From Wikipedia, the free encyclopedia
Natural color x-ray
photogram of a wine scene
An X-ray, or, much less commonly, X-radiation, is a penetrating form of high-energy electromagnetic radiation. Most X-rays have a wavelength ranging from 10 picometers to 10 nanometers, corresponding to frequencies in the range 30 petahertz to 30 exahertz (30×1015 Hz to 30×1018 Hz) and energies in the range 145 eV to 124 keV. X-ray wavelengths are shorter than those of UV rays and typically longer than those of gamma rays. In many languages, X-radiation is referred to as Röntgen radiation, after the German scientist Wilhelm Conrad Röntgen, who discovered it on November 8, 1895. He named it X-radiation to signify an unknown type of radiation. Spellings of X-ray(s) in English include the variants x-ray(s), xray(s), and X ray(s).
The most familiar use of x-rays is checking for fractures (broken
bones), but x-rays are also used in other ways. For example, chest
x-rays can spot pneumonia. Mammograms use x-rays to look for breast
cancer.
History
Pre-Röntgen observations and research
Before their discovery in 1895, X-rays were just a type of unidentified radiation emanating from experimental discharge tubes. They were noticed by scientists investigating cathode rays produced by such tubes, which are energetic electron beams that were first observed in 1869. Many of the early Crookes tubes
(invented around 1875) undoubtedly radiated X-rays, because early
researchers noticed effects that were attributable to them, as detailed
below. Crookes tubes created free electrons by ionization of the residual air in the tube by a high DC voltage of anywhere between a few kilovolts and 100 kV. This voltage accelerated the electrons coming from the cathode to a high enough velocity that they created X-rays when they struck the anode or the glass wall of the tube.
The earliest experimenter thought to have (unknowingly) produced X-rays was William Morgan. In 1785, he presented a paper to the Royal Society of London
describing the effects of passing electrical currents through a
partially evacuated glass tube, producing a glow created by X-rays. This work was further explored by Humphry Davy and his assistant Michael Faraday.
When Stanford University physics professor Fernando Sanford
created his "electric photography", he also unknowingly generated and
detected X-rays. From 1886 to 1888, he had studied in the Hermann von Helmholtz
laboratory in Berlin, where he became familiar with the cathode rays
generated in vacuum tubes when a voltage was applied across separate
electrodes, as previously studied by Heinrich Hertz and Philipp Lenard. His letter of January 6, 1893 (describing his discovery as "electric photography") to The Physical Review was duly published and an article entitled Without Lens or Light, Photographs Taken With Plate and Object in Darkness appeared in the San Francisco Examiner.
Starting in 1888, Philipp Lenard conducted experiments to see
whether cathode rays could pass out of the Crookes tube into the air. He
built a Crookes tube with a "window" at the end made of thin aluminium,
facing the cathode so the cathode rays would strike it (later called a
"Lenard tube"). He found that something came through, that would expose
photographic plates and cause fluorescence. He measured the penetrating
power of these rays through various materials. It has been suggested
that at least some of these "Lenard rays" were actually X-rays.
In 1889, Ukrainian-born Ivan Puluj, a lecturer in experimental physics at the Prague Polytechnic who since 1877 had been constructing various designs of glass-filled tubes
to investigate their properties, published a paper on how sealed
photographic plates became dark when exposed to the emanations from the
tubes.
Helmholtz formulated mathematical equations for X-rays. He
postulated a dispersion theory before Röntgen made his discovery and
announcement. He based it on the electromagnetic theory of light. However, he did not work with actual X-rays.
In 1894, Nikola Tesla
noticed damaged film in his lab that seemed to be associated with
Crookes tube experiments and began investigating this invisible, radiant energy. After Röntgen identified the X-ray, Tesla began making X-ray images of his own using high voltages and tubes of his own design, as well as Crookes tubes.
Discovery by Röntgen
On November 8, 1895, German physics professor Wilhelm Röntgen stumbled on X-rays while experimenting with Lenard tubes and Crookes tubes
and began studying them. He wrote an initial report "On a new kind of
ray: A preliminary communication" and on December 28, 1895, submitted it
to Würzburg's Physical-Medical Society journal.
