Uranium, 92U
 |
| General properties |
| Name, symbol |
uranium, U |
| Pronunciation |
//
ew-RAY-nee-əm |
| Appearance |
silvery gray metallic; corrodes to a spalling black oxide coat in air |
| Uranium in the periodic table |
|
|
| Atomic number |
92 |
| Standard atomic weight (±) |
238.02891(3)[1] |
| Element category |
actinide |
| Group, block |
group n/a, f-block |
| Period |
period 7 |
| Electron configuration |
[Rn] 5f3 6d1 7s2 |
| per shell |
2, 8, 18, 32, 21, 9, 2 |
| Physical properties |
| Phase |
solid |
| Melting point |
1405.3 K (1132.2 °C, 2070 °F) |
| Boiling point |
4404 K (4131 °C, 7468 °F) |
| Density near r.t. |
19.1 g·cm−3 |
| when liquid, at m.p. |
17.3 g·cm−3 |
| Heat of fusion |
9.14 kJ·mol−1 |
| Heat of vaporization |
417.1 kJ·mol−1 |
| Molar heat capacity |
27.665 J·mol−1·K−1 |
vapor pressure
| P (Pa) |
1 |
10 |
100 |
1 k |
10 k |
100 k |
| at T (K) |
2325 |
2564 |
2859 |
3234 |
3727 |
4402 |
|
| Atomic properties |
| Oxidation states |
6, 5, 4, 3,[2] 2, 1 (a weakly basic oxide) |
| Electronegativity |
Pauling scale: 1.38 |
| Ionization energies |
1st: 597.6 kJ·mol−1
2nd: 1420 kJ·mol−1 |
| Atomic radius |
empirical: 156 pm |
| Covalent radius |
196±7 pm |
| Van der Waals radius |
186 pm |
| Miscellanea |
| Crystal structure |
orthorhombic
|
| Speed of sound thin rod |
3155 m·s−1 (at 20 °C) |
| Thermal expansion |
13.9 µm·m−1·K−1 (at 25 °C) |
| Thermal conductivity |
27.5 W·m−1·K−1 |
| Electrical resistivity |
0.280 µΩ·m (at 0 °C) |
| Magnetic ordering |
paramagnetic |
| Young's modulus |
208 GPa |
| Shear modulus |
111 GPa |
| Bulk modulus |
100 GPa |
| Poisson ratio |
0.23 |
| Vickers hardness |
1960–2500 MPa |
| Brinell hardness |
2350–3850 MPa |
| CAS Registry Number |
7440-61-1 |
| History |
| Naming |
after planet Uranus, itself named after Greek god of the sky Uranus |
| Discovery |
Martin Heinrich Klaproth (1789) |
| First isolation |
Eugène-Melchior Péligot (1841) |
| Most stable isotopes |
| Main article: Isotopes of uranium |
|
|
|
Uranium is a
chemical element with symbol
U and
atomic number 92. It is a silvery-white
metal in the
actinide series of the
periodic table. A uranium atom has 92
protons and 92
electrons, of which 6 are
valence electrons. Uranium is weakly
radioactive because all its
isotopes are unstable (with half-lives of the 6 naturally known isotopes, uranium-233 to uranium-238, varying between 69 years and 4.5 billion years). The most common isotopes of uranium are
uranium-238 (which has 146
neutrons and accounts for almost 99.3% of the uranium found in nature) and
uranium-235 (which has 143 neutrons, accounting for 0.7% of the element found naturally). Uranium has the second highest
atomic weight of the
primordially occurring elements, lighter only than
plutonium.
[4] Its
density is about 70% higher than that of
lead, but slightly lower than that of
gold or
tungsten. It occurs naturally in low concentrations of a few
parts per million in soil, rock and water, and is commercially
extracted from uranium-bearing
minerals such as
uraninite.
In nature, uranium is found as uranium-238 (99.2739–99.2752%), uranium-235 (0.7198–0.7202%), and a very small amount of
uranium-234 (0.0050–0.0059%).
[5] Uranium decays slowly by emitting an
alpha particle. The
half-life of uranium-238 is about 4.47
billion years and that of uranium-235 is 704
million years,
[6] making them useful in dating the
age of the Earth.
Many contemporary uses of uranium exploit its unique
nuclear properties. Uranium-235 has the distinction of being the only naturally occurring
fissile isotope. Uranium-238 is fissionable by fast neutrons, and is
fertile, meaning it can be
transmuted to fissile
plutonium-239 in a
nuclear reactor. Another fissile isotope,
uranium-233, can be produced from natural
thorium and is also important in nuclear technology. While uranium-238 has a small probability for
spontaneous fission or even induced fission with fast neutrons, uranium-235 and to a lesser degree uranium-233 have a much higher fission cross-section for slow neutrons. In sufficient concentration, these isotopes maintain a sustained
nuclear chain reaction. This generates the heat in nuclear power reactors, and produces the fissile material for
nuclear weapons.
Depleted uranium (
238U) is used in
kinetic energy penetrators and
armor plating.
[7]
Uranium is used as a colorant in
uranium glass producing orange-red to lemon yellow hues. It was also used for tinting and shading in early
photography. The 1789
discovery of uranium in the mineral
pitchblende is credited to
Martin Heinrich Klaproth, who named the new element after the planet
Uranus.
Eugène-Melchior Péligot was the first person to isolate the metal and its radioactive properties were discovered in 1896 by
Henri Becquerel. Research by
Enrico Fermi and others, such as
J. Robert Oppenheimer starting in 1934 led to its use as a fuel in the nuclear power industry and in
Little Boy, the
first nuclear weapon used in war. An ensuing
arms race during the
Cold War between the
United States and the
Soviet Union produced tens of thousands of nuclear weapons that used uranium metal and uranium-derived
plutonium-239. The security of those weapons and their fissile material following the
breakup of the Soviet Union in 1991 is an ongoing concern for public health and safety.
