From Wikipedia, the free encyclopedia
As the temperature decreases, the peak of the black-body radiation curve
moves to lower intensities and longer wavelengths. The black-body
radiation graph is also compared with the classical model of Rayleigh
and Jeans.
Black-body radiation is the
thermal electromagnetic radiation within or surrounding a body in
thermodynamic equilibrium with its environment, or emitted by a
black body
(an opaque and non-reflective body). It has a specific spectrum and
intensity that depends only on the body's temperature, which is assumed
for the sake of calculations and theory to be uniform and constant.
[1][2][3][4]
The thermal radiation spontaneously emitted by many ordinary objects
can be approximated as black-body radiation. A perfectly insulated
enclosure that is in thermal equilibrium internally contains black-body
radiation and will emit it through a hole made in its wall, provided the
hole is small enough to have negligible effect upon the equilibrium.
A black-body at room temperature appears black, as most of the energy it radiates is
infra-red
and cannot be perceived by the human eye. Because the human eye cannot
perceive color at very low light intensities, a black body, viewed in
the dark at the lowest just faintly visible temperature, subjectively
appears grey (but only because the human eye is sensitive only to black
and white at very low intensities - in reality, the frequency of the
light in the visible range would still be red, although the intensity
would be too low to discern as red), even though its objective physical
spectrum peaks in the infrared range.
[5] When it becomes a little hotter, it appears dull red. As its temperature increases further it eventually becomes blue-white.
Although planets and stars are neither in thermal equilibrium with their surroundings nor perfect
black bodies, black-body radiation is used as a first approximation for the energy they emit.
[6] Black holes
are near-perfect black bodies, in the sense that they absorb all the
radiation that falls on them. It has been proposed that they emit
black-body radiation (called
Hawking radiation), with a temperature that depends on the mass of the black hole.
[7]
The term
black body was introduced by
Gustav Kirchhoff in 1860. Black-body radiation is also called
thermal radiation,
cavity radiation,
complete radiation or
temperature radiation.
Spectrum
Black-body radiation has a characteristic, continuous
frequency spectrum that depends only on the body's temperature,
[8] called the Planck spectrum or
Planck's law. The spectrum is peaked at a characteristic frequency that shifts to higher frequencies with increasing temperature, and at
room temperature most of the emission is in the
infrared region of the
electromagnetic spectrum.
[9][10][11]
As the temperature increases past about 500 degrees
Celsius,
black bodies start to emit significant amounts of visible light. Viewed
in the dark by the human eye, the first faint glow appears as a
"ghostly" grey (the visible light is actually red, but low intensity
light activates only the eye's grey-level sensors). With rising
temperature, the glow becomes visible even when there is some background
surrounding light: first as a dull red, then yellow, and eventually a
"dazzling bluish-white" as the temperature rises.
[12][13] When the body appears white, it is emitting a substantial fraction of its energy as
ultraviolet radiation. The Sun, with an
effective temperature of approximately 5800 K,
[14] is an approximate black body with an emission spectrum peaked in the central, yellow-green part of the
visible spectrum, but with significant power in the ultraviolet as well.
Black-body radiation provides insight into the
thermodynamic equilibrium state of cavity radiation. If each
Fourier mode
of the equilibrium radiation in an otherwise empty cavity with
perfectly reflective walls is considered as a degree of freedom capable
of exchanging energy, then, according to the
equipartition theorem
of classical physics, there would be an equal amount of energy in each
mode. Since there are an infinite number of modes this implies infinite
heat capacity
(infinite energy at any non-zero temperature), as well as an unphysical
spectrum of emitted radiation that grows without bound with increasing
frequency, a problem known as the
ultraviolet catastrophe. Instead, in quantum theory the
occupation numbers
of the modes are quantized, cutting off the spectrum at high frequency
in agreement with experimental observation and resolving the
catastrophe. The study of the laws of black bodies and the failure of
classical physics to describe them helped establish the foundations of
quantum mechanics.
Explanation
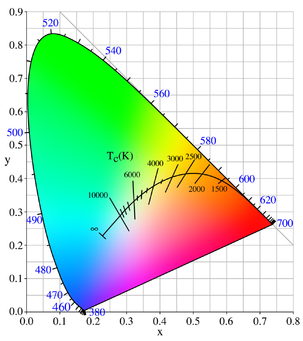
Color of a black body from 800 K to 12200 K. This range of colors
approximates the range of colors of stars of different temperatures, as
seen or photographed in the night sky.
All normal (
baryonic) matter emits electromagnetic radiation when it has a temperature above
absolute zero. The radiation represents a conversion of a body's thermal energy into electromagnetic energy, and is therefore called
thermal radiation. It is a
spontaneous process of radiative distribution of
entropy.
