticals) could decrease the effectiveness of
radiotherapy and
chemotherapy.
[21][68][69] On the other hand, other reviews have suggested that antioxidants could reduce
side effects or increase survival times.
[70][71]
Uses in technology
Food preservatives
Antioxidants are used as
food additives to help
guard against food deterioration. Exposure to oxygen and sunlight are the two main factors in the oxidation of food, so food is preserved by keeping in the dark and sealing it in containers or even coating it in wax, as with cucumbers. However, as oxygen is also important for plant
respiration, storing plant materials in
anaerobic conditions produces unpleasant flavors and unappealing colors.
[72] Consequently, packaging of fresh fruits and vegetables contains an ~8% oxygen atmosphere. Antioxidants are an especially important class of preservatives as, unlike bacterial or
fungal spoilage, oxidation reactions still occur relatively rapidly in frozen or refrigerated food.
[73] These preservatives include natural antioxidants such as ascorbic acid (AA, E300) and tocopherols (E306), as well as synthetic antioxidants such as
propyl gallate (PG, E310),
tertiary butylhydroquinone (TBHQ),
butylated hydroxyanisole (BHA, E320) and
butylated hydroxytoluene (BHT, E321).
[74][75]
The most common molecules attacked by oxidation are unsaturated fats; oxidation causes them to turn
rancid.
[76] Since oxidized lipids are often discolored and usually have unpleasant tastes such as metallic or
sulfurous flavors, it is important to avoid oxidation in fat-rich foods. Thus, these foods are rarely preserved by drying; instead, they are preserved by
smoking,
salting or
fermenting. Even less fatty foods such as fruits are sprayed with sulfurous antioxidants prior to air drying. Oxidation is often catalyzed by metals, which is why fats such as butter should never be wrapped in
aluminium foil or kept in metal containers. Some fatty foods such as olive oil are partially protected from oxidation by their natural content of antioxidants, but remain sensitive to photooxidation.
[77] Antioxidant preservatives are also added to fat based cosmetics such as lipstick and
moisturizers to prevent rancidity.
Industrial uses
Antioxidants are frequently added to industrial products. A common use is as
stabilizers in
fuels and
lubricants to prevent oxidation, and in gasolines to prevent the polymerization that leads to the formation of engine-fouling residues.
[78] In 2007, the worldwide market for industrial antioxidants had a total volume of around 0.88 million tons. This created a revenue of circa 3.7 billion US-dollars (2.4 billion Euros).
[79]
They are widely used to prevent the oxidative degradation of
polymers such as rubbers, plastics and
adhesives that causes a loss of strength and flexibility in these materials.
[80] Polymers containing
double bonds in their main chains, such as
natural rubber and
polybutadiene, are especially susceptible to
oxidation and
ozonolysis. They can be protected by
antiozonants. Solid polymer products start to crack on exposed surfaces as the material degrades and the chains break. The mode of cracking varies between oxygen and
ozone attack, the former causing a "crazy paving" effect, while ozone attack produces deeper cracks aligned at right angles to the tensile strain in the product. Oxidation and
UV degradation are also frequently linked, mainly because
UV radiation creates free radicals by bond breakage. The free radicals then react with oxygen to produce
peroxy radicals which cause yet further damage, often in a
chain reaction. Other polymers susceptible to oxidation include
polypropylene and
polyethylene. The former is more sensitive owing to the presence of
secondary carbon atoms present in every repeat unit. Attack occurs at this point because the free radical formed is more stable than one formed on a
primary carbon atom. Oxidation of polyethylene tends to occur at weak links in the chain, such as branch points in
low density polyethylene.