This was the first paper written on X-rays. Röntgen referred to the
radiation as "X", to indicate that it was an unknown type of radiation.
The name stuck, although (over Röntgen's great objections) many of his
colleagues suggested calling them Röntgen rays. They are still referred to as such in many languages, including German, Hungarian, Ukrainian, Danish, Polish, Bulgarian, Swedish, Finnish, Estonian, Slovenian, Turkish, Russian, Latvian, Lithuanian, Japanese, Dutch, Georgian, Hebrew, and Norwegian. Röntgen received the first Nobel Prize in Physics for his discovery.
There are conflicting accounts of his discovery because Röntgen
had his lab notes burned after his death, but this is a likely
reconstruction by his biographers:
Röntgen was investigating cathode rays from a Crookes tube which he had
wrapped in black cardboard so that the visible light from the tube
would not interfere, using a fluorescent screen painted with barium platinocyanide.
He noticed a faint green glow from the screen, about 1 meter (3.3 ft)
away. Röntgen realized some invisible rays coming from the tube were
passing through the cardboard to make the screen glow. He found they
could also pass through books and papers on his desk. Röntgen threw
himself into investigating these unknown rays systematically. Two months
after his initial discovery, he published his paper.
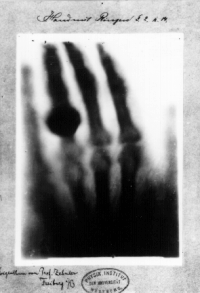
Röntgen discovered their medical use when he made a picture of his
wife's hand on a photographic plate formed due to X-rays. The photograph
of his wife's hand was the first photograph of a human body part using
X-rays. When she saw the picture, she said "I have seen my death."
The discovery of X-rays stimulated a veritable sensation.
Röntgen's biographer Otto Glasser estimated that, in 1896 alone, as many
as 49 essays and 1044 articles about the new rays were published.
This was probably a conservative estimate, if one considers that nearly
every paper around the world extensively reported about the new
discovery, with a magazine such as Science dedicating as many as 23 articles to it in that year alone.
Sensationalist reactions to the new discovery included publications
linking the new kind of rays to occult and paranormal theories, such as
telepathy.
Advances in radiology
Taking an X-ray image with early
Crookes tube apparatus, late 1800s. The Crookes tube is visible in center. The standing man is viewing his hand with a
fluoroscope screen. The seated man is taking a
radiograph of his hand by placing it on a
photographic plate. No precautions against radiation exposure are taken; its hazards were not known at the time.
Surgical removal of a bullet whose location was diagnosed with X-rays (see inset) in 1897
Röntgen immediately noticed X-rays could have medical applications.
Along with his 28 December Physical-Medical Society submission, he sent a
letter to physicians he knew around Europe (January 1, 1896). News (and the creation of "shadowgrams") spread rapidly with Scottish electrical engineer Alan Archibald Campbell-Swinton
being the first after Röntgen to create an X-ray (of a hand). Through
February, there were 46 experimenters taking up the technique in North
America alone.
The first use of X-rays under clinical conditions was by John Hall-Edwards in Birmingham, England
on 11 January 1896, when he radiographed a needle stuck in the hand of
an associate. On February 14, 1896, Hall-Edwards was also the first to
use X-rays in a surgical operation.
In early 1896, several weeks after Röntgen's discovery, Ivan Romanovich Tarkhanov irradiated frogs and insects with X-rays, concluding that the rays "not only photograph, but also affect the living function". At around the same time, the zoological illustrator James Green began to use X-rays to examine fragile specimens. George Albert Boulenger first mentioned this work in a paper he delivered before the Zoological Society of London in May 1896. The book Sciagraphs of British Batrachians and Reptiles
(sciagraph is an obsolete name for an X-ray photograph), by Green and
James H. Gardiner, with a foreword by Boulenger, was published in 1897.
