From Wikipedia, the free encyclopedia
The
electron is a
subatomic particle, symbol
e− or
β−, whose
electric charge is negative one
elementary charge. Electrons belong to the first
generation of the
lepton particle family, and are generally thought to be
elementary particles because they have no known components or substructure. The electron has a
mass that is approximately
1/1836 that of the
proton.
Quantum mechanical properties of the electron include an intrinsic
angular momentum (
spin) of a half-integer value, expressed in units of the
reduced Planck constant,
ħ. As it is a
fermion, no two electrons can occupy the same
quantum state, in accordance with the
Pauli exclusion principle. Like all elementary particles, electrons exhibit properties of
both particles and waves: they can collide with other particles and can be
diffracted like light. The
wave properties of electrons are easier to observe with experiments than those of other particles like
neutrons and protons because electrons have a lower mass and hence a longer
de Broglie wavelength for a given energy.
Electrons play an essential role in numerous
physical phenomena, such as
electricity,
magnetism,
chemistry and
thermal conductivity, and they also participate in
gravitational,
electromagnetic and
weak interactions.
[11] Since an electron has charge, it has a surrounding
electric field, and if that electron is moving relative to an observer, it will generate a
magnetic field. Electromagnetic fields produced from other sources will affect the motion of an electron according to the
Lorentz force law.
Electrons radiate or absorb energy in the form of photons when they are
accelerated. Laboratory instruments are capable of trapping individual
electrons as well as
electron plasma by the use of electromagnetic fields. Special
telescopes can detect electron plasma in outer space. Electrons are involved in many applications such as
electronics,
welding,
cathode ray tubes,
electron microscopes,
radiation therapy,
lasers,
gaseous ionization detectors and
particle accelerators.
Interactions involving electrons with other subatomic particles are of interest in fields such as
chemistry and
nuclear physics. The
Coulomb force interaction between the positive
protons within
atomic nuclei and the negative electrons without, allows the composition of the two known as
atoms. Ionization or differences in the proportions of negative electrons versus positive nuclei changes the
binding energy of an atomic system. The exchange or sharing of the electrons between two or more atoms is the main cause of
chemical bonding.
[12] In 1838, British natural philosopher
Richard Laming first hypothesized the concept of an indivisible quantity of electric charge to explain the
chemical properties of atoms.
[3] Irish physicist
George Johnstone Stoney named this charge 'electron' in 1891, and
J. J. Thomson and his team of British physicists identified it as a particle in 1897. Electrons can also participate in
nuclear reactions, such as
nucleosynthesis in stars, where they are known as
beta particles. Electrons can be created through
beta decay of
radioactive isotopes and in high-energy collisions, for instance when
cosmic rays enter the atmosphere. The
antiparticle of the electron is called the
positron; it is identical to the electron except that it carries electrical and other
charges of the opposite sign. When an
electron collides with a positron, both particles can be
annihilated, producing
gamma ray photons.
History
Discovery of effect of electric force
The
ancient Greeks noticed that
amber attracted small objects when rubbed with fur. Along with
lightning, this phenomenon is one of humanity's earliest recorded experiences with
electricity.
[15] In his 1600 treatise
De Magnete, the English scientist
William Gilbert coined the
New Latin term
electricus, to refer to this property of attracting small objects after being rubbed.
[16] Both
electric and
electricity are derived from the Latin
ēlectrum (also the root of the
alloy of the same name), which came from the Greek word for amber,
ἤλεκτρον (
ēlektron).
Discovery of two kinds of charges
In the early 1700s,
Francis Hauksbee and French chemist
Charles François du Fay
independently discovered what they believed were two kinds of
frictional electricity—one generated from rubbing glass, the other from
rubbing resin. From this, du Fay theorized that electricity consists of
two
electrical fluids,
vitreous and
resinous, that are separated by friction, and that neutralize each other when combined.
[17] American scientist
Ebenezer Kinnersley later also independently reached the same conclusion.
[18]:118 A decade later
Benjamin Franklin
proposed that electricity was not from different types of electrical
fluid, but a single electrical fluid showing an excess (+) or deficit
(-). He gave them the modern
charge nomenclature of positive and negative respectively.
[19]
Franklin thought of the charge carrier as being positive, but he did
not correctly identify which situation was a surplus of the charge
carrier, and which situation was a deficit.
[20]
Between 1838 and 1851, British natural philosopher
Richard Laming developed the idea that an atom is composed of a core of matter surrounded by subatomic particles that had unit
electric charges.
[2] Beginning in 1846, German physicist
William Weber theorized that electricity was composed of positively and negatively charged fluids, and their interaction was governed by the
inverse square law. After studying the phenomenon of
electrolysis in 1874, Irish physicist
George Johnstone Stoney suggested that there existed a "single definite quantity of electricity", the charge of a
monovalent ion. He was able to estimate the value of this elementary charge
e by means of
Faraday's laws of electrolysis.
[21] However, Stoney believed these charges were permanently attached to atoms and could not be removed. In 1881, German physicist
Hermann von Helmholtz
argued that both positive and negative charges were divided into
elementary parts, each of which "behaves like atoms of electricity".
[3]
Stoney initially coined the term
electrolion in 1881. Ten years later, he switched to
electron
to describe these elementary charges, writing in 1894: "... an estimate
was made of the actual amount of this most remarkable fundamental unit
of electricity, for which I have since ventured to suggest the name
electron". A 1906 proposal to change to
electrion failed because
Hendrik Lorentz preferred to keep
electron.
[22][23] The word
electron is a combination of the words
electric and
ion.
[24] The suffix
-on which is now used to designate other subatomic particles, such as a proton or neutron, is in turn derived from electron.
[25][26]
Discovery of free electrons outside matter
A beam of electrons deflected in a circle by a magnetic field
[27]
The German physicist
Johann Wilhelm Hittorf studied electrical conductivity in
rarefied gases: in 1869, he discovered a glow emitted from the
cathode that increased in size with decrease in gas pressure. In 1876, the German physicist
Eugen Goldstein showed that the rays from this glow cast a shadow, and he dubbed the rays
cathode rays.