Conversely all normal matter absorbs electromagnetic radiation to
some degree. An object that absorbs all radiation falling on it, at all
wavelengths,
is called a black body. When a black body is at a uniform temperature,
its emission has a characteristic frequency distribution that depends on
the temperature. Its emission is called black-body radiation.
The concept of the black body is an idealization, as perfect black bodies do not exist in nature.
[15] Graphite and
lamp black,
with emissivities greater than 0.95, however, are good approximations
to a black material. Experimentally, black-body radiation may be
established best as the ultimately stable steady state equilibrium
radiation in a cavity in a rigid body, at a uniform temperature, that is
entirely opaque and is only partly reflective.
[15]
A closed box of graphite walls at a constant temperature with a small
hole on one side produces a good approximation to ideal black-body
radiation emanating from the opening.
[16][17]
Black-body radiation has the unique absolutely stable distribution of
radiative intensity that can persist in thermodynamic equilibrium in a
cavity.
[15]
In equilibrium, for each frequency the total intensity of radiation
that is emitted and reflected from a body (that is, the net amount of
radiation leaving its surface, called the
spectral radiance) is determined solely by the equilibrium temperature, and does not depend upon the shape, material or structure of the body.
[18]
For a black body (a perfect absorber) there is no reflected radiation,
and so the spectral radiance is due entirely to emission. In addition, a
black body is a diffuse emitter (its emission is independent of
direction). Consequently, black-body radiation may be viewed as the
radiation from a black body at thermal equilibrium.
Black-body radiation becomes a visible glow of light if the temperature of the object is high enough. The
Draper point is the temperature at which all solids glow a dim red, about 798 K.
[19][20]
At 1000 K, a small opening in the wall of a large uniformly heated
opaque-walled cavity (let us call it an oven), viewed from outside,
looks red; at 6000 K, it looks white. No matter how the oven is
constructed, or of what material, as long as it is built so that almost
all light entering is absorbed by its walls, it will contain a good
approximation to black-body radiation. The spectrum, and therefore
color, of the light that comes out will be a function of the cavity
temperature alone. A graph of the amount of energy inside the oven per
unit volume and per unit frequency interval plotted versus frequency, is
called the
black-body curve. Different curves are obtained by varying the temperature.
The temperature of a
Pāhoehoe
lava flow can be estimated by observing its color. The result agrees
well with other measurements of temperatures of lava flows at about
1,000 to 1,200 °C (1,830 to 2,190 °F).
Two bodies that are at the same temperature stay in mutual thermal equilibrium, so a body at temperature
T surrounded by a cloud of light at temperature
T on average will emit as much light into the cloud as it absorbs, following Prevost's exchange principle, which refers to
radiative equilibrium. The principle of
detailed balance says that in thermodynamic equilibrium every elementary process works equally in its forward and backward sense.
[21][22]
Prevost also showed that the emission from a body is logically
determined solely by its own internal state. The causal effect of
thermodynamic absorption on thermodynamic (spontaneous) emission is not
direct, but is only indirect as it affects the internal state of the
body. This means that at thermodynamic equilibrium the amount of every
wavelength in every direction of thermal radiation emitted by a body at
temperature
T, black or not, is equal to the corresponding amount that the body absorbs because it is surrounded by light at temperature
T.
[23]
When the body is black, the absorption is obvious: the amount of
light absorbed is all the light that hits the surface. For a black body
much bigger than the wavelength, the light energy absorbed at any
wavelength
λ per unit time is strictly proportional to the
black-body curve. This means that the black-body curve is the amount of
light energy emitted by a black body, which justifies the name. This is
the condition for the applicability of
Kirchhoff's law of thermal radiation: the black-body curve is characteristic of thermal light, which depends only on the
temperature
of the walls of the cavity, provided that the walls of the cavity are
completely opaque and are not very reflective, and that the cavity is in
thermodynamic equilibrium.
[24]
When the black body is small, so that its size is comparable to the
wavelength of light, the absorption is modified, because a small object
is not an efficient absorber of light of long wavelength, but the
principle of strict equality of emission and absorption is always upheld
in a condition of thermodynamic equilibrium.
In the laboratory, black-body radiation is approximated by the radiation from a small hole in a large cavity, a
hohlraum,
in an entirely opaque body that is only partly reflective, that is
maintained at a constant temperature. (This technique leads to the
alternative term
cavity radiation.) Any light entering the hole