[8] See
Nuclear proliferation.
Characteristics

A neutron-induced nuclear fission event involving uranium-235
When
refined, uranium is a silvery white, weakly radioactive
metal. It has a
Mohs hardness of 6, sufficient to scratch glass and approximately equal to that of
titanium,
rhodium,
manganese and
niobium. It is
malleable,
ductile, slightly
paramagnetic, strongly
electropositive and a poor
electrical conductor.
[9][10] Uranium metal has a very high
density of 18,800 kg/m
3, denser than
lead (11,340 kg/m
3), but slightly less dense than
tungsten (19,300 kg/m
3) and
gold (19,320) kg/m
3.
Uranium metal reacts with almost all non-metal elements (with an exception of the
noble gases) and their
compounds, with reactivity increasing with temperature.
[11] Hydrochloric and
nitric acids dissolve uranium, but non-oxidizing acids other than hydrochloric acid attack the element very slowly.
[9] When finely divided, it can react with cold water; in air, uranium metal becomes coated with a dark layer of
uranium oxide.
[10] Uranium in ores is extracted chemically and converted into
uranium dioxide or other chemical forms usable in industry.
Uranium-235 was the first isotope that was found to be
fissile. Other naturally occurring isotopes are fissionable, but not fissile. Upon bombardment with slow neutrons, its uranium-235
isotope will most of the time divide into two smaller
nuclei, releasing nuclear
binding energy and more neutrons. If too many of these neutrons are absorbed by other uranium-235 nuclei, a
nuclear chain reaction occurs that results in a burst of heat or (in special circumstances) an explosion. In a nuclear reactor, such a chain reaction is slowed and controlled by a
neutron poison, absorbing some of the free neutrons. Such neutron absorbent materials are often part of reactor
control rods (see
nuclear reactor physics for a description of this process of reactor control).
As little as 15 lb (7 kg) of uranium-235 can be used to make an atomic bomb.
[12] The first nuclear bomb used in war,
Little Boy, relied on uranium fission, while the very first nuclear explosive (
The gadget) and the bomb that destroyed Nagasaki (
Fat Man) were plutonium bombs.
Uranium metal has three
allotropic forms:
[13]
Applications
Military
The major application of uranium in the military sector is in high-density penetrators. This ammunition consists of
depleted uranium (DU) alloyed with 1–2% other elements. At high impact speed, the density, hardness, and
pyrophoricity of the projectile enable destruction of heavily armored targets. Tank armor and other removable
vehicle armor are also hardened with depleted uranium plates. The use of depleted uranium became politically and environmentally contentious after the use of depleted uranium munitions by the US, UK and other countries during wars in the Persian Gulf and the Balkans raised questions of uranium compounds left in the soil (see
Gulf War Syndrome).
[12]
Depleted uranium is also used as a shielding material in some containers used to store and transport radioactive materials. While the metal itself is radioactive, its high density makes it more effective than
lead in halting radiation from strong sources such as
radium.
[9] Other uses of depleted uranium include counterweights for aircraft control surfaces, as ballast for missile
re-entry vehicles and as a shielding material.
[10] Due to its high density, this material is found in
inertial guidance systems and in
gyroscopic compasses.
[10] Depleted uranium is preferred over similarly dense metals due to its ability to be easily machined and cast as well as its relatively low cost.
[14] The main risk of exposure to depleted uranium is chemical poisoning by
uranium oxide rather than radioactivity (uranium being only a weak
alpha emitter).
During the later stages of
World War II, the entire
Cold War, and to a lesser extent afterwards, uranium-235 has been used as the fissile explosive material to produce nuclear weapons. Initially, two major types of fission bombs were built: a relatively simple device that uses uranium-235 and a more complicated mechanism that uses
plutonium-239 derived from uranium-238. Later, a much more complicated and far more powerful type of fission/fusion bomb (
thermonuclear weapon) was built, that uses a plutonium-based device to cause a mixture of
tritium and
deuterium to undergo
nuclear fusion. Such bombs are jacketed in a non-fissile (unenriched) uranium case, and they derive more than half their power from the fission of this material by
fast neutrons from the nuclear fusion process.
[15]
Civilian

The most visible civilian use of uranium is as the thermal power source used in
nuclear power plants
The main use of uranium in the civilian sector is to fuel
nuclear power plants. One kilogram of uranium-235 can theoretically produce about
20 terajoules of energy (2
×10
13 joules), assuming complete fission; as much
energy as 1500
tonnes of
coal.
[7]
Commercial
nuclear power plants use fuel that is typically enriched to around 3% uranium-235.
[7] The
CANDU and
Magnox designs are the only commercial reactors capable of using unenriched uranium fuel. Fuel used for
United States Navy reactors is typically highly enriched in
uranium-235 (the exact values are
classified). In a
breeder reactor, uranium-238 can also be converted into
plutonium through the following reaction:
[10]

Before (and, occasionally, after) the discovery of radioactivity, uranium was primarily used in small amounts for yellow glass and pottery glazes, such as
uranium glass and in
Fiestaware.
[16]
The discovery and isolation of
radium in uranium ore (pitchblende) by
Marie Curie sparked the development of uranium mining to extract the radium, which was used to make glow-in-the-dark paints for clock and aircraft dials.
[17] This left a prodigious quantity of uranium as a waste product, since it takes three tonnes of uranium to extract one
gram of radium. This waste product was diverted to the glazing industry, making uranium glazes very inexpensive and abundant. Besides the pottery glazes,
uranium tile glazes accounted for the bulk of the use, including common bathroom and kitchen tiles which can be produced in green, yellow, mauve, black, blue, red and other colors.