[28] During the 1870s, the English chemist and physicist Sir
William Crookes developed the first cathode ray tube to have a
high vacuum inside.
[29] He then showed that the luminescence rays appearing within the tube carried energy and moved from the cathode to the
anode.
Furthermore, by applying a magnetic field, he was able to deflect the
rays, thereby demonstrating that the beam behaved as though it were
negatively charged.
[30][31]
In 1879, he proposed that these properties could be explained by what
he termed 'radiant matter'. He suggested that this was a fourth state of
matter, consisting of negatively charged
molecules that were being projected with high velocity from the cathode.
[32]
The German-born British physicist
Arthur Schuster expanded upon Crookes' experiments by placing metal plates parallel to the cathode rays and applying an
electric potential
between the plates. The field deflected the rays toward the positively
charged plate, providing further evidence that the rays carried negative
charge. By measuring the amount of deflection for a given level of
current, in 1890 Schuster was able to estimate the
charge-to-mass ratio
of the ray components. However, this produced a value that was more
than a thousand times greater than what was expected, so little credence
was given to his calculations at the time.
[30][33]
In 1892
Hendrik Lorentz suggested that the mass of these particles (electrons) could be a consequence of their electric charge.
[34]
In 1896, the British physicist
J. J. Thomson, with his colleagues
John S. Townsend and
H. A. Wilson,
[13]
performed experiments indicating that cathode rays really were unique
particles, rather than waves, atoms or molecules as was believed
earlier.
[5] Thomson made good estimates of both the charge
e and the mass
m,
finding that cathode ray particles, which he called "corpuscles," had
perhaps one thousandth of the mass of the least massive ion known:
hydrogen.
[5][14] He showed that their charge-to-mass ratio,
e/
m,
was independent of cathode material. He further showed that the
negatively charged particles produced by radioactive materials, by
heated materials and by illuminated materials were universal.
[5][35] The name electron was again proposed for these particles by the Irish physicist
George Johnstone Stoney, and the name has since gained universal acceptance.
While studying naturally
fluorescing minerals in 1896, the French physicist
Henri Becquerel discovered that they emitted radiation without any exposure to an external energy source. These
radioactive materials became the subject of much interest by scientists, including the New Zealand physicist
Ernest Rutherford who discovered they emitted particles. He designated these particles
alpha and
beta, on the basis of their ability to penetrate matter.
[36] In 1900, Becquerel showed that the beta rays emitted by
radium could be deflected by an electric field, and that their mass-to-charge ratio was the same as for cathode rays.
[37] This evidence strengthened the view that electrons existed as components of atoms.
[38][39]
The electron's charge was more carefully measured by the American physicists
Robert Millikan and
Harvey Fletcher in their
oil-drop experiment
of 1909, the results of which were published in 1911. This experiment
used an electric field to prevent a charged droplet of oil from falling
as a result of gravity. This device could measure the electric charge
from as few as 1–150 ions with an error margin of less than 0.3%.
Comparable experiments had been done earlier by Thomson's team,
[5] using clouds of charged water droplets generated by electrolysis,
[13] and in 1911 by
Abram Ioffe,
who independently obtained the same result as Millikan using charged
microparticles of metals, then published his results in 1913.
[40]
However, oil drops were more stable than water drops because of their
slower evaporation rate, and thus more suited to precise experimentation
over longer periods of time.
[41]
Around the beginning of the twentieth century, it was found that
under certain conditions a fast-moving charged particle caused a
condensation of
supersaturated water vapor along its path. In 1911,
Charles Wilson used this principle to devise his
cloud chamber so he could photograph the tracks of charged particles, such as fast-moving electrons.
[42]
Atomic theory
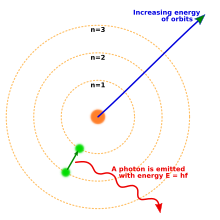
The
Bohr model of the atom, showing states of electron with energy
quantized by the number n. An electron dropping to a lower orbit emits a photon equal to the energy difference between the orbits.
By 1914, experiments by physicists
Ernest Rutherford,
Henry Moseley,
James Franck and
Gustav Hertz had largely established the structure of an atom as a dense
nucleus of positive charge surrounded by lower-mass electrons.
[43] In 1913, Danish physicist
Niels Bohr
postulated that electrons resided in quantized energy states, with
their energies determined by the angular momentum of the electron's
orbit about the nucleus. The electrons could move between those states,
or orbits, by the emission or absorption of photons of specific
frequencies. By means of these quantized orbits, he accurately explained
the
spectral lines of the hydrogen atom.
[44]
However, Bohr's model failed to account for the relative intensities of
the spectral lines and it was unsuccessful in explaining the spectra of
more complex atoms.
[43]
Chemical bonds between atoms were explained by
Gilbert Newton Lewis, who in 1916 proposed that a
covalent bond between two atoms is maintained by a pair of electrons shared between them.
[45] Later, in 1927,
Walter Heitler and
Fritz London gave the full explanation of the electron-pair formation and chemical bonding in terms of
quantum mechanics.
[46] In 1919, the American chemist
Irving Langmuir
elaborated on the Lewis' static model of the atom and suggested that
all electrons were distributed in successive "concentric (nearly)
spherical shells, all of equal thickness".
[47]
In turn, he divided the shells into a number of cells each of which
contained one pair of electrons. With this model Langmuir was able to
qualitatively explain the
chemical properties of all elements in the periodic table,
[46] which were known to largely repeat themselves according to the
periodic law.
[48]
In 1924, Austrian physicist
Wolfgang Pauli
observed that the shell-like structure of the atom could be explained
by a set of four parameters that defined every quantum energy state, as
long as each state was occupied by no more than a single electron. This
prohibition against more than one electron occupying the same quantum
energy state became known as the
Pauli exclusion principle.
[49]
The physical mechanism to explain the fourth parameter, which had two
distinct possible values, was provided by the Dutch physicists
Samuel Goudsmit and
George Uhlenbeck.
In 1925, they suggested that an electron, in addition to the angular
momentum of its orbit, possesses an intrinsic angular momentum and
magnetic dipole moment.