The first medical X-ray made in the United States was obtained
using a discharge tube of Pului's design. In January 1896, on reading of
Röntgen's discovery, Frank Austin of Dartmouth College
tested all of the discharge tubes in the physics laboratory and found
that only the Pului tube produced X-rays. This was a result of Pului's
inclusion of an oblique "target" of mica, used for holding samples of fluorescent
material, within the tube. On 3 February 1896, Gilman Frost, professor
of medicine at the college, and his brother Edwin Frost, professor of
physics, exposed the wrist of Eddie McCarthy, whom Gilman had treated
some weeks earlier for a fracture, to the X-rays and collected the
resulting image of the broken bone on gelatin photographic plates obtained from Howard Langill, a local photographer also interested in Röntgen's work.
1896 plaque published in
"Nouvelle Iconographie de la Salpetrière", a medical journal. In the left a hand deformity, in the right same hand seen using
radiography. The authors named the technique Röntgen photography.
Many experimenters, including Röntgen himself in his original
experiments, came up with methods to view X-ray images "live" using some
form of luminescent screen. Röntgen used a screen coated with barium platinocyanide.
On February 5, 1896, live imaging devices were developed by both
Italian scientist Enrico Salvioni (his "cryptoscope") and Professor
McGie of Princeton University (his "Skiascope"), both using barium platinocyanide. American inventor Thomas Edison
started research soon after Röntgen's discovery and investigated
materials' ability to fluoresce when exposed to X-rays, finding that calcium tungstate
was the most effective substance. In May 1896, he developed the first
mass-produced live imaging device, his "Vitascope", later called the fluoroscope, which became the standard for medical X-ray examinations. Edison dropped X-ray research around 1903, before the death of Clarence Madison Dally, one of his glassblowers. Dally had a habit of testing X-ray tubes on his own hands, developing a cancer in them so tenacious that both arms were amputated in a futile attempt to save his life; in 1904, he became the first known death attributed to X-ray exposure. During the time the fluoroscope was being developed, Serbian American physicist Mihajlo Pupin,
using a calcium tungstate screen developed by Edison, found that using a
fluorescent screen decreased the exposure time it took to create an
X-ray for medical imaging from an hour to a few minutes.
In 1901, U.S. President William McKinley was shot twice in an assassination attempt. While one bullet only grazed his sternum, another had lodged somewhere deep inside his abdomen and could not be found. A worried McKinley aide sent word to inventor Thomas Edison to rush an X-ray machine to Buffalo to find the stray bullet. It arrived but was not used. While the shooting itself had not been lethal, gangrene had developed along the path of the bullet, and McKinley died of septic shock due to bacterial infection six days later.
Hazards discovered
With
the widespread experimentation with X‑rays after their discovery in
1895 by scientists, physicians, and inventors came many stories of
burns, hair loss, and worse in technical journals of the time. In
February 1896, Professor John Daniel and Dr. William Lofland Dudley of Vanderbilt University
reported hair loss after Dr. Dudley was X-rayed. A child who had been
shot in the head was brought to the Vanderbilt laboratory in 1896.
Before trying to find the bullet, an experiment was attempted, for which
Dudley "with his characteristic devotion to science" volunteered. Daniel reported that 21 days after taking a picture of Dudley's skull
(with an exposure time of one hour), he noticed a bald spot 5
centimeters (2 in) in diameter on the part of his head nearest the X-ray
tube: "A plate holder with the plates towards the side of the skull was
fastened and a coin
placed between the skull and the head. The tube was fastened at the
other side at a distance of one-half inch [1.3 cm] from the hair."
In August 1896, Dr. HD. Hawks, a graduate of Columbia College,
suffered severe hand and chest burns from an X-ray demonstration. It was
reported in Electrical Review and led to many other reports of problems associated with X-rays being sent in to the publication. Many experimenters including Elihu Thomson at Edison's lab, William J. Morton, and Nikola Tesla
also reported burns. Elihu Thomson deliberately exposed a finger to an
X-ray tube over a period of time and suffered pain, swelling, and
blistering. Other effects were sometimes blamed for the damage including ultraviolet rays and (according to Tesla) ozone. Many physicians claimed there were no effects from X-ray exposure at all. On August 3, 1905, in San Francisco, California, Elizabeth Fleischman, an American X-ray pioneer, died from complications as a result of her work with X-rays.