Uranium glass used as lead-in seals in a vacuum
capacitor
Uranium was also used in
photographic chemicals (especially
uranium nitrate as a
toner),
[10] in lamp filaments for
stage lighting bulbs,
[18] to improve the appearance of
dentures,
[19] and in the leather and wood industries for stains and dyes. Uranium salts are
mordants of silk or wool. Uranyl acetate and uranyl formate are used as electron-dense "stains" in
transmission electron microscopy, to increase the contrast of biological specimens in ultrathin sections and in
negative staining of
viruses, isolated
cell organelles and
macromolecules.
The discovery of the radioactivity of uranium ushered in additional scientific and practical uses of the element. The long
half-life of the isotope uranium-238 (4.51
×10
9 years) makes it well-suited for use in estimating the age of the earliest
igneous rocks and for other types of
radiometric dating, including
uranium-thorium dating,
uranium-lead dating and
uranium-uranium dating. Uranium metal is used for
X-ray targets in the making of high-energy X-rays.
[10]
History
Prehistoric naturally occurring fission
In 1972 the French physicist
Francis Perrin discovered fifteen ancient and no longer active natural nuclear fission reactors in three separate ore deposits at the
Oklo mine in
Gabon,
West Africa, collectively known as the
Oklo Fossil Reactors. The ore deposit is 1.7 billion years old; then, uranium-235 constituted about 3% of the total uranium on Earth.
[20] This is high enough to permit a sustained nuclear fission chain reaction to occur, provided other supporting conditions exist. The capacity of the surrounding sediment to contain the
nuclear waste products has been cited by the U.S. federal government as supporting evidence for the feasibility to store spent nuclear fuel at the
Yucca Mountain nuclear waste repository.
[20]
Pre-discovery use
The use of uranium in its natural
oxide form dates back to at least the year 79
CE, when it was used to add a yellow color to
ceramic glazes.
[10] Yellow glass with 1% uranium oxide was found in a
Roman villa on Cape
Posillipo in the
Bay of Naples,
Italy, by R. T. Gunther of the
University of Oxford in 1912.
[21] Starting in the late
Middle Ages, pitchblende was extracted from the
Habsburg silver mines in
Joachimsthal,
Bohemia (now Jáchymov in the
Czech Republic), and was used as a coloring agent in the local
glassmaking industry.
[22] In the early 19th century, the world's only known sources of uranium ore were these mines.
Discovery
The
discovery of the element is credited to the German chemist
Martin Heinrich Klaproth. While he was working in his experimental laboratory in
Berlin in 1789, Klaproth was able to precipitate a yellow compound (likely
sodium diuranate) by dissolving
pitchblende in
nitric acid and neutralizing the solution with
sodium hydroxide.
[22] Klaproth assumed the yellow substance was the oxide of a yet-undiscovered element and heated it with
charcoal to obtain a black powder, which he thought was the newly discovered metal itself (in fact, that powder was an oxide of uranium).
[22][23] He named the newly discovered element after the planet
Uranus, (named after the primordial
Greek god of the sky), which had been discovered eight years earlier by
William Herschel.
[24]
In 1841,
Eugène-Melchior Péligot, Professor of Analytical Chemistry at the
Conservatoire National des Arts et Métiers (Central School of Arts and Manufactures) in
Paris, isolated the first sample of uranium metal by heating
uranium tetrachloride with
potassium.
[22][25] Uranium was not seen as being particularly dangerous during much of the 19th century, leading to the development of various uses for the element. One such use for the oxide was the aforementioned but no longer secret coloring of pottery and glass.
Henri Becquerel discovered
radioactivity by using uranium in 1896.
[11] Becquerel made the discovery in Paris by leaving a sample of a uranium salt, K
2UO
2(SO
4)
2 (potassium uranyl sulfate), on top of an unexposed
photographic plate in a drawer and noting that the plate had become "fogged".
[26] He determined that a form of invisible light or rays emitted by uranium had exposed the plate.
Fission research

Cubes and cuboids of uranium produced during the Manhattan project
A team led by
Enrico Fermi in 1934 observed that bombarding uranium with neutrons produces the emission of
beta rays (
electrons or
positrons from the elements produced; see
beta particle).
[27] The fission products were at first mistaken for new elements of atomic numbers 93 and 94, which the Dean of the Faculty of Rome, Orso Mario Corbino, christened
ausonium and
hesperium, respectively.
[28][29][30][31] The experiments leading to the discovery of uranium's ability to fission (break apart) into lighter elements and release
binding energy were conducted by
Otto Hahn and
Fritz Strassmann[27] in Hahn's laboratory in Berlin.
Lise Meitner and her nephew, the physicist
Otto Robert Frisch, published the physical explanation in February 1939 and named the process "
nuclear fission".
[32] Soon after, Fermi hypothesized that the fission of uranium might release enough neutrons to sustain a fission reaction. Confirmation of this hypothesis came in 1939, and later work found that on average about 2.5 neutrons are released by each fission of the rare uranium isotope uranium-235.
[27] Further work found that the far more common uranium-238 isotope can be
transmuted into plutonium, which, like uranium-235, is also fissionable by thermal neutrons. These discoveries led numerous countries to begin working on the development of nuclear weapons and
nuclear power.
On 2 December 1942, as part of the
Manhattan Project, another team led by Enrico Fermi was able to initiate the first artificial self-sustained
nuclear chain reaction,
Chicago Pile-1. Working in a lab below the stands of
Stagg Field at the
University of Chicago, the team created the conditions needed for such a reaction by piling together 400 short tons (360 metric tons) of
graphite, 58 short tons (53 metric tons) of
uranium oxide, and six short tons (5.5 metric tons) of uranium metal, a majority of which was supplied by
Westinghouse Lamp Plant in a makeshift production process.
[27][33]
Nuclear weaponry
Two major types of atomic bombs were developed by the United States during
World War II: a uranium-based device (codenamed "
Little Boy") whose fissile material was highly
enriched uranium, and a plutonium-based device (see
Trinity test and "
Fat Man") whose plutonium was derived from uranium-238. The uranium-based Little Boy device became the first nuclear weapon used in war when it was detonated over the
Japanese city of
Hiroshima on 6 August 1945. Exploding with a yield equivalent to 12,500 tonnes of
TNT, the blast and thermal wave of the bomb destroyed nearly 50,000 buildings and killed approximately 75,000 people (see
Atomic bombings of Hiroshima and Nagasaki).