[43][50]
This is analogous to the rotation of the Earth on its axis as it orbits
the Sun. The intrinsic angular momentum became known as
spin, and explained the previously mysterious splitting of spectral lines observed with a high-resolution
spectrograph; this phenomenon is known as
fine structure splitting.
[51]
Quantum mechanics
In his 1924 dissertation
Recherches sur la théorie des quanta (Research on Quantum Theory), French physicist
Louis de Broglie hypothesized that all matter can be represented as a
de Broglie wave in the manner of
light.
[52] That is, under the appropriate conditions, electrons and other matter would show properties of either particles or waves. The
corpuscular properties of a particle are demonstrated when it is shown to have a localized position in space along its trajectory at any given moment.
[53] The wave-like nature of light is displayed, for example, when a beam of
light is passed through parallel slits thereby creating
interference patterns. In 1927
George Paget Thomson,
discovered the interference effect was produced when a beam of
electrons was passed through thin metal foils and by American physicists
Clinton Davisson and
Lester Germer by the reflection of electrons from a crystal of
nickel.
[54]
In quantum mechanics, the behavior of an electron in an atom is described by an
orbital,
which is a probability distribution rather than an orbit. In the
figure, the shading indicates the relative probability to "find" the
electron, having the energy corresponding to the given
quantum numbers, at that point.
De Broglie's prediction of a wave nature for electrons led
Erwin Schrödinger to postulate a wave equation for electrons moving under the influence of the nucleus in the atom. In 1926, this equation, the
Schrödinger equation, successfully described how electron waves propagated.
[55]
Rather than yielding a solution that determined the location of an
electron over time, this wave equation also could be used to predict the
probability of finding an electron near a position, especially a
position near where the electron was bound in space, for which the
electron wave equations did not change in time. This approach led to a
second formulation of
quantum mechanics
(the first by Heisenberg in 1925), and solutions of Schrödinger's
equation, like Heisenberg's, provided derivations of the energy states
of an electron in a hydrogen atom that were equivalent to those that had
been derived first by Bohr in 1913, and that were known to reproduce
the hydrogen spectrum.
[56]
Once spin and the interaction between multiple electrons were
describable, quantum mechanics made it possible to predict the
configuration of electrons in atoms with atomic numbers greater than
hydrogen.
[57]
In 1928, building on Wolfgang Pauli's work,
Paul Dirac produced a model of the electron – the
Dirac equation, consistent with
relativity theory, by applying relativistic and symmetry considerations to the
hamiltonian formulation of the quantum mechanics of the electro-magnetic field.
[58]
In order to resolve some problems within his relativistic equation,
Dirac developed in 1930 a model of the vacuum as an infinite sea of
particles with negative energy, later dubbed the
Dirac sea. This led him to predict the existence of a positron, the
antimatter counterpart of the electron.
[59] This particle was discovered in 1932 by
Carl Anderson, who proposed calling standard electrons
negatons and using
electron as a generic term to describe both the positively and negatively charged variants.
In 1947
Willis Lamb, working in collaboration with graduate student
Robert Retherford,
found that certain quantum states of the hydrogen atom, which should
have the same energy, were shifted in relation to each other, the
difference came to be called the
Lamb shift. About the same time,
Polykarp Kusch, working with
Henry M. Foley,
discovered the magnetic moment of the electron is slightly larger than
predicted by Dirac's theory. This small difference was later called
anomalous magnetic dipole moment of the electron. This difference was later explained by the theory of
quantum electrodynamics, developed by
Sin-Itiro Tomonaga,
Julian Schwinger and
Richard Feynman in the late 1940s.
[60]
Particle accelerators
With the development of the
particle accelerator during the first half of the twentieth century, physicists began to delve deeper into the properties of
subatomic particles.
[61] The first successful attempt to accelerate electrons using
electromagnetic induction was made in 1942 by
Donald Kerst. His initial
betatron reached energies of 2.3 MeV, while subsequent betatrons achieved 300 MeV. In 1947,
synchrotron radiation was discovered with a 70 MeV electron synchrotron at
General Electric. This radiation was caused by the acceleration of electrons through a magnetic field as they moved near the speed of light.
[62]
With a beam energy of 1.5 GeV, the first high-energy
particle
collider was
ADONE, which began operations in 1968.
[63]
This device accelerated electrons and positrons in opposite directions,
effectively doubling the energy of their collision when compared to
striking a static target with an electron.
[64] The
Large Electron–Positron Collider (LEP) at
CERN, which was operational from 1989 to 2000, achieved collision energies of 209 GeV and made important measurements for the
Standard Model of particle physics.
[65][66]
Confinement of individual electrons
Individual electrons can now be easily confined in ultra small (
L = 20 nm,
W = 20 nm) CMOS transistors operated at cryogenic temperature over a range of −269 °C (4
K) to about −258 °C (15
K).
[67]
The electron wavefunction spreads in a semiconductor lattice and
negligibly interacts with the valence band electrons, so it can be
treated in the single particle formalism, by replacing its mass with the
effective mass tensor.
Characteristics
Classification
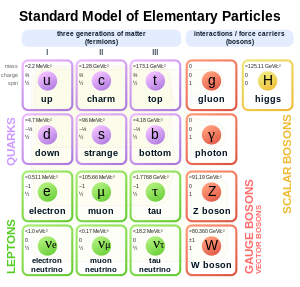
Standard Model of elementary particles. The electron (symbol e) is on the left.
In the
Standard Model of particle physics, electrons belong to the group of subatomic particles called
leptons, which are believed to be fundamental or
elementary particles. Electrons have the lowest mass of any charged lepton (or electrically charged particle of any type) and belong to the first-
generation of fundamental particles.
[68] The second and third generation contain charged leptons, the
muon and the
tau, which are identical to the electron in charge,
spin and
interactions, but are more massive. Leptons differ from the other basic constituent of matter, the
quarks, by their lack of
strong interaction. All members of the lepton group are fermions, because they all have half-odd integer spin; the electron has spin
1/2.