would have to reflect off the walls of the cavity multiple times before
it escaped, in which process it is nearly certain to be absorbed.
Absorption occurs regardless of the
wavelength
of the radiation entering (as long as it is small compared to the
hole). The hole, then, is a close approximation of a theoretical black
body and, if the cavity is heated, the
spectrum
of the hole's radiation (i.e., the amount of light emitted from the
hole at each wavelength) will be continuous, and will depend only on the
temperature and the fact that the walls are opaque and at least partly
absorptive, but not on the particular material of which they are built
nor on the material in the cavity (compare with
emission spectrum).
Calculating the black-body curve was a major challenge in
theoretical physics during the late nineteenth century. The problem was solved in 1901 by
Max Planck in the formalism now known as
Planck's law of black-body radiation.
[25] By making changes to
Wien's radiation law (not to be confused with
Wien's displacement law) consistent with
thermodynamics and
electromagnetism,
he found a mathematical expression fitting the experimental data
satisfactorily. Planck had to assume that the energy of the oscillators
in the cavity was quantized, i.e., it existed in integer multiples of
some quantity.
Einstein built on this idea and proposed the quantization of electromagnetic radiation itself in 1905 to explain the
photoelectric effect. These theoretical advances eventually resulted in the superseding of classical electromagnetism by
quantum electrodynamics. These quanta were called
photons and the black-body cavity was thought of as containing a
gas of photons. In addition, it led to the development of quantum probability distributions, called
Fermi–Dirac statistics and
Bose–Einstein statistics, each applicable to a different class of particles,
fermions and
bosons.
The wavelength at which the radiation is strongest is given by Wien's
displacement law, and the overall power emitted per unit area is given
by the
Stefan–Boltzmann law.
So, as temperature increases, the glow color changes from red to yellow
to white to blue. Even as the peak wavelength moves into the
ultra-violet, enough radiation continues to be emitted in the blue
wavelengths that the body will continue to appear blue. It will never
become invisible—indeed, the radiation of visible light increases
monotonically with temperature.
[26]
The Stefan-Boltzmann law also says that the total radiant heat energy
emitted from a surface is proportional to the fourth power of its
absolute temperature. The law was formulated by Josef Stefan in 1879 and
later derived by Ludwig Boltzmann. The formula E = σT
4 is
given, where E is the radiant heat emitted from a unit of area in one
second, T is the temperature in Kelvin, and sigma (σ) is the
Stefan-Boltzmann constant, which is equal to 5.670367 x 10
-8 watts per meter
2 per K
4.
[27]
The
radiance or observed intensity is not a function of direction. Therefore, a black body is a perfect
Lambertian radiator.
Real objects never behave as full-ideal black bodies, and instead the
emitted radiation at a given frequency is a fraction of what the ideal
emission would be. The
emissivity
of a material specifies how well a real body radiates energy as
compared with a black body. This emissivity depends on factors such as
temperature, emission angle, and wavelength. However, it is typical in
engineering to assume that a surface's spectral emissivity and
absorptivity do not depend on wavelength, so that the emissivity is a
constant. This is known as the
gray body assumption.
With non-black surfaces, the deviations from ideal black-body
behavior are determined by both the surface structure, such as roughness
or granularity, and the chemical composition. On a "per wavelength"
basis, real objects in states of
local thermodynamic equilibrium still follow
Kirchhoff's Law:
emissivity equals absorptivity, so that an object that does not absorb
all incident light will also emit less radiation than an ideal black
body; the incomplete absorption can be due to some of the incident light
being transmitted through the body or to some of it being reflected at
the surface of the body.
In
astronomy, objects such as
stars
are frequently regarded as black bodies, though this is often a poor
approximation. An almost perfect black-body spectrum is exhibited by the
cosmic microwave background radiation.
Hawking radiation is the hypothetical black-body radiation emitted by
black holes,
at a temperature that depends on the mass, charge, and spin of the
hole. If this prediction is correct, black holes will very gradually
shrink and evaporate over time as they lose mass by the emission of
photons and other particles.
A black body radiates energy at all frequencies, but its intensity
rapidly tends to zero at high frequencies (short wavelengths). For
example, a black body at room temperature (300 K) with one square meter
of surface area will emit a photon in the visible range (390–750 nm) at
an average rate of one photon every 41 seconds, meaning that for most
practical purposes, such a black body does not emit in the visible
range.
[30]
Equations
Planck's law of black-body radiation
Planck's law states that
[31]