[26] Initially it was believed that uranium was relatively rare, and that
nuclear proliferation could be avoided by simply buying up all known uranium stocks, but within a decade large deposits of it were discovered in many places around the world.
[34]
Reactors

Four light bulbs lit with electricity generated from the first artificial electricity-producing nuclear reactor,
EBR-I (1951)
The
X-10 Graphite Reactor at
Oak Ridge National Laboratory (ORNL) in Oak Ridge, Tennessee, formerly known as the Clinton Pile and X-10 Pile, was the world's second artificial nuclear reactor (after Enrico Fermi's Chicago Pile) and was the first reactor designed and built for continuous operation.
Argonne National Laboratory's
Experimental Breeder Reactor I, located at the Atomic Energy Commission's National Reactor Testing Station near
Arco, Idaho, became the first nuclear reactor to create electricity on 20 December 1951.
[35] Initially, four 150-watt light bulbs were lit by the reactor, but improvements eventually enabled it to power the whole facility (later, the town of Arco became the first in the world to have all its
electricity come from nuclear power generated by
BORAX-III, another reactor designed and operated by
Argonne National Laboratory).
[36][37] The world's first commercial scale nuclear power station,
Obninsk in the
Soviet Union, began generation with its reactor AM-1 on 27 June 1954. Other early nuclear power plants were
Calder Hall in
England which began generation on 17 October 1956
[38] and the
Shippingport Atomic Power Station in
Pennsylvania which began on 26 May 1958. Nuclear power was used for the first time for propulsion by a
submarine, the
USS Nautilus, in 1954.
[27][39]
Contamination and the Cold War legacy
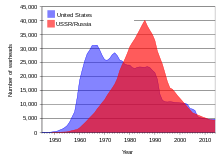
U.S. and USSR/Russian nuclear weapons stockpiles, 1945–2005
Above-ground
nuclear tests by the Soviet Union and the United States in the 1950s and early 1960s and by
France into the 1970s and 1980s
[14] spread a significant amount of
fallout from uranium daughter isotopes around the world.
[40] Additional fallout and pollution occurred from several
nuclear accidents.
[41][42]
Uranium miners have a higher incidence of
cancer. An excess risk of lung cancer among
Navajo uranium miners, for example, has been documented and linked to their occupation.
[43] The
Radiation Exposure Compensation Act, a 1990 law in the USA, required $100,000 in "compassion payments" to uranium miners diagnosed with cancer or other respiratory ailments.
[44]
During the
Cold War between the Soviet Union and the United States, huge stockpiles of uranium were amassed and tens of thousands of nuclear weapons were created using enriched uranium and plutonium made from uranium. Since the
break-up of the Soviet Union in 1991, an estimated 600 short tons (540 metric tons) of highly enriched weapons grade uranium (enough to make 40,000 nuclear warheads) have been stored in often inadequately guarded facilities in the
Russian Federation and several other former Soviet states.
[12] Police in
Asia,
Europe, and
South America on at least 16 occasions from 1993 to 2005 have
intercepted shipments of smuggled bomb-grade uranium or plutonium, most of which was from ex-Soviet sources.
[12] From 1993 to 2005 the Material Protection, Control, and Accounting Program, operated by the
federal government of the United States, spent approximately
US $550 million to help safeguard uranium and plutonium stockpiles in Russia.
[12] This money was used for improvements and security enhancements at research and storage facilities.
Scientific American reported in February 2006 that in some of the facilities security consisted of chain link fences which were in severe states of disrepair. According to an interview from the article, one facility had been storing samples of enriched (weapons grade) uranium in a broom closet before the improvement project; another had been keeping track of its stock of nuclear warheads using index cards kept in a shoe box.
[45]
Occurrence
Biotic and abiotic

Uraninite, also known as pitchblende, is the most common ore mined to extract uranium

The evolution of Earth's
radiogenic heat flow over time: contribution from
235U in pink and from
238U in light blue
Uranium is a
naturally occurring element that can be found in low levels within all rock, soil, and water. Uranium is the 51st element in order of
abundance in the Earth's crust. Uranium is also the highest-numbered element to be found naturally in significant quantities on Earth and is almost always found combined with other elements.
[10] Along with all elements having
atomic weights higher than that of
iron, it is only naturally formed in
supernovae.
[46] The decay of uranium,
thorium, and
potassium-40 in the Earth's
mantle is thought to be the main source of heat
[47][48] that keeps the
outer core liquid and drives
mantle convection, which in turn drives
plate tectonics.
Uranium's average concentration in the
Earth's
crust is (depending on the reference) 2 to 4 parts per million,
[9][14] or about 40 times as abundant as
silver.
[11] The Earth's crust from the surface to 25 km (15 mi) down is calculated to contain 10
17 kg (2
×10
17 lb) of uranium while the
oceans may contain 10
13 kg (2
×10
13 lb).
[9] The concentration of uranium in soil ranges from 0.7 to 11 parts per million (up to 15 parts per million in farmland soil due to use of phosphate
fertilizers), and its concentration in sea water is 3 parts per billion.
[14]
Uranium is more plentiful than
antimony,
tin,
cadmium,
mercury, or silver, and it is about as abundant as
arsenic or
molybdenum.
[10][14] Uranium is found in hundreds of minerals including uraninite (the most common uranium
ore),
carnotite,
autunite,
uranophane,
torbernite, and
coffinite.
[10] Significant concentrations of uranium occur in some substances such as
phosphate rock deposits, and minerals such as
lignite, and
monazite sands in uranium-rich ores
[10] (it is recovered commercially from sources with as little as 0.1% uranium
[11]).