[69]
Fundamental properties
The
invariant mass of an electron is approximately
9.109×10−31 kilograms,
[70] or
5.489×10−4 atomic mass units. On the basis of
Einstein's principle of
mass–energy equivalence, this mass corresponds to a rest energy of
0.511 MeV. The ratio between the mass of a
proton and that of an electron is about 1836.
[10][71] Astronomical measurements show that the
proton-to-electron mass ratio has held the same value, as is predicted by the Standard Model, for at least half the
age of the universe.
[72]
Electrons have an
electric charge of
−1.602×10−19 coulombs,
[70] which is used as a standard unit of charge for subatomic particles, and is also called the
elementary charge. This elementary charge has a relative standard uncertainty of
2.2×10−8.
[70]
Within the limits of experimental accuracy, the electron charge is
identical to the charge of a proton, but with the opposite sign.
[73] As the symbol
e is used for the
elementary charge, the electron is commonly symbolized by
e−, where the minus sign indicates the negative charge. The positron is symbolized by
e+ because it has the same properties as the electron but with a positive rather than negative charge.
[69][70]
The electron has an intrinsic
angular momentum or spin of
1/2.
[70] This property is usually stated by referring to the electron as a
spin-1/2 particle.
[69] For such particles the spin magnitude is
√3/2 ħ while the result of the measurement of a
projection of the spin on any axis can only be ±
ħ/2. In addition to spin, the electron has an intrinsic
magnetic moment along its spin axis.
[70] It is approximately equal to one
Bohr magneton,
[74][note 4] which is a physical constant equal to
9.27400915(23)×10−24 joules per tesla.
[70] The orientation of the spin with respect to the momentum of the electron defines the property of elementary particles known as
helicity.
[75]
The electron has no known
substructure[1][76] and it is assumed to be a
point particle with a
point charge and no spatial extent.
[9] In
classical physics,
the angular momentum and magnetic moment of an object depend upon its
physical dimensions. Hence, the concept of a dimensionless electron
possessing these properties contrasts to experimental observations in
Penning traps which point to finite non-zero radius of the electron.
[citation needed] A possible explanation of this paradoxical situation is given below in the "
Virtual particles" subsection by taking into consideration the
Foldy-Wouthuysen transformation.
The issue of the radius of the electron is a challenging problem
of the modern theoretical physics. The admission of the hypothesis of a
finite radius of the electron is incompatible to the premises of the
theory of relativity. On the other hand, a point-like electron (zero
radius) generates serious mathematical difficulties due to the
self-energy of the electron tending to infinity.
[77]
Observation of a single electron in a
Penning trap suggests the upper limit of the particle's radius to be 10
−22 meters.
[78]
The upper bound of the electron radius of 10
−18 meters
[79] can be derived using the
uncertainty relation in energy.
There
is also a physical constant called the "
classical electron radius", with the much larger value of
2.8179×10−15 m,
greater than the radius of the proton. However, the terminology comes
from a simplistic calculation that ignores the effects of
quantum mechanics; in reality, the so-called classical electron radius has little to do with the true fundamental structure of the electron.
[80][note 5]
There are
elementary particles that spontaneously
decay into less massive particles. An example is the
muon, with a
mean lifetime of
2.2×10−6 seconds, which decays into an electron, a muon
neutrino and an electron
antineutrino.
The electron, on the other hand, is thought to be stable on theoretical
grounds: the electron is the least massive particle with non-zero
electric charge, so its decay would violate
charge conservation.
[81] The experimental lower bound for the electron's mean lifetime is
6.6×1028 years, at a 90%
confidence level.
[7][82][83]
Quantum properties
As with all particles, electrons can act as waves. This is called the
wave–particle duality and can be demonstrated using the
double-slit experiment.
The wave-like nature of the electron allows it to pass through
two parallel slits simultaneously, rather than just one slit as would be
the case for a classical particle. In quantum mechanics, the wave-like
property of one particle can be described mathematically as a
complex-valued function, the
wave function, commonly denoted by the Greek letter psi (
ψ). When the
absolute value of this function is
squared, it gives the probability that a particle will be observed near a location—a
probability density.
[84]:162–218
Electrons are
identical particles
because they cannot be distinguished from each other by their intrinsic
physical properties. In quantum mechanics, this means that a pair of
interacting electrons must be able to swap positions without an
observable change to the state of the system. The wave function of
fermions, including electrons, is antisymmetric, meaning that it changes
sign when two electrons are swapped; that is,
ψ(r1, r2) = −ψ(r2, r1), where the variables
r1 and
r2
correspond to the first and second electrons, respectively. Since the
absolute value is not changed by a sign swap, this corresponds to equal
probabilities.
Bosons, such as the photon, have symmetric wave functions instead.
[84]:162–218
In the case of antisymmetry, solutions of the wave equation for interacting electrons result in a
zero probability that each pair will occupy the same location or state. This is responsible for the
Pauli exclusion principle,
which precludes any two electrons from occupying the same quantum
state. This principle explains many of the properties of electrons. For
example, it causes groups of bound electrons to occupy different
orbitals in an atom, rather than all overlapping each other in the same orbit.
[84]:162–218
Virtual particles
In a simplified picture, every photon spends some time as a
combination of a virtual electron plus its antiparticle, the virtual
positron, which rapidly
annihilate each other shortly thereafter.
[85]
The combination of the energy variation needed to create these
particles, and the time during which they exist, fall under the
threshold of detectability expressed by the
Heisenberg uncertainty relation, Δ
E · Δ
t ≥
ħ. In effect, the energy needed to create these virtual particles, Δ
E, can be "borrowed" from the
vacuum for a period of time, Δ
t, so that their product is no more than the
reduced Planck constant,
ħ ≈ 6.6×10−16 eV·s. Thus, for a virtual electron, Δ
t is at most
1.3×10−21 s.
[86]
A schematic depiction of virtual electron–positron pairs appearing at random near an electron (at lower left)
While an electron–positron virtual pair is in existence, the
coulomb force from the ambient
electric field
surrounding an electron causes a created positron to be attracted to
the original electron, while a created electron experiences a repulsion.
This causes what is called
vacuum polarization. In effect, the vacuum behaves like a medium having a
dielectric permittivity more than
unity.