| Fuel additive |
Components[81] |
Applications[81] |
| AO-22 |
N,N'-di-2-butyl-1,4-phenylenediamine |
Turbine oils, transformer oils, hydraulic fluids, waxes, and greases |
| AO-24 |
N,N'-di-2-butyl-1,4-phenylenediamine |
Low-temperature oils |
| AO-29 |
2,6-di-tert-butyl-4-methylphenol |
Turbine oils, transformer oils, hydraulic fluids, waxes, greases, and gasolines |
| AO-30 |
2,4-dimethyl-6-tert-butylphenol |
Jet fuels and gasolines, including aviation gasolines |
| AO-31 |
2,4-dimethyl-6-tert-butylphenol |
Jet fuels and gasolines, including aviation gasolines |
| AO-32 |
2,4-dimethyl-6-tert-butylphenol and 2,6-di-tert-butyl-4-methylphenol |
Jet fuels and gasolines, including aviation gasolines |
| AO-37 |
2,6-di-tert-butylphenol |
Jet fuels and gasolines, widely approved for aviation fuels |
Oxidative challenge in biology

A
paradox in
metabolism is that, while the vast majority of complex
life on Earth requires
oxygen for its existence, oxygen is a highly reactive molecule that damages living organisms by producing
reactive oxygen species.
[82] Consequently, organisms contain a complex network of antioxidant
metabolites and
enzymes that work together to prevent oxidative damage to cellular components such as
DNA,
proteins and
lipids.
[1][83] In general, antioxidant systems either prevent these reactive species from being formed, or remove them before they can damage vital components of the cell.
[1][82] However, reactive oxygen species also have useful cellular functions, such as
redox signaling. Thus, the function of antioxidant systems is not to remove oxidants entirely, but instead to keep them at an optimum level.
[84]
The reactive oxygen species produced in cells include
hydrogen peroxide (H
2O
2),
hypochlorous acid (HClO), and
free radicals such as the
hydroxyl radical (·OH) and the
superoxide anion (O
2−).
[85] The hydroxyl radical is particularly unstable and will react rapidly and non-specifically with most biological molecules. This species is produced from hydrogen peroxide in
metal-catalyzed redox reactions such as the
Fenton reaction.
[86] These oxidants can damage cells by starting chemical chain reactions such as lipid peroxidation, or by oxidizing DNA or proteins.
[1] Damage to DNA can cause
mutations and possibly cancer, if not reversed by
DNA repair mechanisms,
[87][88] while damage to
proteins causes enzyme inhibition,
denaturation and
protein degradation.
[89]
The use of oxygen as part of the process for generating metabolic energy produces reactive oxygen species.
[90] In this process, the superoxide anion is produced as a
by-product of several steps in the
electron transport chain.
[91] Particularly important is the reduction of
coenzyme Q in
complex III, since a highly reactive free radical is formed as an intermediate (Q
·−). This unstable intermediate can lead to electron "leakage", when electrons jump directly to oxygen and form the superoxide anion, instead of moving through the normal series of well-controlled reactions of the electron transport chain.
[92] Peroxide is also produced from the oxidation of reduced
flavoproteins, such as
complex I.
[93] However, although these enzymes can produce oxidants, the relative importance of the electron transfer chain to other processes that generate peroxide is unclear.
[94][95] In plants,
algae, and
cyanobacteria, reactive oxygen species are also produced during
photosynthesis,
[96] particularly under conditions of high
light intensity.
[97] This effect is partly offset by the involvement of
carotenoids in
photoinhibition, and in algae and cyanobacteria, by large amount of
iodide and
selenium,
[98] which involves these antioxidants reacting with over-reduced forms of the
photosynthetic reaction centres to prevent the production of reactive oxygen species.
[99][100]
Metabolites
Antioxidants are classified into two broad divisions, depending on whether they are soluble in water (
hydrophilic) or in lipids (
lipophilic). In general, water-soluble antioxidants react with oxidants in the cell
cytosol and the
blood plasma, while lipid-soluble antioxidants protect
cell membranes from lipid peroxidation.
[1] These compounds may be synthesized in the body or obtained from the diet.
[83] The different antioxidants are present at a wide range of concentrations in
body fluids and tissues, with some such as glutathione or
ubiquinone mostly present within cells, while others such as
uric acid are more evenly distributed (see table below). Some antioxidants are only found in a few organisms and these compounds can be important in
pathogens and can be
virulence factors.
[101]
The relative importance and interactions between these different antioxidants is a very complex question, with the various metabolites and enzyme systems having
synergistic and interdependent effects on one another.
[102][103] The action of one antioxidant may therefore depend on the proper function of other members of the antioxidant system.