where
- Bν(T) is the spectral radiance (the power per unit solid angle and per unit of area normal to the propagation) density of frequency ν radiation per unit frequency at thermal equilibrium at temperature T.
- h is the Planck constant;
- c is the speed of light in a vacuum;
- k is the Boltzmann constant;
- ν is the frequency of the electromagnetic radiation;
- T is the absolute temperature of the body.
For a black body surface the spectral radiance density (defined per
unit of area normal to the propagation) is independent of the angle

of emission with respect to the normal. However, this means that, following
Lambert's cosine law,

is the radiance density per unit area of emitting surface as the
surface area involved in generating the radiance is increased by a
factor

with respect to an area normal to the propagation direction. At oblique
angles, the solid angle spans involved do get smaller, resulting in
lower aggregate intensities.
Wien's displacement law
Wien's displacement law
shows how the spectrum of black-body radiation at any temperature is
related to the spectrum at any other temperature. If we know the shape
of the spectrum at one temperature, we can calculate the shape at any
other temperature. Spectral intensity can be expressed as a function of
wavelength or of frequency.
A consequence of Wien's displacement law is that the wavelength at which the intensity
per unit wavelength of the radiation produced by a black body is at a maximum,

, is a function only of the temperature:

where the constant
b, known as Wien's displacement constant, is equal to
2.8977729(17)×10−3 K m.
[32]
Planck's law was also stated above as a function of frequency. The intensity maximum for this is given by
 .[33]
.[33]
Stefan–Boltzmann law
By integrating

over the frequency the integrated radiance

is

by using

with

and with

being the
Stefan–Boltzmann constant. The radiance

is then

per unit of emitting surface.
On a side note, at a distance d, the intensity

per area

of radiating surface is the useful expression

when the receiving surface is perpendicular to the radiation.
By subsequently integrating over the solid angle

(where

) the
Stefan–Boltzmann law is calculated, stating that the power
j* emitted per unit area of the surface of a black body is directly proportional to the fourth power of its absolute temperature:

by using

Human-body emission
 |
 |
| Much of a person's energy is radiated away in the form of infrared
light. Some materials are transparent in the infrared, but opaque to
visible light, as is the plastic bag in this infrared image (bottom).
Other materials are transparent to visible light, but opaque or
reflective in the infrared, noticeable by the darkness of the man's
glasses. |
The human body radiates energy as
infrared light. The net power radiated is the difference between the power emitted and the power absorbed:

Applying the Stefan–Boltzmann law,

where
A and
T are the body surface area and temperature,

is the
emissivity, and
T0 is the ambient temperature.
The total surface area of an adult is about 2 m
2, and the mid- and far-infrared
emissivity of skin and most clothing is near unity, as it is for most nonmetallic surfaces.
[34][35] Skin temperature is about 33 °C,
[36] but clothing reduces the surface temperature to about 28 °C when the ambient temperature is 20 °C.
[37] Hence, the net radiative heat loss is about