Thus the effective charge of an electron is actually smaller than its
true value, and the charge decreases with increasing distance from the
electron.
[87][88] This polarization was confirmed experimentally in 1997 using the Japanese
TRISTAN particle accelerator.
[89] Virtual particles cause a comparable
shielding effect for the mass of the electron.
[90]
The interaction with virtual particles also explains the small
(about 0.1%) deviation of the intrinsic magnetic moment of the electron
from the Bohr magneton (the
anomalous magnetic moment).
[74][91] The extraordinarily precise agreement of this predicted difference with
the experimentally determined value is viewed as one of the great
achievements of
quantum electrodynamics.
[92]
The apparent paradox (mentioned above in the properties
subsection) of a point particle electron having intrinsic angular
momentum and magnetic moment can be explained by the formation of
virtual photons
in the electric field generated by the electron. These photons cause
the electron to shift about in a jittery fashion (known as
zitterbewegung),
[93] which results in a net circular motion with
precession. This motion produces both the spin and the magnetic moment of the electron.
[9][94] In atoms, this creation of virtual photons explains the
Lamb shift observed in
spectral lines.
[87]
Interaction
An
electron generates an electric field that exerts an attractive force on
a particle with a positive charge, such as the proton, and a repulsive
force on a particle with a negative charge. The strength of this force
in nonrelativistic approximation is determined by
Coulomb's inverse square law.
[95]:58–61 When an electron is in motion, it generates a
magnetic field.
[84]:140 The
Ampère-Maxwell law relates the magnetic field to the mass motion of electrons (the
current) with respect to an observer. This property of induction supplies the magnetic field that drives an
electric motor.
[96] The electromagnetic field of an arbitrary moving charged particle is expressed by the
Liénard–Wiechert potentials, which are valid even when the particle's speed is close to that of light (
relativistic).
[95]:429–434
A particle with charge q (at left) is moving with velocity v through a magnetic field B that is oriented toward the viewer. For an electron, q is negative so it follows a curved trajectory toward the top.
When an electron is moving through a magnetic field, it is subject to the
Lorentz force that acts perpendicularly to the plane defined by the magnetic field and the electron velocity. This
centripetal force causes the electron to follow a
helical trajectory through the field at a radius called the
gyroradius. The acceleration from this curving motion induces the electron to radiate energy in the form of synchrotron radiation.
[84]:160[97][note 6] The energy emission in turn causes a recoil of the electron, known as the
Abraham–Lorentz–Dirac Force, which creates a friction that slows the electron. This force is caused by a
back-reaction of the electron's own field upon itself.
[98]
Photons mediate electromagnetic interactions between particles in
quantum electrodynamics. An isolated electron at a constant velocity cannot emit or absorb a real photon; doing so would violate
conservation of energy and
momentum.
Instead, virtual photons can transfer momentum between two charged
particles. This exchange of virtual photons, for example, generates the
Coulomb force.
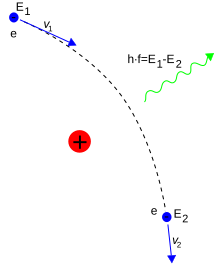
[99] Energy emission can occur when a moving electron is deflected by a
charged particle, such as a proton. The acceleration of the electron
results in the emission of
Bremsstrahlung radiation.
[100]
Here, Bremsstrahlung is produced by an electron e deflected by the electric field of an atomic nucleus. The energy change E2 − E1 determines the frequency f of the emitted photon.
An inelastic collision between a photon (light) and a solitary (free) electron is called
Compton scattering.
This collision results in a transfer of momentum and energy between the
particles, which modifies the wavelength of the photon by an amount
called the
Compton shift.
[note 7] The maximum magnitude of this wavelength shift is
h/
mec, which is known as the
Compton wavelength.
[101] For an electron, it has a value of
2.43×10−12 m.
[70] When the wavelength of the light is long (for instance, the wavelength of the
visible light is 0.4–0.7 μm) the wavelength shift becomes negligible. Such interaction between the light and free electrons is called
Thomson scattering or linear Thomson scattering.
[102]
The relative strength of the electromagnetic interaction between
two charged particles, such as an electron and a proton, is given by the
fine-structure constant.
This value is a dimensionless quantity formed by the ratio of two
energies: the electrostatic energy of attraction (or repulsion) at a
separation of one Compton wavelength, and the rest energy of the charge.
It is given by
α ≈
7.297353×10−3, which is approximately equal to
1/137.
[70]
When electrons and positrons collide, they
annihilate each other, giving rise to two or more gamma ray photons. If the electron and positron have negligible momentum, a
positronium atom can form before annihilation results in two or three gamma ray photons totalling 1.022 MeV.
[103][104] On the other hand, a high-energy photon can transform into an electron and a positron by a process called
pair production, but only in the presence of a nearby charged particle, such as a nucleus.
[105][106]
In the theory of
electroweak interaction, the
left-handed component of electron's wavefunction forms a
weak isospin doublet with the
electron neutrino. This means that during
weak interactions, electron neutrinos behave like electrons. Either member of this doublet can undergo a
charged current interaction by emitting or absorbing a
W
and be converted into the other member. Charge is conserved during this
reaction because the W boson also carries a charge, canceling out any
net change during the transmutation. Charged current interactions are
responsible for the phenomenon of
beta decay in a
radioactive atom. Both the electron and electron neutrino can undergo a
neutral current interaction via a
Z0 exchange, and this is responsible for neutrino-electron
elastic scattering.
[107]
Atoms and molecules
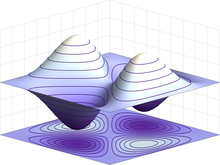
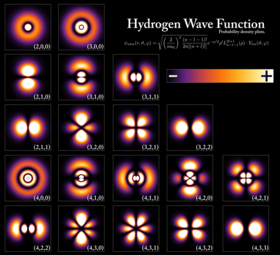
Probability
densities for the first few hydrogen atom orbitals, seen in
cross-section. The energy level of a bound electron determines the
orbital it occupies, and the color reflects the probability of finding
the electron at a given position.