Citrobacter species can have concentrations of uranium in their bodies 300 times higher than in the surrounding environment
Some bacteria such as
S. putrefaciens and
G. metallireducens have been shown to reduce U(VI) to U(IV).
[49]
Some organisms, such as the lichen
Trapelia involuta or
microorganisms such as the
bacterium Citrobacter, can absorb concentrations of uranium that are up to 300 times higher than in their environment.
[50] Citrobacter species absorb
uranyl ions when given
glycerol phosphate (or other similar organic phosphates). After one day, one gram of bacteria can encrust themselves with nine grams of uranyl phosphate crystals; this creates the possibility that these organisms could be used in
bioremediation to
decontaminate uranium-polluted water.
[22][51] The proteobacterium
Geobacter has also been shown to bioremediate uranium in ground water.
[52] The mycorrhizal fungus
Glomus intraradices increases uranium content in the roots of its symbiotic plant.
[53]
In nature, uranium(VI) forms highly soluble carbonate complexes at alkaline pH. This leads to an increase in mobility and availability of uranium to groundwater and soil from nuclear wastes which leads to health hazards. However, it is difficult to precipitate uranium as phosphate in the presence of excess carbonate at alkaline pH. A
Sphingomonas sp. strain BSAR-1 has been found to express a high activity alkaline phosphatase (PhoK) that has been applied for bioprecipitation of uranium as uranyl phosphate species from alkaline solutions. The precipitation ability was enhanced by overexpressing PhoK protein in
E. coli.
[54]
Plants absorb some uranium from soil. Dry weight concentrations of uranium in plants range from 5 to 60 parts per billion, and ash from burnt wood can have concentrations up to 4 parts per million.
[22] Dry weight concentrations of uranium in
food plants are typically lower with one to two micrograms per day ingested through the food people eat.
[22]
Production and mining
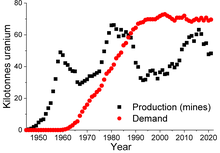
World uranium production (mines) and demand
[55]

Yellowcake is a concentrated mixture of uranium oxides that is further refined to extract pure uranium
The worldwide production of uranium in 2010 amounted to 53,663
tonnes, of which 17,803 t (33.2%) was mined in
Kazakhstan. Other important uranium mining countries are
Canada (9,783 t),
Australia (5,900 t),
Namibia (4,496 t),
Niger (4,198 t) and
Russia (3,562 t).
[55]
Uranium ore is mined in several ways: by
open pit,
underground,
in-situ leaching, and
borehole mining (see
uranium mining).
[7] Low-grade uranium ore mined typically contains 0.01 to 0.25% uranium oxides. Extensive measures must be employed to extract the metal from its ore.
[56] High-grade ores found in
Athabasca Basin deposits in
Saskatchewan, Canada can contain up to 23% uranium oxides on average.
[57] Uranium ore is crushed and rendered into a fine powder and then leached with either an
acid or
alkali. The
leachate is subjected to one of several sequences of precipitation, solvent extraction, and ion exchange. The resulting mixture, called
yellowcake, contains at least 75% uranium oxides U
3O
8. Yellowcake is then
calcined to remove impurities from the milling process before refining and conversion.
[58]
Commercial-grade uranium can be produced through the
reduction of uranium
halides with
alkali or
alkaline earth metals.
[10] Uranium metal can also be prepared through
electrolysis of
KUF
5 or
UF
4, dissolved in molten
calcium chloride (
CaCl
2) and
sodium chloride (
NaCl) solution.
[10] Very pure uranium is produced through the
thermal decomposition of uranium halides on a hot filament.
[10]
Resources and reserves
It is estimated that 5.5 million tonnes of uranium exists in ore reserves that are economically viable at US$59 per lb of uranium,
[59] while 35 million tonnes are classed as mineral resources (reasonable prospects for eventual economic extraction).
[60] Prices went from about $10/lb in May 2003 to $138/lb in July 2007. This has caused a big increase in spending on exploration,
[59] with US$200 million being spent world wide in 2005, a 54% increase on the previous year.
[60] This trend continued through 2006, when expenditure on exploration rocketed to over $774 million, an increase of over 250% compared to 2004. The
OECD Nuclear Energy Agency said exploration figures for 2007 would likely match those for 2006.
[59]
Australia has 31% of the world's known uranium ore reserves
[61] and the world's largest single uranium deposit, located at the
Olympic Dam Mine in
South Australia.
[62] There is a significant reserve of uranium in
Bakouma a
sub-prefecture in the
prefecture of
Mbomou in
Central African Republic.
Some nuclear fuel comes from nuclear weapons being dismantled,
[63] such as from the
Megatons to Megawatts Program.
An additional 4.6 billion tonnes of uranium are estimated to be in
sea water (
Japanese scientists in the 1980s showed that extraction of uranium from sea water using
ion exchangers was technically feasible).
[64][65] There have been experiments to extract uranium from sea water,
[66] but the yield has been low due to the carbonate present in the water. In 2012,
ORNL researchers announced the successful development of a new absorbent material dubbed HiCap which performs surface retention of solid or gas molecules, atoms or ions and also effectively removes toxic metals from water, according to results verified by researchers at Pacific Northwest National Laboratory.
[67][68]
Supplies
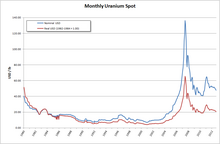
In 2005, seventeen countries produced concentrated uranium oxides, with
Canada (27.9% of world production) and
Australia (22.8%) being the largest producers and
Kazakhstan (10.5%),
Russia (8.0%),
Namibia (7.5%),
Niger (7.4%),
Uzbekistan (5.5%), the
United States (2.5%),
Argentina (2.1%),
Ukraine (1.9%) and
China (1.7%) also producing significant amounts.
[70] Kazakhstan continues to increase production and may have become the world's largest producer of uranium by 2009 with an expected production of 12,826 tonnes, compared to Canada with 11,100 t and Australia with 9,430 t.