Hall-Edwards developed a cancer (then called X-ray dermatitis)
sufficiently advanced by 1904 to cause him to write papers and give
public addresses on the dangers of X-rays. He lost his personal battle
and his left arm had to be amputated at the elbow in 1908, and four fingers on his right arm soon thereafter, leaving only a thumb. He died of cancer in 1926. His left hand is kept at Birmingham University.
20th century and beyond
A patient being examined with a thoracic
fluoroscope in 1940, which displayed continuous moving images. This image was used to argue that
radiation exposure during the X-ray procedure would be negligible.
The many applications of X-rays immediately generated enormous
interest. Workshops began making specialized versions of Crookes tubes
for generating X-rays and these first-generation cold cathode or Crookes X-ray tubes were used until about 1920.
A typical early 20th century medical X-ray system consisted of a Ruhmkorff coil connected to a cold cathode Crookes X-ray tube. A spark gap was typically connected to the high voltage side in parallel to the tube and used for diagnostic purposes.
The spark gap allowed detecting the polarity of the sparks, measuring
voltage by the length of the sparks thus determining the "hardness" of
the vacuum of the tube, and it provided a load in the event the X-ray
tube was disconnected. To detect the hardness of the tube, the spark gap
was initially opened to the widest setting. While the coil was
operating, the operator reduced the gap until sparks began to appear. A
tube in which the spark gap began to spark at around 6.4 centimeters
(2.5 in) was considered soft (low vacuum) and suitable for thin body
parts such as hands and arms. A 13-centimeter (5 in) spark indicated the
tube was suitable for shoulders and knees. An 18-to-23-centimeter (7 to
9 in) spark would indicate a higher vacuum suitable for imaging the
abdomen of larger individuals. Since the spark gap was connected in
parallel to the tube, the spark gap had to be opened until the sparking
ceased in order to operate the tube for imaging. Exposure time for
photographic plates was around half a minute for a hand to a couple of
minutes for a thorax. The plates may have a small addition of
fluorescent salt to reduce exposure times.
Crookes tubes were unreliable. They had to contain a small
quantity of gas (invariably air) as a current will not flow in such a
tube if they are fully evacuated. However, as time passed, the X-rays
caused the glass to absorb the gas, causing the tube to generate
"harder" X-rays until it soon stopped operating. Larger and more
frequently used tubes were provided with devices for restoring the air,
known as "softeners". These often took the form of a small side tube
that contained a small piece of mica, a mineral
that traps relatively large quantities of air within its structure. A
small electrical heater heated the mica, causing it to release a small
amount of air, thus restoring the tube's efficiency. However, the mica
had a limited life, and the restoration process was difficult to
control.
In 1904, John Ambrose Fleming invented the thermionic diode, the first kind of vacuum tube. This used a hot cathode that caused an electric current to flow in a vacuum.
This idea was quickly applied to X-ray tubes, and hence heated-cathode
X-ray tubes, called "Coolidge tubes", completely replaced the
troublesome cold cathode tubes by about 1920.
In about 1906, the physicist Charles Barkla discovered that X-rays could be scattered by gases, and that each element had a characteristic X-ray spectrum. He won the 1917 Nobel Prize in Physics for this discovery.
In 1912, Max von Laue, Paul Knipping, and Walter Friedrich first observed the diffraction of X-rays by crystals. This discovery, along with the early work of Paul Peter Ewald, William Henry Bragg, and William Lawrence Bragg, gave birth to the field of X-ray crystallography.
In 1913, Henry Moseley performed crystallography experiments with X-rays emanating from various metals and formulated Moseley's law which relates the frequency of the X-rays to the atomic number of the metal.
The Coolidge X-ray tube was invented the same year by William D. Coolidge.
It made possible the continuous emissions of X-rays. Modern X-ray tubes
are based on this design, often employing the use of rotating targets
which allow for significantly higher heat dissipation than static
targets, further allowing higher quantity X-ray output for use in high
powered applications such as rotational CT scanners.