An electron can be
bound to the nucleus of an atom by the
attractive Coulomb force. A system of one or more electrons bound to a
nucleus is called an atom. If the number of electrons is different from
the nucleus' electrical charge, such an atom is called an
ion. The wave-like behavior of a bound electron is described by a function called an
atomic orbital.
Each orbital has its own set of quantum numbers such as energy, angular
momentum and projection of angular momentum, and only a discrete set of
these orbitals exist around the nucleus. According to the Pauli
exclusion principle each orbital can be occupied by up to two electrons,
which must differ in their
spin quantum number.
Electrons can transfer between different orbitals by the emission
or absorption of photons with an energy that matches the difference in
potential.
[108] Other methods of orbital transfer include collisions with particles, such as electrons, and the
Auger effect.
[109] To escape the atom, the energy of the electron must be increased above its
binding energy to the atom. This occurs, for example, with the
photoelectric effect, where an incident photon exceeding the atom's
ionization energy is absorbed by the electron.
[110]
The orbital angular momentum of electrons is
quantized.
Because the electron is charged, it produces an orbital magnetic moment
that is proportional to the angular momentum. The net magnetic moment
of an atom is equal to the vector sum of orbital and spin magnetic
moments of all electrons and the nucleus. The magnetic moment of the
nucleus is negligible compared with that of the electrons. The magnetic
moments of the electrons that occupy the same orbital (so called, paired
electrons) cancel each other out.
[111]
The
chemical bond between atoms occurs as a result of electromagnetic interactions, as described by the laws of quantum mechanics.
[112] The strongest bonds are formed by the
sharing or
transfer of electrons between atoms, allowing the formation of
molecules.
[12] Within a molecule, electrons move under the influence of several nuclei, and occupy
molecular orbitals; much as they can occupy atomic orbitals in isolated atoms.
[113] A fundamental factor in these molecular structures is the existence of
electron pairs.
These are electrons with opposed spins, allowing them to occupy the
same molecular orbital without violating the Pauli exclusion principle
(much like in atoms). Different molecular orbitals have different
spatial distribution of the electron density. For instance, in bonded
pairs (i.e. in the pairs that actually bind atoms together) electrons
can be found with the maximal probability in a relatively small volume
between the nuclei. By contrast, in non-bonded pairs electrons are
distributed in a large volume around nuclei.
[114]
Conductivity
A
lightning discharge consists primarily of a flow of electrons.
[115] The electric potential needed for lightning can be generated by a triboelectric effect.
[116][117]
If a body has more or fewer electrons than are required to balance
the positive charge of the nuclei, then that object has a net electric
charge. When there is an excess of electrons, the object is said to be
negatively charged. When there are fewer electrons than the number of
protons in nuclei, the object is said to be positively charged. When the
number of electrons and the number of protons are equal, their charges
cancel each other and the object is said to be electrically neutral. A
macroscopic body can develop an electric charge through rubbing, by the
triboelectric effect.
[118]
Independent electrons moving in vacuum are termed
free
electrons. Electrons in metals also behave as if they were free. In
reality the particles that are commonly termed electrons in metals and
other solids are quasi-electrons—
quasiparticles, which have the same electrical charge, spin, and magnetic moment as real electrons but might have a different mass.
[119] When free electrons—both in vacuum and metals—move, they produce a
net flow of charge called an
electric current,
which generates a magnetic field. Likewise a current can be created by a
changing magnetic field. These interactions are described
mathematically by
Maxwell's equations.
[120]
At a given temperature, each material has an
electrical conductivity that determines the value of electric current when an
electric potential is applied. Examples of good conductors include metals such as copper and gold, whereas glass and
Teflon are poor conductors. In any
dielectric material, the electrons remain bound to their respective atoms and the material behaves as an
insulator. Most
semiconductors have a variable level of conductivity that lies between the extremes of conduction and insulation.
[121] On the other hand,
metals have an
electronic band structure
containing partially filled electronic bands. The presence of such
bands allows electrons in metals to behave as if they were free or
delocalized electrons.
These electrons are not associated with specific atoms, so when an
electric field is applied, they are free to move like a gas (called
Fermi gas)
[122] through the material much like free electrons.
Because of collisions between electrons and atoms, the
drift velocity
of electrons in a conductor is on the order of millimeters per second.
However, the speed at which a change of current at one point in the
material causes changes in currents in other parts of the material, the
velocity of propagation, is typically about 75% of light speed.
[123] This occurs because electrical signals propagate as a wave, with the velocity dependent on the
dielectric constant of the material.
[124]
Metals make relatively good conductors of heat, primarily because
the delocalized electrons are free to transport thermal energy between
atoms. However, unlike electrical conductivity, the thermal conductivity
of a metal is nearly independent of temperature. This is expressed
mathematically by the
Wiedemann–Franz law,
[122] which states that the ratio of
thermal conductivity
to the electrical conductivity is proportional to the temperature. The
thermal disorder in the metallic lattice increases the electrical
resistivity of the material, producing a temperature dependence for electric current.
[125]
When cooled below a point called the
critical temperature, materials can undergo a phase transition in which they lose all resistivity to electric current, in a process known as
superconductivity. In
BCS theory, this behavior is modeled by pairs of electrons entering a quantum state known as a
Bose–Einstein condensate. These
Cooper pairs have their motion coupled to nearby matter via lattice vibrations called
phonons, thereby avoiding the collisions with atoms that normally create electrical resistance.
[126] (Cooper pairs have a radius of roughly 100 nm, so they can overlap each other.)
[127] However, the mechanism by which
higher temperature superconductors operate remains uncertain.
Electrons inside conducting solids, which are quasi-particles themselves, when tightly confined at temperatures close to
absolute zero, behave as though they had split into three other
quasiparticles:
spinons,
orbitons and
holons.
[128][129] The former carries spin and magnetic moment, the next carries its orbital location while the latter electrical charge.