[71][72] In the late 1960s, UN geologists also discovered major uranium deposits and other rare mineral reserves in
Somalia. The find was the largest of its kind, with industry experts estimating the deposits at over 25% of the world's then known uranium reserves of 800,000 tons.
[73]
The ultimate available uranium is believed to be sufficient for at least the next 85 years,
[60] although some studies indicate underinvestment in the late twentieth century may produce supply problems in the 21st century.
[74] Uranium deposits seem to be log-normal distributed. There is a 300-fold increase in the amount of uranium recoverable for each tenfold decrease in ore grade.
[75] In other words, there is little high grade ore and proportionately much more low grade ore available.
Compounds
Oxidation states and oxides
Oxides
Calcined uranium yellowcake as produced in many large mills contains a distribution of uranium oxidation species in various forms ranging from most oxidized to least oxidized. Particles with short residence times in a calciner will generally be less oxidized than those with long retention times or particles recovered in the stack scrubber. Uranium content is usually referenced to
U
3O
8, which dates to the days of the
Manhattan project when
U
3O
8 was used as an analytical chemistry reporting standard.
Phase relationships in the uranium-oxygen system are complex. The most important oxidation states of uranium are uranium(IV) and uranium(VI), and their two corresponding
oxides are, respectively,
uranium dioxide (
UO
2) and
uranium trioxide (
UO
3).
[76] Other
uranium oxides such as uranium monoxide (UO), diuranium pentoxide (
U
2O
5), and uranium peroxide (
UO
4·2H
2O) also exist.
The most common forms of uranium oxide are
triuranium octoxide (
U
3O
8) and
UO
2.
[77] Both oxide forms are solids that have low solubility in water and are relatively stable over a wide range of environmental conditions. Triuranium octoxide is (depending on conditions) the most stable compound of uranium and is the form most commonly found in nature. Uranium dioxide is the form in which uranium is most commonly used as a nuclear reactor fuel.
[77] At ambient temperatures,
UO
2 will gradually convert to
U
3O
8. Because of their stability, uranium oxides are generally considered the preferred chemical form for storage or disposal.
[77]
Aqueous chemistry

Uranium in its oxidation states III, IV, V, VI
Salts of many
oxidation states of uranium are water-
soluble and may be studied in
aqueous solutions. The most common ionic forms are
U3+ (brown-red),
U4+ (green),
UO+
2 (unstable), and
UO2+
2 (yellow), for U(III), U(IV), U(V), and U(VI), respectively.
[78] A few
solid and semi-metallic compounds such as UO and US exist for the formal oxidation state uranium(II), but no simple ions are known to exist in solution for that state. Ions of
U3+ liberate
hydrogen from
water and are therefore considered to be highly unstable. The
UO2+
2 ion represents the uranium(VI) state and is known to form compounds such as
uranyl carbonate,
uranyl chloride and
uranyl sulfate.
UO2+
2 also forms
complexes with various
organic chelating agents, the most commonly encountered of which is
uranyl acetate.
[78]
Unlike the uranyl salts of uranium and
polyatomic ion uranium-oxide cationic forms, the
uranates, salts containing a polyatomic uranium-oxide anion, are generally not water-soluble.
Carbonates
The interactions of carbonate anions with uranium(VI) cause the
Pourbaix diagram to change greatly when the medium is changed from water to a carbonate containing solution. While the vast majority of carbonates are insoluble in water (students are often taught that all carbonates other than those of alkali metals are insoluble in water), uranium carbonates are often soluble in water. This is because a U(VI) cation is able to bind two terminal oxides and three or more carbonates to form anionic complexes.
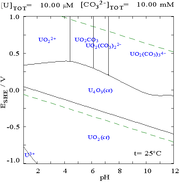
Pourbaix diagrams[79]
|
|
|
| Uranium in a non-complexing aqueous medium (e.g. perchloric acid/sodium hydroxide).[79] |
Uranium in carbonate solution |
|
|
|
| Relative concentrations of the different chemical forms of uranium in a non-complexing aqueous medium (e.g. perchloric acid/sodium hydroxide).[79] |
Relative concentrations of the different chemical forms of uranium in an aqueous carbonate solution.[79] |
Effects of pH
The uranium fraction diagrams in the presence of carbonate illustrate this further: when the pH of a uranium(VI) solution increases, the uranium is converted to a hydrated uranium oxide hydroxide and at high pHs it becomes an anionic hydroxide complex.
When carbonate is added, uranium is converted to a series of carbonate complexes if the pH is increased. One effect of these reactions is increased solubility of uranium in the pH range 6 to 8, a fact which has a direct bearing on the long term stability of spent uranium dioxide nuclear fuels.
Hydrides, carbides and nitrides
Uranium metal heated to 250 to 300 °C (482 to 572 °F) reacts with
hydrogen to form
uranium hydride. Even higher temperatures will reversibly remove the hydrogen. This property makes uranium hydrides convenient starting materials to create reactive uranium powder along with various uranium
carbide,
nitride, and
halide compounds.
[80] Two crystal modifications of uranium hydride exist: an α form that is obtained at low temperatures and a β form that is created when the formation temperature is above 250 °C.
[80]
Uranium carbides and
uranium nitrides are both relatively
inert semimetallic compounds that are minimally soluble in
acids, react with water, and can ignite in
air to form
U
3O
8.
[80] Carbides of uranium include uranium monocarbide (U
C), uranium dicarbide (
UC
2), and diuranium tricarbide (
U
2C
3). Both UC and
UC
2 are formed by adding carbon to molten uranium or by exposing the metal to
carbon monoxide at high temperatures. Stable below 1800 °C,
U
2C
3 is prepared by subjecting a heated mixture of UC and
UC
2 to mechanical stress.