[83] The amount of protection provided by any one antioxidant will also depend on its concentration, its reactivity towards the particular reactive oxygen species being considered, and the status of the antioxidants with which it interacts.
[83]
Some compounds contribute to antioxidant defense by
chelating transition metals and preventing them from catalyzing the production of free radicals in the cell. Particularly important is the ability to sequester iron, which is the function of
iron-binding proteins such as
transferrin and
ferritin.
[95] Selenium and
zinc are commonly referred to as
antioxidant nutrients, but these
chemical elements have no antioxidant action themselves and are instead required for the activity of some antioxidant enzymes, as is discussed below.
Uric acid
Uric acid is by far the highest concentration antioxidant in human blood. Uric acid (UA) is an antioxidant oxypurine produced from
xanthine by the enzyme
xanthine oxidase, and is an intermediate product of
purine metabolism.
[116] In almost all land animals,
urate oxidase further catalyzes the oxidation of uric acid to
allantoin,
[117] but in humans and most higher primates, the urate oxidase gene is nonfunctional, so that UA is not further broken down.
[117][118] The evolutionary reasons for this loss of urate conversion to allantoin remain the topic of active speculation.
[119][120] The antioxidant effects of uric acid have led researchers to suggest this mutation was beneficial to early primates and humans.
[120][121] Studies of high altitude acclimatization support the hypothesis that urate acts as an antioxidant by mitigating the oxidative stress caused by high-altitude hypoxia.
[122] In animal studies that investigate diseases facilitated by oxidative stress, introduction of UA both prevents the disease or reduces it, leading researchers to propose this is due to UA's antioxidant properties.
[123] Studies of UA's antioxidant mechanism support this proposal.
[124]
With respect to multiple sclerosis, Gwen Scott explains the significance of uric acid as an antioxidant by proposing that "Serum UA levels are inversely associated with the incidence of
MS in humans because MS patients have low serum UA levels and individuals with hyperuricemia (gout) rarely develop the disease. Moreover, the administration of UA is therapeutic in experimental allergic encephalomyelitis (EAE), an animal model of MS."
[123][125][126] In sum, while the mechanism of UA as an antioxidant is well-supported, the claim that its levels affect MS risk is still controversial,
[127][128] and requires more research.
Likewise, UA has the highest concentration of any blood antioxidant
[110] and provides over half of the total antioxidant capacity of human serum.
[129] Uric acid's antioxidant activities are also complex, given that it does not react with some oxidants, such as
superoxide, but does act against
peroxynitrite,
[130] peroxides, and
hypochlorous acid.
[116] Concerns over elevated UA's contribution to
gout must be considered as one of many risk factors.
[131] By itself, UA-related risk of gout at high levels (415–530 μmol/L) is only 0.5% per year with an increase to 4.5% per year at UA
supersaturation levels (535+ μmol/L).
[132] Many of these aforementioned studies determined UA's antioxidant actions within normal physiological levels,
[122][130] and some found antioxidant activity at levels as high as 285 μmol/L.
[133]
Vitamin C
Ascorbic acid or "
vitamin C" is a
monosaccharide oxidation-reduction (
redox)
catalyst found in both animals and plants. As one of the enzymes needed to make ascorbic acid has been lost by
mutation during
primate evolution, humans must obtain it from the diet; it is therefore a vitamin.
[134] Most other animals are able to produce this compound in their bodies and do not require it in their diets.
[135] Ascorbic acid is required for the conversion of the
procollagen to
collagen by oxidizing
proline residues to
hydroxyproline. In other cells, it is maintained in its
reduced form by reaction with glutathione, which can be catalysed by
protein disulfide isomerase and
glutaredoxins.
[136][137] Ascorbic acid is a redox catalyst which can reduce, and thereby neutralize, reactive oxygen species such as hydrogen peroxide.
[138] In addition to its direct antioxidant effects, ascorbic acid is also a
substrate for the redox enzyme
ascorbate peroxidase, a function that is particularly important in stress resistance in plants.
[139] Ascorbic acid is present at high levels in all parts of plants and can reach concentrations of 20
millimolar in
chloroplasts.