Motion and energy
According to
Einstein's theory of
special relativity, as an electron's speed approaches the
speed of light, from an observer's point of view its
relativistic mass
increases, thereby making it more and more difficult to accelerate it
from within the observer's frame of reference. The speed of an electron
can approach, but never reach, the speed of light in a vacuum,
c. However, when relativistic electrons—that is, electrons moving at a speed close to
c—are injected into a dielectric medium such as water, where the local speed of light is significantly less than
c,
the electrons temporarily travel faster than light in the medium. As
they interact with the medium, they generate a faint light called
Cherenkov radiation.
[130]
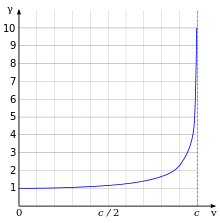
Lorentz factor as a function of velocity. It starts at value 1 and goes to infinity as v approaches c.
The effects of special relativity are based on a quantity known as the
Lorentz factor, defined as

where
v is the speed of the particle. The kinetic energy
Ke of an electron moving with velocity
v is:

where
me is the mass of electron. For example, the
Stanford linear accelerator can
accelerate an electron to roughly 51 GeV.
[131]
Since an electron behaves as a wave, at a given velocity it has a characteristic
de Broglie wavelength. This is given by
λe =
h/
p where
h is the
Planck constant and
p is the momentum.
[52] For the 51 GeV electron above, the wavelength is about
2.4×10−17 m, small enough to explore structures well below the size of an atomic nucleus.
[132]
Formation
Pair production
of an electron and positron, caused by the close approach of a photon
with an atomic nucleus. The lightning symbol represents an exchange of a
virtual photon, thus an electric force acts. The angle between the
particles is very small.
[133]
The
Big Bang theory is the most widely accepted scientific theory to explain the early stages in the evolution of the Universe.
[134] For the first millisecond of the Big Bang, the temperatures were over 10 billion
kelvins and photons had mean energies over a million
electronvolts.
These photons were sufficiently energetic that they could react with
each other to form pairs of electrons and positrons. Likewise,
positron-electron pairs annihilated each other and emitted energetic
photons:
γ
+
γ
↔
e+ +
e−
An equilibrium between electrons, positrons and photons was
maintained during this phase of the evolution of the Universe. After 15
seconds had passed, however, the temperature of the universe dropped
below the threshold where electron-positron formation could occur. Most
of the surviving electrons and positrons annihilated each other,
releasing gamma radiation that briefly reheated the universe.
[135]
For reasons that remain uncertain, during the annihilation
process there was an excess in the number of particles over
antiparticles. Hence, about one electron for every billion
electron-positron pairs survived. This excess matched the excess of
protons over antiprotons, in a condition known as
baryon asymmetry, resulting in a net charge of zero for the universe.
[136][137] The surviving protons and neutrons began to participate in reactions with each other—in the process known as
nucleosynthesis, forming isotopes of hydrogen and
helium, with trace amounts of
lithium. This process peaked after about five minutes.
[138] Any leftover neutrons underwent negative
beta decay with a half-life of about a thousand seconds, releasing a proton and electron in the process,
n
→
p
+
e− +
ν
e
For about the next
300000–
400000 years, the excess electrons remained too energetic to bind with
atomic nuclei.
[139] What followed is a period known as
recombination, when neutral atoms were formed and the expanding universe became transparent to radiation.
[140]
Roughly one million years after the big bang, the first generation of
stars began to form.
[140] Within a star,
stellar nucleosynthesis
results in the production of positrons from the fusion of atomic
nuclei. These antimatter particles immediately annihilate with
electrons, releasing gamma rays. The net result is a steady reduction in
the number of electrons, and a matching increase in the number of
neutrons. However, the process of
stellar evolution
can result in the synthesis of radioactive isotopes. Selected isotopes
can subsequently undergo negative beta decay, emitting an electron and
antineutrino from the nucleus.
[141] An example is the
cobalt-60 (
60Co) isotope, which decays to form
nickel-60 (
60Ni
).
[142]
An extended air shower generated by an energetic cosmic ray striking the Earth's atmosphere
At the end of its lifetime, a star with more than about 20
solar masses can undergo
gravitational collapse to form a
black hole.
[143] According to
classical physics, these massive stellar objects exert a
gravitational attraction that is strong enough to prevent anything, even
electromagnetic radiation, from escaping past the
Schwarzschild radius. However, quantum mechanical effects are believed to potentially allow the emission of
Hawking radiation at this distance. Electrons (and positrons) are thought to be created at the
event horizon of these
stellar remnants.
When a pair of virtual particles (such as an electron and
positron) is created in the vicinity of the event horizon, random
spatial positioning might result in one of them to appear on the
exterior; this process is called
quantum tunnelling. The
gravitational potential
of the black hole can then supply the energy that transforms this
virtual particle into a real particle, allowing it to radiate away into
space.
[144]
In exchange, the other member of the pair is given negative energy,
which results in a net loss of mass-energy by the black hole. The rate
of Hawking radiation increases with decreasing mass, eventually causing
the black hole to evaporate away until, finally, it explodes.
[145]
Cosmic rays are particles traveling through space with high energies. Energy events as high as
3.0×1020 eV have been recorded.
[146] When these particles collide with nucleons in the
Earth's atmosphere, a shower of particles is generated, including
pions.
[147] More than half of the cosmic radiation observed from the Earth's surface consists of
muons. The particle called a muon is a lepton produced in the upper atmosphere by the decay of a pion.
π− →
μ− +
ν
μ
A muon, in turn, can decay to form an electron or positron.
[148]
μ− →
e− +
ν
e +
ν
μ
Observation
Remote observation of electrons requires detection of their radiated
energy. For example, in high-energy environments such as the
corona of a star, free electrons form a
plasma that radiates energy due to
Bremsstrahlung radiation. Electron gas can undergo
plasma oscillation,
which is waves caused by synchronized variations in electron density,
and these produce energy emissions that can be detected by using
radio telescopes.
[150]
The
frequency of a
photon
is proportional to its energy. As a bound electron transitions between
different energy levels of an atom, it absorbs or emits photons at
characteristic frequencies. For instance, when atoms are irradiated by a
source with a broad spectrum, distinct
absorption lines
appear in the spectrum of transmitted radiation. Each element or
molecule displays a characteristic set of spectral lines, such as the
hydrogen spectral series.