[81] Uranium nitrides obtained by direct exposure of the metal to
nitrogen include uranium mononitride (UN), uranium dinitride (
UN
2), and diuranium trinitride (
U
2N
3).
[81]
Halides
All uranium fluorides are created using
uranium tetrafluoride (
UF
4);
UF
4 itself is prepared by hydrofluorination of uranium dioxide.
[80] Reduction of
UF
4 with hydrogen at 1000 °C produces uranium trifluoride (
UF
3). Under the right conditions of temperature and pressure, the reaction of solid
UF
4 with gaseous
uranium hexafluoride (
UF
6) can form the intermediate fluorides of
U
2F
9,
U
4F
17, and
UF
5.
[80]
At room temperatures,
UF
6 has a high
vapor pressure, making it useful in the
gaseous diffusion process to separate the rare uranium-235 from the common uranium-238 isotope. This compound can be prepared from uranium dioxide and uranium hydride by the following process:
[80]
- UO
2 + 4 HF → UF
4 + 2 H
2O (500 °C, endothermic)
- UF
4 + F
2 → UF
6 (350 °C, endothermic)
The resulting
UF
6, a white solid, is highly
reactive (by fluorination), easily
sublimes (emitting a vapor that behaves as a nearly
ideal gas), and is the most volatile compound of uranium known to exist.
[80]
One method of preparing
uranium tetrachloride (
UCl
4) is to directly combine
chlorine with either uranium metal or uranium hydride. The reduction of
UCl
4 by hydrogen produces uranium trichloride (
UCl
3) while the higher chlorides of uranium are prepared by reaction with additional chlorine.
[80] All uranium chlorides react with water and air.
Bromides and
iodides of uranium are formed by direct reaction of, respectively,
bromine and
iodine with uranium or by adding
UH
3 to those element's acids.
[80] Known examples include:
UBr
3,
UBr
4,
UI
3, and
UI
4. Uranium oxyhalides are water-soluble and include
UO
2F
2,
UOCl
2,
UO
2Cl
2, and
UO
2Br
2. Stability of the oxyhalides decrease as the
atomic weight of the component halide increases.
[80]
Isotopes
Natural concentrations
Natural uranium consists of three major
isotopes:
uranium-238 (99.28%
natural abundance), uranium-235 (0.71%), and
uranium-234 (0.0054%). All three are
radioactive, emitting
alpha particles, with the exception that all three of these isotopes have small probabilities of undergoing
spontaneous fission, rather than
alpha emission.
Uranium-238 is the most stable isotope of uranium, with a
half-life of about 4.468
×10
9 years, roughly the
age of the Earth. Uranium-235 has a half-life of about 7.13
×10
8 years, and uranium-234 has a half-life of about 2.48
×10
5 years.
[82] For natural uranium, about 49% of its alpha rays are emitted by each of
238U atom, and also 49% by
234U (since the latter is formed from the former) and about 2.0% of them by the
235U. When the Earth was young, probably about one-fifth of its uranium was uranium-235, but the percentage of
234U was probably much lower than this.
Uranium-238 is usually an α emitter (occasionally, it undergoes spontaneous fission), decaying through the
"Uranium Series" of nuclear decay, which has 18 members, all of which eventually decay into
lead-206, by a variety of different decay paths.
[11]
The
decay series of 235U, which is called the
actinium series has 15 members, all of which eventually decay into lead-207.
[11] The constant rates of decay in these decay series makes the comparison of the ratios of parent to daughter elements useful in
radiometric dating.
Uranium-234 is a member of the
"Uranium Series", and it decays to lead-206 through a series of relatively short-lived isotopes.
Uranium-233 is made from
thorium-232 by neutron bombardment, usually in a nuclear reactor, and
233U is also fissile.
[10] Its decay series ends with
thallium-205.
Uranium-235 is important for both
nuclear reactors and
nuclear weapons, because it is the only uranium isotope existing in nature on Earth in any significant amount that is
fissile. This means it can be split into two or three fragments (
fission products) by thermal neutrons.
[11]
Uranium-238 is not fissile, but is a fertile isotope, because after
neutron activation it can produce
plutonium-239, another fissile isotope. Indeed, the
238U nucleus can absorb one neutron to produce the radioactive isotope
uranium-239.
239U decays by
beta emission to
neptunium-239, also a beta-emitter, that decays in its turn, within a few days into plutonium-239.
239Pu was used as fissile material in the first
atomic bomb detonated in the "
Trinity test" on 15 July 1945 in
New Mexico.
[27]
Enrichment

Cascades of
gas centrifuges are used to enrich uranium ore to concentrate its fissionable isotopes
In nature, uranium is found as uranium-238 (99.2742%) and uranium-235 (0.7204%).
Isotope separation concentrates (enriches) the fissionable uranium-235 for nuclear weapons and most nuclear power plants, except for
gas cooled reactors and
pressurised heavy water reactors. Most neutrons released by a fissioning atom of uranium-235 must impact other uranium-235 atoms to sustain the
nuclear chain reaction. The concentration and amount of uranium-235 needed to achieve this is called a '
critical mass'.
To be considered 'enriched', the uranium-235 fraction should be between 3% and 5%.
[83] This process produces huge quantities of uranium that is depleted of uranium-235 and with a correspondingly increased fraction of uranium-238, called depleted uranium or 'DU'. To be considered 'depleted', the uranium-235 isotope concentration should be no more than 0.3%.
[84] The price of uranium has risen since 2001, so enrichment tailings containing more than 0.35% uranium-235 are being considered for re-enrichment, driving the price of depleted uranium hexafluoride above $130 per kilogram in July 2007 from $5 in 2001.
[84]
The
gas centrifuge process, where gaseous
uranium hexafluoride (
UF
6) is separated by the difference in molecular weight between
235UF
6 and
238UF
6 using high-speed
centrifuges, is the cheapest and leading enrichment process.