Chandra's
image of the galaxy cluster Abell 2125 reveals a complex of several
massive multimillion-degree-Celsius gas clouds in the process of
merging.
The use of X-rays for medical purposes (which developed into the field of radiation therapy) was pioneered by Major John Hall-Edwards in Birmingham, England. Then in 1908, he had to have his left arm amputated because of the spread of X-ray dermatitis on his arm.
Medical science also used the motion picture to study human
physiology. In 1913, a motion picture was made in Detroit showing a
hard-boiled egg inside a human stomach. This early X-ray movie was
recorded at a rate of one still image every four seconds. Dr Lewis Gregory Cole of New York was a pioneer of the technique, which he called "serial radiography". In 1918, X-rays were used in association with motion picture cameras to capture the human skeleton in motion.
In 1920, it was used to record the movements of tongue and teeth in the
study of languages by the Institute of Phonetics in England.
In 1914, Marie Curie developed radiological cars to support soldiers injured in World War I.
The cars would allow for rapid X-ray imaging of wounded soldiers so
battlefield surgeons could quickly and more accurately operate.
From the early 1920s through to the 1950s, X-ray machines were developed to assist in the fitting of shoes and were sold to commercial shoe stores. Concerns regarding the impact of frequent or poorly controlled use were expressed in the 1950s, leading to the practice's eventual end that decade.
The X-ray microscope was developed during the 1950s.
The Chandra X-ray Observatory,
launched on July 23, 1999, has been allowing the exploration of the
very violent processes in the universe which produce X-rays. Unlike
visible light, which gives a relatively stable view of the universe, the
X-ray universe is unstable. It features stars being torn apart by black holes, galactic collisions, and novae, and neutron stars that build up layers of plasma that then explode into space.
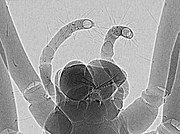
Phase-contrast X-ray image of spider
An X-ray laser device was proposed as part of the Reagan Administration's Strategic Defense Initiative in the 1980s, but the only test of the device (a sort of laser "blaster" or death ray,
powered by a thermonuclear explosion) gave inconclusive results. For
technical and political reasons, the overall project (including the
X-ray laser) was defunded (though was later revived by the second Bush Administration as National Missile Defense using different technologies).
Phase-contrast X-ray imaging
refers to a variety of techniques that use phase information of an
X-ray beam to form the image. Due to its good sensitivity to density
differences, it is especially useful for imaging soft tissues. It has
become an important method for visualizing cellular and histological
structures in a wide range of biological and medical studies. There are
several technologies being used for X-ray phase-contrast imaging, all
utilizing different principles to convert phase variations in the X-rays
emerging from an object into intensity variations. These include propagation-based phase contrast, Talbot interferometry, refraction-enhanced imaging, and X-ray interferometry.
These methods provide higher contrast compared to normal
absorption-based X-ray imaging, making it possible to distinguish from
each other details that have almost similar density. A disadvantage is
that these methods require more sophisticated equipment, such as synchrotron or microfocus X-ray sources, X-ray optics, and high resolution X-ray detectors.
Energy ranges
Soft and hard X-rays
X-rays with high photon energies above 5–10 keV (below 0.2–0.1 nm wavelength) are called hard X-rays, while those with lower energy (and longer wavelength) are called soft X-rays. The intermediate range with photon energies of several keV is often referred to as tender X-rays. Due to their penetrating ability, hard X-rays are widely used to image the inside of objects, e.g., in medical radiography and airport security. The term X-ray is metonymically used to refer to a radiographic
image produced using this method, in addition to the method itself.
Since the wavelengths of hard X-rays are similar to the size of atoms,
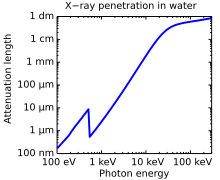
they are also useful for determining crystal structures by X-ray crystallography. By contrast, soft X-rays are easily absorbed in air; the attenuation length of 600 eV (~2 nm) X-rays in water is less than 1 micrometer.