[140]
Glutathione
Glutathione is a
cysteine-containing
peptide found in most forms of aerobic life.
[141] It is not required in the diet and is instead synthesized in cells from its constituent
amino acids.
[142] Glutathione has antioxidant properties since the
thiol group in its
cysteine moiety is a reducing agent and can be reversibly oxidized and reduced. In cells, glutathione is maintained in the reduced form by the enzyme
glutathione reductase and in turn reduces other metabolites and enzyme systems, such as ascorbate in the
glutathione-ascorbate cycle,
glutathione peroxidases and
glutaredoxins, as well as reacting directly with oxidants.
[136] Due to its high concentration and its central role in maintaining the cell's redox state, glutathione is one of the most important cellular antioxidants.
[141] In some organisms glutathione is replaced by other thiols, such as by
mycothiol in the
Actinomycetes,
bacillithiol in some
Gram-positive bacteria,
[143][144] or by
trypanothione in the
Kinetoplastids.
[145][146]
Melatonin
Melatonin is a powerful antioxidant.
[147] Melatonin easily crosses cell membranes and the
blood–brain barrier.
[148] Unlike other antioxidants, melatonin does not undergo
redox cycling, which is the ability of a molecule to undergo repeated
reduction and
oxidation. Redox cycling may allow other antioxidants (such as vitamin C) to act as
pro-oxidants and promote free radical formation. Melatonin, once oxidized, cannot be reduced to its former state because it forms several stable end-products upon reacting with free radicals. Therefore, it has been referred to as a terminal (or suicidal) antioxidant.
[149]
Vitamin E
Vitamin E is the collective name for a set of eight related
tocopherols and
tocotrienols, which are
fat-soluble vitamins with antioxidant properties.
[150][151] Of these, α-tocopherol has been most studied as it has the highest
bioavailability, with the body preferentially absorbing and metabolising this form.
[152]
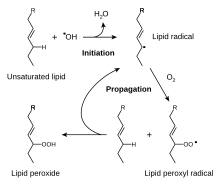
It has been claimed that the α-tocopherol form is the most important lipid-soluble antioxidant, and that it protects membranes from oxidation by reacting with lipid radicals produced in the lipid peroxidation chain reaction.
[150][153] This removes the free radical intermediates and prevents the propagation reaction from continuing. This reaction produces oxidised α-tocopheroxyl radicals that can be recycled back to the active reduced form through reduction by other antioxidants, such as ascorbate, retinol or ubiquinol.
[154] This is in line with findings showing that α-tocopherol, but not water-soluble antioxidants, efficiently protects glutathione peroxidase 4 (
GPX4)-deficient cells from cell death.
[155] GPx4 is the only known enzyme that efficiently reduces lipid-hydroperoxides within biological membranes.
However, the roles and importance of the various forms of vitamin E are presently unclear,
[156][157] and it has even been suggested that the most important function of α-tocopherol is as a
signaling molecule, with this molecule having no significant role in antioxidant metabolism.
[158][159] The functions of the other forms of vitamin E are even less well-understood, although γ-tocopherol is a
nucleophile that may react with
electrophilic mutagens,
[152] and tocotrienols may be important in protecting
neurons from damage.
[160]
Pro-oxidant activities
Antioxidants that are reducing agents can also act as pro-oxidants. For example, vitamin C has antioxidant activity when it reduces oxidizing substances such as hydrogen peroxide,
[161] however, it will also reduce metal ions that generate free radicals through the
Fenton reaction.
[86][162]
- 2 Fe3+ + Ascorbate → 2 Fe2+ + Dehydroascorbate
- 2 Fe2+ + 2 H2O2 → 2 Fe3+ + 2 OH· + 2 OH−
The relative importance of the antioxidant and pro-oxidant activities of antioxidants is an area of current research, but vitamin C, which exerts its effects as a vitamin by oxidizing polypeptides, appears to have a mostly antioxidant action in the human body.
[162][163] However, less data is available for other dietary antioxidants, such as vitamin E,
[164] or the
polyphenols.