The total energy radiated in one day is about 8
MJ, or 2000 kcal (food
calories).
Basal metabolic rate for a 40-year-old male is about 35 kcal/(m
2·h),
[38] which is equivalent to 1700 kcal per day, assuming the same 2 m
2 area. However, the mean metabolic rate of sedentary adults is about 50% to 70% greater than their basal rate.
[39]
There are other important thermal-loss mechanisms, including
convection and
evaporation. Conduction is negligible – the
Nusselt number is much greater than unity. Evaporation by
perspiration
is only required if radiation and convection are insufficient to
maintain a steady-state temperature (but evaporation from the lungs
occurs regardless). Free-convection rates are comparable, albeit
somewhat lower, than radiative rates.
[40]
Thus, radiation accounts for about two-thirds of thermal energy loss in
cool, still air. Given the approximate nature of many of the
assumptions, this can only be taken as a crude estimate. Ambient air
motion, causing forced convection, or evaporation reduces the relative
importance of radiation as a thermal-loss mechanism.
Application of
Wien's law to human-body emission results in a peak wavelength of

For this reason, thermal imaging devices for human subjects are most sensitive in the 7–14 micrometer range.
Temperature relation between a planet and its star
The black-body law may be used to estimate the temperature of a planet orbiting the Sun.
Earth's longwave thermal
radiation intensity, from clouds, atmosphere and ground
The temperature of a planet depends on several factors:
The analysis only considers the Sun's heat for a planet in a Solar System.
The
Stefan–Boltzmann law gives the total
power (energy/second) the Sun is emitting:

The Earth only has an absorbing area equal to a two dimensional disk, rather than the surface of a sphere.

where
 is the Stefan–Boltzmann constant,
is the Stefan–Boltzmann constant, is the effective temperature of the Sun, and
is the effective temperature of the Sun, and is the radius of the Sun.
is the radius of the Sun.
The Sun emits that power equally in all directions. Because of this,
the planet is hit with only a tiny fraction of it. The power from the
Sun that strikes the planet (at the top of the atmosphere) is:

where
 is the radius of the planet and
is the radius of the planet and is the distance between the Sun and the planet.
is the distance between the Sun and the planet.
Because of its high temperature, the Sun emits to a large extent in
the ultraviolet and visible (UV-Vis) frequency range. In this frequency
range, the planet reflects a fraction

of this energy where

is the
albedo or reflectance of the planet in the UV-Vis range. In other words, the planet absorbs a fraction

of the Sun's light, and reflects the rest. The power absorbed by the planet and its atmosphere is then:

Even though the planet only absorbs as a circular area

, it emits equally in all directions as a sphere. If the planet were a perfect black body, it would emit according to the
Stefan–Boltzmann law

where

is the temperature of the planet. This temperature, calculated for the case of the planet acting as a black body by setting

, is known as the
effective temperature.
The actual temperature of the planet will likely be different,
depending on its surface and atmospheric properties. Ignoring the
atmosphere and greenhouse effect, the planet, since it is at a much
lower temperature than the Sun, emits mostly in the infrared (IR)
portion of the spectrum. In this frequency range, it emits

of the radiation that a black body would emit where

is the average emissivity in the IR range. The power emitted by the planet is then:

For a body in
radiative exchange equilibrium with its surroundings, the rate at which it emits radiant energy is equal to the rate at which it absorbs it:
[41][42]

Substituting the expressions for solar and planet power in equations
1–6 and simplifying yields the estimated temperature of the planet,
ignoring greenhouse effect,
TP:

In other words, given the assumptions made, the temperature of a
planet depends only on the surface temperature of the Sun, the radius of
the Sun, the distance between the planet and the Sun, the albedo and
the IR emissivity of the planet.
Notice that a gray ( flat spectrum ) ball where

comes to the same temperature as a black body no matter how dark or light gray .
Virtual temperature of Earth
Substituting the measured values for the Sun and Earth yields:
 [43]
[43] [43]
[43] [43]
[43] [44]
[44]
With the average emissivity

set to unity, the
effective temperature of the Earth is:

or −18.8 °C.
This is the temperature of the Earth if it radiated as a perfect
black body in the infrared, assuming an unchanging albedo and ignoring
greenhouse effects (which can raise the surface temperature of a body above what it would be if it were a perfect black body in all spectrums
[45]).
The Earth in fact radiates not quite as a perfect black body in the
infrared which will raise the estimated temperature a few degrees above
the effective temperature. If we wish to estimate what the temperature
of the Earth would be if it had no atmosphere, then we could take the
albedo and emissivity of the Moon as a good estimate. The albedo and
emissivity of the Moon are about 0.1054
[46] and 0.95
[47] respectively, yielding an estimated temperature of about 1.36 °C.
Estimates of the Earth's average albedo vary in the range 0.3–0.4,
resulting in different estimated effective temperatures. Estimates are
often based on the
solar constant
(total insolation power density) rather than the temperature, size, and
distance of the Sun. For example, using 0.4 for albedo, and an
insolation of 1400 W m
−2, one obtains an effective temperature of about 245 K.
[48] Similarly using albedo 0.3 and solar constant of 1372 W m
−2, one obtains an effective temperature of 255 K.
[49][50][51]
Cosmology
The
cosmic microwave background radiation observed today is the most perfect black-body radiation ever observed in nature, with a temperature of about 2.7 K.
[52] It is a "snapshot" of the radiation at the time of
decoupling
between matter and radiation in the early universe. Prior to this time,
most matter in the universe was in the form of an ionized plasma in
thermal, though not full thermodynamic, equilibrium with radiation.
According to Kondepudi and Prigogine, at very high temperatures (above 10
10 K;
such temperatures existed in the very early universe), where the
thermal motion separates protons and neutrons in spite of the strong
nuclear forces, electron-positron pairs appear and disappear
spontaneously and are in thermal equilibrium with electromagnetic
radiation. These particles form a part of the black body spectrum, in
addition to the electromagnetic radiation.
[53]
Doppler effect for a moving black body
The
relativistic Doppler effect causes a shift in the frequency
f of light originating from a source that is moving in relation to the observer, so that the wave is observed to have frequency
f':

where
v is the velocity of the source in the observer's rest frame,
θ is the angle between the velocity vector and the observer-source direction measured in the reference frame of the source, and
c is the
speed of light.
[54] This can be simplified for the special cases of objects moving directly towards (
θ = π) or away (
θ = 0) from the observer, and for speeds much less than
c.
Through Planck's law the temperature spectrum of a black body is
proportionally related to the frequency of light and one may substitute
the temperature (
T) for the frequency in this equation.
For the case of a source moving directly towards or away from the observer, this reduces to