[26] The
gaseous diffusion process had been the leading method for enrichment and was used in the
Manhattan Project. In this process, uranium hexafluoride is repeatedly
diffused through a
silver-
zinc membrane, and the different isotopes of uranium are separated by diffusion rate (since uranium 238 is heavier it diffuses slightly slower than uranium-235).
[26] The
molecular laser isotope separation method employs a
laser beam of precise energy to sever the bond between uranium-235 and fluorine. This leaves uranium-238 bonded to fluorine and allows uranium-235 metal to precipitate from the solution.
[7] An alternative laser method of enrichment is known as
atomic vapor laser isotope separation (AVLIS) and employs visible
tunable lasers such as
dye lasers.
[85] Another method used is liquid thermal diffusion.
[9]
Human exposure
A person can be exposed to uranium (or its radioactive daughters such as
radon) by inhaling dust in air or by ingesting contaminated water and food. The amount of uranium in air is usually very small; however, people who work in factories that process
phosphate fertilizers, live near government facilities that made or tested nuclear weapons, live or work near a modern battlefield where depleted uranium
weapons have been used, or live or work near a
coal-fired power plant, facilities that mine or process uranium ore, or enrich uranium for reactor fuel, may have increased exposure to uranium.
[86][87] Houses or structures that are over uranium deposits (either natural or man-made slag deposits) may have an increased incidence of exposure to radon gas.
Most ingested uranium is excreted during
digestion. Only 0.5% is absorbed when insoluble forms of uranium, such as its oxide, are ingested, whereas absorption of the more soluble
uranyl ion can be up to 5%.
[22] However, soluble uranium compounds tend to quickly pass through the body whereas insoluble uranium compounds, especially when inhaled by way of dust into the
lungs, pose a more serious exposure hazard. After entering the bloodstream, the absorbed uranium tends to
bioaccumulate and stay for many years in
bone tissue because of uranium's affinity for phosphates.
[22] Uranium is not absorbed through the skin, and
alpha particles released by uranium cannot penetrate the skin.
Incorporated uranium becomes
uranyl ions, which accumulate in bone, liver, kidney, and reproductive tissues. Uranium can be decontaminated from steel surfaces
[88] and
aquifers.
[89]
Effects and precautions
Normal functioning of the
kidney,
brain,
liver,
heart, and other systems can be affected by uranium exposure, because, besides being weakly radioactive, uranium is a
toxic metal.
[22][90][91] Uranium is also a reproductive toxicant.
[92][93] Radiological effects are generally local because alpha radiation, the primary form of
238U decay, has a very short range, and will not penetrate skin.
Uranyl (
UO2+
2) ions, such as from
uranium trioxide or uranyl nitrate and other hexavalent uranium compounds, have been shown to cause birth defects and immune system damage in laboratory animals.
[94] While the CDC has published one study that no human
cancer has been seen as a result of exposure to natural or depleted uranium,
[95] exposure to uranium and its decay products, especially
radon, are widely known and significant health threats.
[14] Exposure to
strontium-90,
iodine-131, and other fission products is unrelated to uranium exposure, but may result from medical procedures or exposure to spent reactor fuel or fallout from nuclear weapons.
[96] Although accidental inhalation exposure to a high concentration of
uranium hexafluoride has resulted in human fatalities, those deaths were associated with generation of highly toxic hydrofluoric acid and
uranyl fluoride rather than with uranium itself.
[97] Finely divided uranium metal presents a fire hazard because uranium is
pyrophoric; small grains will ignite spontaneously in air at room temperature.
[10]
Uranium metal is commonly handled with gloves as a sufficient precaution.
[98] Uranium concentrate is handled and contained so as to ensure that people do not inhale or ingest it.
[98]
Compilation of 2004 review on uranium toxicity[90]
| Body system |
Human studies |
Animal studies |
In vitro |
| Renal |
Elevated levels of protein excretion, urinary catalase and diuresis |
Damage to proximal convoluted tubules, necrotic cells cast from tubular epithelium, glomerular changes (renal failure) |
No studies |
| Brain/CNS |
Decreased performance on neurocognitive tests |
Acute cholinergic toxicity; Dose-dependent accumulation in cortex, midbrain, and vermis; Electrophysiological changes in hippocampus |
No studies |
| DNA |
Increased reports of cancers[44][99][100][101][102][103] |
Increased mutagenicity (in mice) and induction of tumors |
Binucleated cells with micronuclei, Inhibition of cell cycle kinetics and proliferation; Sister chromatid induction, tumorigenic phenotype |
| Bone/muscle |
No studies |
Inhibition of periodontal bone formation; and alveolar wound healing |
No studies |
| Reproductive |
Uranium miners have more first born female children |
Moderate to severe focal tubular atrophy; vacuolization of Leydig cells |
No studies |
| Lungs/respiratory |
No adverse health effects reported |
Severe nasal congestion and hemorrhage, lung lesions and fibrosis, edema and swelling, lung cancer |
No studies |
| Gastrointestinal |
Vomiting, diarrhea, albuminuria |
No studies |
No studies |
| Liver |
No effects seen at exposure dose |
Fatty livers, focal necrosis |
No studies |
| Skin |
No exposure assessment data available |
Swollen vacuolated epidermal cells, damage to hair follicles and sebaceous glands |
No studies |
| Tissues surrounding embedded DU fragments |
Elevated uranium urine concentrations |
Elevated uranium urine concentrations, perturbations in biochemical and neuropsychological testing |
No studies |
| Immune system |
Chronic fatigue, rash, ear and eye infections, hair and weight loss, cough. May be due to combined chemical exposure rather than DU alone |
No studies |
No studies |
| Eyes |
No studies |
Conjunctivitis, irritation inflammation, edema, ulceration of conjunctival sacs |
No studies |
| Blood |
No studies |
Decrease in RBC count and hemoglobin concentration |
No studies |
| Cardiovascular |
Myocarditis resulting from the uranium ingestion, which ended 6 months after ingestion |
No effects |
No studies |