[165][166] Likewise, the pathogenesis of diseases involving hyperuricemia likely involve uric acid's direct and indirect pro-oxidant properties.
That is, paradoxically, agents which are normally considered antioxidants can act as conditional
pro-oxidants and actually increase oxidative stress. Besides ascorbate, medically important conditional pro-oxidants include uric acid and sulfhydryl amino acids such as
homocysteine. Typically, this involves some transition-series metal such as copper or iron as catalyst. The potential role of the pro-oxidant role of uric acid in (e.g.) atherosclerosis and ischemic stroke is considered above. Another example is the postulated role of
homocysteine in atherosclerosis.
Negative health effects
Some antioxidant supplements may promote disease and increase mortality in humans under certain conditions.
[61][166] Hypothetically, free radicals induce an endogenous response that protects against exogenous radicals (and possibly other toxic compounds).
[167] Free radicals may increase life span.
[166] This increase may be prevented by antioxidants, providing direct evidence that toxic radicals may
mitohormetically exert life extending and health promoting effects.
[61][166]
Enzyme systems

Enzymatic pathway for detoxification of reactive oxygen species.
As with the chemical antioxidants, cells are protected against oxidative stress by an interacting network of antioxidant enzymes.
[1][82] Here, the superoxide released by processes such as
oxidative phosphorylation is first converted to hydrogen peroxide and then further reduced to give water. This detoxification pathway is the result of multiple enzymes, with superoxide dismutases catalysing the first step and then catalases and various peroxidases removing hydrogen peroxide. As with antioxidant metabolites, the contributions of these enzymes to antioxidant defenses can be hard to separate from one another, but the generation of
transgenic mice lacking just one antioxidant enzyme can be informative.
[168]
Superoxide dismutase, catalase and peroxiredoxins
Superoxide dismutases (SODs) are a class of closely related enzymes that catalyze the breakdown of the superoxide anion into oxygen and hydrogen peroxide.
[169][170] SOD enzymes are present in almost all aerobic cells and in extracellular fluids.
[171] Superoxide dismutase enzymes contain metal ion cofactors that, depending on the isozyme, can be copper, zinc,
manganese or iron. In humans, the copper/zinc SOD is present in the
cytosol, while manganese SOD is present in the
mitochondrion.
[170] There also exists a third form of SOD in
extracellular fluids, which contains copper and zinc in its active sites.
[172] The mitochondrial isozyme seems to be the most biologically important of these three, since mice lacking this enzyme die soon after birth.
[173] In contrast, the mice lacking copper/zinc SOD (Sod1) are viable but have numerous pathologies and a reduced lifespan (see article on
superoxide), while mice without the extracellular SOD have minimal defects (sensitive to
hyperoxia).
[168][174] In plants, SOD isozymes are present in the cytosol and mitochondria, with an iron SOD found in
chloroplasts that is absent from
vertebrates and
yeast.
[175]
Catalases are enzymes that catalyse the conversion of hydrogen peroxide to water and oxygen, using either an iron or manganese cofactor.
[176][177] This protein is localized to
peroxisomes in most
eukaryotic cells.
[178] Catalase is an unusual enzyme since, although hydrogen peroxide is its only substrate, it follows a
ping-pong mechanism. Here, its cofactor is oxidised by one molecule of hydrogen peroxide and then regenerated by transferring the bound oxygen to a second molecule of substrate.
[179] Despite its apparent importance in hydrogen peroxide removal, humans with genetic deficiency of catalase — "
acatalasemia" — or mice
genetically engineered to lack catalase completely, suffer few ill effects.
[180][181]
Peroxiredoxins are peroxidases that catalyze the reduction of hydrogen peroxide,
organic hydroperoxides, as well as
peroxynitrite.
[183] They are divided into three classes: typical 2-cysteine peroxiredoxins; atypical 2-cysteine peroxiredoxins; and 1-cysteine peroxiredoxins.
[184] These enzymes share the same basic catalytic mechanism, in which a redox-active cysteine (the peroxidatic cysteine) in the
active site is oxidized to a
sulfenic acid by the peroxide substrate.
[185] Over-oxidation of this cysteine residue in peroxiredoxins inactivates these enzymes, but this can be reversed by the action of
sulfiredoxin.