Here
v > 0 indicates a receding source, and
v < 0 indicates an approaching source.
This is an important effect in astronomy, where the velocities of stars and galaxies can reach significant fractions of
c. An example is found in the
cosmic microwave background radiation, which exhibits a dipole anisotropy from the Earth's motion relative to this black-body radiation field.
History
Balfour Stewart
In 1858,
Balfour Stewart
described his experiments on the thermal radiative emissive and
absorptive powers of polished plates of various substances, compared
with the powers of lamp-black surfaces, at the same temperature.
[23] Stewart chose lamp-black surfaces as his reference because of various previous experimental findings, especially those of
Pierre Prevost and of
John Leslie.
He wrote "Lamp-black, which absorbs all the rays that fall upon it, and
therefore possesses the greatest possible absorbing power, will possess
also the greatest possible radiating power." More an experimenter than a
logician, Stewart failed to point out that his statement presupposed an
abstract general principle, that there exist either ideally in theory
or really in nature bodies or surfaces that respectively have one and
the same unique universal greatest possible absorbing power, likewise
for radiating power, for every wavelength and equilibrium temperature.
Stewart measured radiated power with a thermo-pile and sensitive
galvanometer read with a microscope. He was concerned with selective
thermal radiation, which he investigated with plates of substances that
radiated and absorbed selectively for different qualities of radiation
rather than maximally for all qualities of radiation. He discussed the
experiments in terms of rays which could be reflected and refracted, and
which obeyed the Stokes-
Helmholtz reciprocity
principle (though he did not use an eponym for it). He did not in this
paper mention that the qualities of the rays might be described by their
wavelengths, nor did he use spectrally resolving apparatus such as
prisms or diffraction gratings. His work was quantitative within these
constraints. He made his measurements in a room temperature environment,
and quickly so as to catch his bodies in a condition near the thermal
equilibrium in which they had been prepared by heating to equilibrium
with boiling water. His measurements confirmed that substances that emit
and absorb selectively respect the principle of selective equality of
emission and absorption at thermal equilibrium.
Stewart offered a theoretical proof that this should be the case
separately for every selected quality of thermal radiation, but his
mathematics was not rigorously valid.
[55] He made no mention of thermodynamics in this paper, though he did refer to conservation of
vis viva.
He proposed that his measurements implied that radiation was both
absorbed and emitted by particles of matter throughout depths of the
media in which it propagated. He applied the Helmholtz reciprocity
principle to account for the material interface processes as distinct
from the processes in the interior material. He did not postulate
unrealizable perfectly black surfaces. He concluded that his experiments
showed that in a cavity in thermal equilibrium, the heat radiated from
any part of the interior bounding surface, no matter of what material it
might be composed, was the same as would have been emitted from a
surface of the same shape and position that would have been composed of
lamp-black. He did not state explicitly that the lamp-black-coated
bodies that he used as reference must have had a unique common spectral
emittance function that depended on temperature in a unique way.
Gustav Kirchhoff
In 1859, not knowing of Stewart's work,
Gustav Robert Kirchhoff
reported the coincidence of the wavelengths of spectrally resolved
lines of absorption and of emission of visible light. Importantly for
thermal physics, he also observed that bright lines or dark lines were
apparent depending on the temperature difference between emitter and
absorber.
[56]
Kirchhoff then went on to consider bodies that emit and absorb heat
radiation, in an opaque enclosure or cavity, in equilibrium at
temperature
T.
Here is used a notation different from Kirchhoff's. Here, the emitting power
E(T, i) denotes a dimensioned quantity, the total radiation emitted by a body labeled by index
i at temperature
T. The total absorption ratio
a(T, i) of that body is dimensionless, the ratio of absorbed to incident radiation in the cavity at temperature
T
. (In contrast with Balfour Stewart's, Kirchhoff's definition of his
absorption ratio did not refer in particular to a lamp-black surface as
the source of the incident radiation.) Thus the ratio
E(T, i) / a(T, i) of emitting power to absorption ratio is a dimensioned quantity, with the dimensions of emitting power, because
a(T, i) is dimensionless. Also here the wavelength-specific emitting power of the body at temperature
T is denoted by
E(λ, T, i) and the wavelength-specific absorption ratio by
a(λ, T, i) . Again, the ratio
E(λ, T, i) / a(λ, T, i) of emitting power to absorption ratio is a dimensioned quantity, with the dimensions of emitting power.
In a second report made in 1859, Kirchhoff announced a new general
principle or law for which he offered a theoretical and mathematical
proof, though he did not offer quantitative measurements of radiation
powers.
[57] His theoretical proof was and still is considered by some writers to be invalid.
[55][58]
His principle, however, has endured: it was that for heat rays of the