Spectroscopic
measurements of the strength and width of these lines allow the
composition and physical properties of a substance to be determined.
[151][152]
In laboratory conditions, the interactions of individual electrons can be observed by means of
particle detectors, which allow measurement of specific properties such as energy, spin and charge.
[110] The development of the
Paul trap and
Penning trap
allows charged particles to be contained within a small region for long
durations. This enables precise measurements of the particle
properties. For example, in one instance a Penning trap was used to
contain a single electron for a period of 10 months.
[153]
The magnetic moment of the electron was measured to a precision of
eleven digits, which, in 1980, was a greater accuracy than for any other
physical constant.
[154]
The first video images of an electron's energy distribution were captured by a team at
Lund University in Sweden, February 2008. The scientists used extremely short flashes of light, called
attosecond pulses, which allowed an electron's motion to be observed for the first time.
[155][156]
The distribution of the electrons in solid materials can be visualized by
angle-resolved photoemission spectroscopy (ARPES). This technique employs the photoelectric effect to measure the
reciprocal space—a
mathematical representation of periodic structures that is used to
infer the original structure. ARPES can be used to determine the
direction, speed and scattering of electrons within the material.
[157]
Plasma applications
Particle beams
Electron beams are used in
welding.
[159] They allow energy densities up to
107 W·cm−2 across a narrow focus diameter of
0.1–1.3 mm
and usually require no filler material. This welding technique must be
performed in a vacuum to prevent the electrons from interacting with the
gas before reaching their target, and it can be used to join conductive
materials that would otherwise be considered unsuitable for welding.
[160][161]
Electron-beam lithography (EBL) is a method of etching semiconductors at resolutions smaller than a
micrometer.
[162]
This technique is limited by high costs, slow performance, the need to
operate the beam in the vacuum and the tendency of the electrons to
scatter in solids. The last problem limits the resolution to about
10 nm. For this reason, EBL is primarily used for the production of
small numbers of specialized
integrated circuits.
[163]
Electron beam processing is used to irradiate materials in order to change their physical properties or
sterilize medical and food products.
[164]
Electron beams fluidise or quasi-melt glasses without significant
increase of temperature on intensive irradiation: e.g. intensive
electron radiation causes a many orders of magnitude decrease of
viscosity and stepwise decrease of its activation energy.
[165]
Linear particle accelerators generate electron beams for treatment of superficial tumors in
radiation therapy.
Electron therapy can treat such skin lesions as
basal-cell carcinomas
because an electron beam only penetrates to a limited depth before
being absorbed, typically up to 5 cm for electron energies in the range
5–20 MeV. An electron beam can be used to supplement the treatment of
areas that have been irradiated by
X-rays.
[166][167]
Particle accelerators
use electric fields to propel electrons and their antiparticles to high
energies. These particles emit synchrotron radiation as they pass
through magnetic fields. The dependency of the intensity of this
radiation upon spin polarizes the electron beam—a process known as the
Sokolov–Ternov effect.
[note 8] Polarized electron beams can be useful for various experiments.
Synchrotron radiation can also
cool
the electron beams to reduce the momentum spread of the particles.
Electron and positron beams are collided upon the particles'
accelerating to the required energies;
particle detectors observe the resulting energy emissions, which
particle physics studies.
Imaging
Low-energy electron diffraction (LEED) is a method of bombarding a crystalline material with a
collimated beam
of electrons and then observing the resulting diffraction patterns to
determine the structure of the material. The required energy of the
electrons is typically in the range 20–200 eV.
[169] The
reflection high-energy electron diffraction
(RHEED) technique uses the reflection of a beam of electrons fired at
various low angles to characterize the surface of crystalline materials.
The beam energy is typically in the range 8–20 keV and the angle of
incidence is 1–4°.
[170][171]
The
electron microscope
directs a focused beam of electrons at a specimen. Some electrons
change their properties, such as movement direction, angle, and relative
phase and energy as the beam interacts with the material. Microscopists
can record these changes in the electron beam to produce atomically
resolved images of the material.
[172] In blue light, conventional
optical microscopes have a diffraction-limited resolution of about 200 nm.
[173] By comparison, electron microscopes are limited by the
de Broglie wavelength of the electron. This wavelength, for example, is equal to 0.0037 nm for electrons accelerated across a 100,000-
volt potential.
[174] The
Transmission Electron Aberration-Corrected Microscope is capable of sub-0.05 nm resolution, which is more than enough to resolve individual atoms.
[175]
This capability makes the electron microscope a useful laboratory
instrument for high resolution imaging. However, electron microscopes
are expensive instruments that are costly to maintain.
Two main types of electron microscopes exist:
transmission and
scanning. Transmission electron microscopes function like
overhead projectors, with a beam of electrons passing through a slice of material then being projected by lenses on a
photographic slide or a
charge-coupled device. Scanning electron microscopes
rasteri
a finely focused electron beam, as in a TV set, across the studied
sample to produce the image. Magnifications range from 100× to
1,000,000× or higher for both microscope types. The
scanning tunneling microscope
uses quantum tunneling of electrons from a sharp metal tip into the
studied material and can produce atomically resolved images of its
surface.
Other applications
In the
free-electron laser (FEL), a
relativistic electron beam passes through a pair of
undulators that contain arrays of
dipole magnets whose fields point in alternating directions. The electrons emit synchrotron radiation that
coherently interacts with the same electrons to strongly amplify the radiation field at the
resonance frequency. FEL can emit a coherent high-
brilliance electromagnetic radiation with a wide range of frequencies, from
microwaves
to soft X-rays. These devices are used in manufacturing, communication,
and in medical applications, such as soft tissue surgery.
[179]
Electrons are important in
cathode ray tubes, which have been extensively used as display devices in laboratory instruments,
computer monitors and
television sets.
[180] In a
photomultiplier tube, every photon striking the
photocathode initiates an avalanche of electrons that produces a detectable current pulse.
[181] Vacuum tubes
use the flow of electrons to manipulate electrical signals, and they
played a critical role in the development of electronics technology.
However, they have been largely supplanted by
solid-state devices such as the
transistor.