[186] Peroxiredoxins seem to be important in antioxidant metabolism, as mice lacking peroxiredoxin 1 or 2 have shortened lifespan and suffer from
hemolytic anaemia, while plants use peroxiredoxins to remove hydrogen peroxide generated in chloroplasts.
[187][188][189]
Thioredoxin and glutathione systems
The
thioredoxin system contains the 12-k
Da protein thioredoxin and its companion
thioredoxin reductase.
[190] Proteins related to thioredoxin are present in all sequenced organisms. Plants, such as
Arabidopsis thaliana, have a particularly great diversity of isoforms.
[191] The active site of thioredoxin consists of two
neighboring cysteines, as part of a highly conserved CXXC
motif, that can cycle between an active dithiol form (reduced) and an oxidized
disulfide form. In its active state, thioredoxin acts as an efficient reducing agent, scavenging reactive oxygen species and maintaining other proteins in their reduced state.
[192] After being oxidized, the active thioredoxin is regenerated by the action of thioredoxin reductase, using
NADPH as an
electron donor.
[193]
The
glutathione system includes glutathione,
glutathione reductase,
glutathione peroxidases and
glutathione S-transferases.
[141] This system is found in animals, plants and microorganisms.
[141][194] Glutathione peroxidase is an enzyme containing four
selenium-
cofactors that catalyzes the breakdown of hydrogen peroxide and organic hydroperoxides. There are at least four different glutathione peroxidase
isozymes in animals.
[195] Glutathione peroxidase 1 is the most abundant and is a very efficient scavenger of hydrogen peroxide, while glutathione peroxidase 4 is most active with lipid hydroperoxides. Surprisingly, glutathione peroxidase 1 is dispensable, as mice lacking this enzyme have normal lifespans,
[196] but they are hypersensitive to induced oxidative stress.
[197] In addition, the glutathione
S-transferases show high activity with lipid peroxides.
[198] These enzymes are at particularly high levels in the liver and also serve in
detoxification metabolism.
[199]
Oxidative stress in disease
Oxidative stress is thought to contribute to the development of a wide range of diseases including
Alzheimer's disease,
[200][201] Parkinson's disease,
[202] the pathologies caused by
diabetes,
[203][204] rheumatoid arthritis,
[205] and
neurodegeneration in
motor neuron diseases.
[206] In many of these cases, it is unclear if oxidants trigger the disease, or if they are produced as a secondary consequence of the disease and from general tissue damage;
[85] One case in which this link is particularly well-understood is the role of oxidative stress in
cardiovascular disease. Here,
low density lipoprotein (LDL) oxidation appears to trigger the process of
atherogenesis, which results in
atherosclerosis, and finally cardiovascular disease.
[207][208]
Oxidative damage in DNA can cause cancer. Several antioxidant enzymes such as superoxide dismutase, catalase, glutathione peroxidase, glutathione reductase, glutathione S-transferase etc. protect DNA from oxidative stress. It has been proposed that polymorphisms in these enzymes are associated with DNA damage and subsequently the individual's risk of cancer susceptibility.
[209]
A
low calorie diet extends median and
maximum lifespan in many animals. This effect may involve a reduction in oxidative stress.
[210] While there is some evidence to support the role of oxidative stress in aging in model organisms such as
Drosophila melanogaster and
Caenorhabditis elegans,
[211][212] the evidence in mammals is less clear.
[32][33][34] Indeed, a 2009 review of experiments in mice concluded that almost all manipulations of antioxidant systems had no effect on aging.
[213] Diets high in fruit and vegetables, which are high in antioxidants, promote health and reduce the effects of aging; antioxidant vitamin supplementation has no detectable effect on the aging process, so the effects of fruit and vegetables may be unrelated to their antioxidant contents.
[214][215] One reason for this might be the fact that consuming antioxidant molecules such as polyphenols and vitamin E will produce changes in other parts of metabolism, and it may be these other effects that are the real reason these compounds are important in human nutrition.
[158][216]
Measurement and levels in food

Fruits and vegetables are good sources of antioxidant vitamins.