Gamma rays
There is no consensus for a definition distinguishing between X-rays and gamma rays. One common practice is to distinguish between the two types of radiation based on their source: X-rays are emitted by electrons, while gamma rays are emitted by the atomic nucleus. This definition has several problems: other processes can also generate these high-energy photons,
or sometimes the method of generation is not known. One common
alternative is to distinguish X- and gamma radiation on the basis of
wavelength (or, equivalently, frequency or photon energy), with
radiation shorter than some arbitrary wavelength, such as 10−11 m (0.1 Å), defined as gamma radiation.
This criterion assigns a photon to an unambiguous category, but is only
possible if wavelength is known. (Some measurement techniques do not
distinguish between detected wavelengths.) However, these two
definitions often coincide since the electromagnetic radiation emitted
by X-ray tubes generally has a longer wavelength and lower photon energy than the radiation emitted by radioactive nuclei.
Occasionally, one term or the other is used in specific contexts due to
historical precedent, based on measurement (detection) technique, or
based on their intended use rather than their wavelength or source.
Thus, gamma-rays generated for medical and industrial uses, for example radiotherapy, in the ranges of 6–20 MeV, can in this context also be referred to as X-rays.
Properties
Ionizing radiation hazard symbol
X-ray photons carry enough energy to ionize atoms and disrupt molecular bonds. This makes it a type of ionizing radiation, and therefore harmful to living tissue. A very high radiation dose over a short period of time causes radiation sickness, while lower doses can give an increased risk of radiation-induced cancer.
In medical imaging, this increased cancer risk is generally greatly
outweighed by the benefits of the examination. The ionizing capability
of X-rays can be utilized in cancer treatment to kill malignant cells using radiation therapy. It is also used for material characterization using X-ray spectroscopy.
Hard X-rays can traverse relatively thick objects without being much absorbed or scattered. For this reason, X-rays are widely used to image the inside of visually opaque objects. The most often seen applications are in medical radiography and airport security scanners, but similar techniques are also important in industry (e.g., industrial radiography and industrial CT scanning) and research (e.g., small animal CT). The penetration depth varies with several orders of magnitude over the X-ray spectrum. This allows the photon energy to be adjusted for the application so as to give sufficient transmission through the object and at the same time provide good contrast in the image.
X-rays have much shorter wavelengths than visible light, which
makes it possible to probe structures much smaller than can be seen
using a normal microscope. This property is used in X-ray microscopy to acquire high-resolution images, and also in X-ray crystallography to determine the positions of atoms in crystals.
Interaction with matter
Attenuation length of X-rays in water showing the oxygen
absorption edge at 540 eV, the energy
−3 dependence of
photoabsorption, as well as a leveling off at higher photon energies due to
Compton scattering.
The attenuation length is about four orders of magnitude longer for
hard X-rays (right half) compared to soft X-rays (left half).
X-rays interact with matter in three main ways, through photoabsorption, Compton scattering, and Rayleigh scattering.
The strength of these interactions depends on the energy of the X-rays
and the elemental composition of the material, but not much on chemical
properties, since the X-ray photon energy is much higher than chemical
binding energies. Photoabsorption or photoelectric absorption is the
dominant interaction mechanism in the soft X-ray regime and for the
lower hard X-ray energies. At higher energies, Compton scattering
dominates.
Photoelectric absorption
The probability of a photoelectric absorption per unit mass is approximately proportional to Z3/E3, where Z is the atomic number and E is the energy of the incident photon.
This rule is not valid close to inner shell electron binding energies
where there are abrupt changes in interaction probability, so called absorption edges. However, the general trend of high absorption coefficients and thus short penetration depths
for low photon energies and high atomic numbers is very strong. For
soft tissue, photoabsorption dominates up to about 26 keV photon energy
where Compton scattering takes over. For higher atomic number
substances, this limit is higher. The high amount of calcium (Z = 20) in bones, together with their high density, is what makes them show up so clearly on medical radiographs.