same wavelength, in equilibrium at a given temperature, the
wavelength-specific ratio of emitting power to absorption ratio has one
and the same common value for all bodies that emit and absorb at that
wavelength. In symbols, the law stated that the wavelength-specific
ratio
E(λ, T, i) / a(λ, T, i) has one and the same value for all bodies, that is for all values of index
i . In this report there was no mention of black bodies.
In 1860, still not knowing of Stewart's measurements for selected
qualities of radiation, Kirchhoff pointed out that it was long
established experimentally that for total heat radiation, of unselected
quality, emitted and absorbed by a body in equilibrium, the dimensioned
total radiation ratio
E(T, i) / a(T, i), has one and the same value common to all bodies, that is, for every value of the material index
i.
[59]
Again without measurements of radiative powers or other new
experimental data, Kirchhoff then offered a fresh theoretical proof of
his new principle of the universality of the value of the
wavelength-specific ratio
E(λ, T, i) / a(λ, T, i) at thermal equilibrium. His fresh theoretical proof was and still is considered by some writers to be invalid.
[55][58]
But more importantly, it relied on a new theoretical postulate of
"perfectly black bodies," which is the reason why one speaks of
Kirchhoff's law. Such black bodies showed complete absorption in their
infinitely thin most superficial surface. They correspond to Balfour
Stewart's reference bodies, with internal radiation, coated with
lamp-black. They were not the more realistic perfectly black bodies
later considered by Planck. Planck's black bodies radiated and absorbed
only by the material in their interiors; their interfaces with
contiguous media were only mathematical surfaces, capable neither of
absorption nor emission, but only of reflecting and transmitting with
refraction.
[60]
Kirchhoff's proof considered an arbitrary non-ideal body labeled
i as well as various perfect black bodies labeled
BB . It required that the bodies be kept in a cavity in thermal equilibrium at temperature
T . His proof intended to show that the ratio
E(λ, T, i) / a(λ, T, i) was independent of the nature
i of the non-ideal body, however partly transparent or partly reflective it was.
His proof first argued that for wavelength
λ and at temperature
T,
at thermal equilibrium, all perfectly black bodies of the same size and
shape have the one and the same common value of emissive power
E(λ, T, BB), with the dimensions of power. His proof noted that the dimensionless wavelength-specific absorption ratio
a(λ, T, BB)
of a perfectly black body is by definition exactly 1. Then for a
perfectly black body, the wavelength-specific ratio of emissive power to
absorption ratio
E(λ, T, BB) / a(λ, T, BB) is again just
E(λ, T, BB),
with the dimensions of power. Kirchhoff considered, successively,
thermal equilibrium with the arbitrary non-ideal body, and with a
perfectly black body of the same size and shape, in place in his cavity
in equilibrium at temperature
T . He
argued that the flows of heat radiation must be the same in each case.
Thus he argued that at thermal equilibrium the ratio
E(λ, T, i) / a(λ, T, i) was equal to
E(λ, T, BB), which may now be denoted
Bλ (λ, T), a continuous function, dependent only on
λ at fixed temperature
T, and an increasing function of
T at fixed wavelength
λ,
at low temperatures vanishing for visible but not for longer
wavelengths, with positive values for visible wavelengths at higher
temperatures, which does not depend on the nature
i
of the arbitrary non-ideal body. (Geometrical factors, taken into
detailed account by Kirchhoff, have been ignored in the foregoing.)
Thus
Kirchhoff's law of thermal radiation can be stated:
For any material at all, radiating and absorbing in thermodynamic equilibrium at any given temperature T, for every wavelength λ,
the ratio of emissive power to absorptive ratio has one universal
value, which is characteristic of a perfect black body, and is an
emissive power which we here represent by Bλ (λ, T) . (For our notation
Bλ (λ, T), Kirchhoff's original notation was simply
e.)
[59][61][62][63][64][65]
Kirchhoff announced that the determination of the function
Bλ (λ, T)
was a problem of the highest importance, though he recognized that
there would be experimental difficulties to be overcome. He supposed
that like other functions that do not depend on the properties of
individual bodies, it would be a simple function. Occasionally by
historians that function
Bλ (λ, T) has been called "Kirchhoff's (emission, universal) function,"
[66][67][68][69]
though its precise mathematical form would not be known for another
forty years, till it was discovered by Planck in 1900. The theoretical
proof for Kirchhoff's universality principle was worked on and debated
by various physicists over the same time, and later.
[58] Kirchhoff stated later in 1860 that his theoretical proof was better than Balfour Stewart's, and in some respects it was so.
[55]
Kirchhoff's 1860 paper did not mention the second law of
thermodynamics, and of course did not mention the concept of entropy
which had not at that time been established. In a more considered
account in a book in 1862, Kirchhoff mentioned the connection of his law
with
Carnot's principle, which is a form of the second law.
[70]
According to Helge Kragh, "Quantum theory owes its origin to the
study of thermal radiation, in particular to the "black-body" radiation
that Robert Kirchhoff had first defined in 1859–1860."
[71]