Antioxidant vitamins are found in vegetables, fruits, eggs, legumes and nuts. Vitamins A, C or E can be destroyed by long-term storage or prolonged cooking.
[217] The effects of cooking and food processing are complex, as these processes can also increase the
bioavailability of antioxidants, such as some carotenoids in vegetables.
[218] Processed food contains fewer antioxidant vitamins than fresh and uncooked foods, as preparation exposes food to heat and oxygen.
[219]
| Antioxidant vitamins |
Foods containing high levels of antioxidant vitamins[56][220][221] |
| Vitamin C (ascorbic acid) |
Fresh or frozen fruits and vegetables |
| Vitamin E (tocopherols, tocotrienols) |
Vegetable oils, nuts and seeds |
| Carotenoids (carotenes as provitamin A) |
Fruit, vegetables and eggs |
Other antioxidants are not vitamins and are instead made in the body. For example,
ubiquinol (coenzyme Q) is poorly absorbed from the gut and is made in humans through the
mevalonate pathway.
[115] Another example is
glutathione, which is made from amino acids. As any glutathione in the gut is broken down to free cysteine,
glycine and
glutamic acid before being absorbed, even large oral doses have little effect on the concentration of glutathione in the body.
[222][223] Although large amounts of sulfur-containing amino acids such as
acetylcysteine can increase glutathione,
[224] no evidence exists that eating high levels of these glutathione precursors is beneficial for healthy adults.
[225] Supplying more of these precursors may be useful as part of the treatment of some diseases, such as
acute respiratory distress syndrome,
protein-energy malnutrition, or preventing the liver damage produced by
paracetamol overdose.
[224][226]
Other compounds in the diet can alter the levels of antioxidants by acting as
pro-oxidants whereby consuming the compound may cause
oxidative stress, possibly resulting in higher levels of antioxidant enzymes.
[27]
Invalidation of ORAC
Measurement of antioxidant content in food is not a straightforward process, as this is a diverse group of compounds with different reactivities to various reactive oxygen species. In
food science, the
oxygen radical absorbance capacity (ORAC) used to be the industry standard for antioxidant strength of whole foods, juices and food additives.
[227][228] However, the
United States Department of Agriculture (USDA) withdrew these ratings in 2012 as biologically invalid, stating that no physiological proof
in vivo existed to support the
free-radical theory, especially for
polyphenols.
[229] Consequently, the ORAC method, derived only from
in vitro experiments, is no longer considered relevant to human diets or
biology.
Alternative
in vitro measurements include the
Folin-Ciocalteu reagent, and the
Trolox equivalent antioxidant capacity assay.
[230]
History
As part of their adaptation from marine life, terrestrial plants began producing non-marine antioxidants such as
ascorbic acid (
vitamin C),
polyphenols and
tocopherols. The evolution of
angiosperm plants between 50 and 200 million years ago resulted in the development of many antioxidant pigments – particularly during the
Jurassic period – as chemical defences against
reactive oxygen species that are byproducts of
photosynthesis.
[231] Originally, the term antioxidant specifically referred to a chemical that prevented the consumption of oxygen. In the late 19th and early 20th centuries, extensive study concentrated on the use of antioxidants in important industrial processes, such as the prevention of metal
corrosion, the
vulcanization of rubber, and the
polymerization of fuels in the
fouling of
internal combustion engines.
[232]
Early research on the role of antioxidants in biology focused on their use in preventing the oxidation of
unsaturated fats, which is the cause of
rancidity.
[233] Antioxidant activity could be measured simply by placing the fat in a closed container with oxygen and measuring the rate of oxygen consumption. However, it was the identification of
vitamins A,
C, and
E as antioxidants that revolutionized the field and led to the realization of the importance of antioxidants in the biochemistry of
living organisms.
[234][235] The possible
mechanisms of action of antioxidants were first explored when it was recognized that a substance with anti-oxidative activity is likely to be one that is itself readily oxidized.
[236] Research into how
vitamin E prevents the process of
lipid peroxidation led to the identification of antioxidants as reducing agents that prevent oxidative reactions, often by
scavenging reactive oxygen species before they can damage cells.
[237]