A photoabsorbed photon transfers all its energy to the electron
with which it interacts, thus ionizing the atom to which the electron
was bound and producing a photoelectron that is likely to ionize more
atoms in its path. An outer electron will fill the vacant electron
position and produce either a characteristic X-ray or an Auger electron. These effects can be used for elemental detection through X-ray spectroscopy or Auger electron spectroscopy.
Compton scattering
Compton scattering is the predominant interaction between X-rays and soft tissue in medical imaging. Compton scattering is an inelastic scattering
of the X-ray photon by an outer shell electron. Part of the energy of
the photon is transferred to the scattering electron, thereby ionizing
the atom and increasing the wavelength of the X-ray. The scattered
photon can go in any direction, but a direction similar to the original
direction is more likely, especially for high-energy X-rays. The
probability for different scattering angles is described by the Klein–Nishina formula. The transferred energy can be directly obtained from the scattering angle from the conservation of energy and momentum.
Rayleigh scattering
Rayleigh scattering is the dominant elastic scattering mechanism in the X-ray regime. Inelastic forward scattering gives rise to the refractive index, which for X-rays is only slightly below 1.
Production
Whenever charged particles (electrons or ions) of sufficient energy hit a material, X-rays are produced.
Production by electrons
Characteristic X-ray emission lines for some common anode materials.
Anode
material |
Atomic
number |
Photon energy [keV] |
Wavelength [nm]
|
| Kα1 |
Kβ1 |
Kα1 |
Kβ1
|
| W
|
74 |
59.3 |
67.2 |
0.0209 |
0.0184
|
| Mo
|
42 |
17.5 |
19.6 |
0.0709 |
0.0632
|
| Cu
|
29 |
8.05 |
8.91 |
0.154 |
0.139
|
| Ag
|
47 |
22.2 |
24.9 |
0.0559 |
0.0497
|
| Ga
|
31 |
9.25 |
10.26 |
0.134 |
0.121
|
| In
|
49 |
24.2 |
27.3 |
0.0512 |
0.455
|
X-rays can be generated by an X-ray tube, a vacuum tube that uses a high voltage to accelerate the electrons released by a hot cathode to a high velocity. The high velocity electrons collide with a metal target, the anode, creating the X-rays. In medical X-ray tubes the target is usually tungsten or a more crack-resistant alloy of rhenium (5%) and tungsten (95%), but sometimes molybdenum for more specialized applications, such as when softer X-rays are needed as in mammography. In crystallography, a copper target is most common, with cobalt often being used when fluorescence from iron content in the sample might otherwise present a problem.
The maximum energy of the produced X-ray photon
is limited by the energy of the incident electron, which is equal to
the voltage on the tube times the electron charge, so an 80 kV tube
cannot create X-rays with an energy greater than 80 keV. When the
electrons hit the target, X-rays are created by two different atomic
processes:
- Characteristic X-ray emission (X-ray electroluminescence): If the electron has enough energy, it can knock an orbital electron out of the inner electron shell
of the target atom. After that, electrons from higher energy levels
fill the vacancies, and X-ray photons are emitted. This process produces
an emission spectrum
of X-rays at a few discrete frequencies, sometimes referred to as
spectral lines. Usually, these are transitions from the upper shells to
the K shell (called K lines), to the L shell (called L lines) and so on.
If the transition is from 2p to 1s, it is called Kα, while if it is
from 3p to 1s it is Kβ. The frequencies of these lines depend on the
material of the target and are therefore called characteristic lines.
The Kα line usually has greater intensity than the Kβ one and is more
desirable in diffraction experiments. Thus the Kβ line is filtered out
by a filter. The filter is usually made of a metal having one proton
less than the anode material (e.g., Ni filter for Cu anode or Nb filter
for Mo anode).
- Bremsstrahlung: This is radiation given off by the electrons as they are scattered by the strong electric field near the high-Z (proton number) nuclei. These X-rays have a continuous spectrum. The frequency of bremsstrahlung is limited by the energy of incident electrons.
So, the resulting output of a tube consists of a continuous
bremsstrahlung spectrum falling off to zero at the tube voltage, plus
several spikes at the characteristic lines. The voltages used in
diagnostic X-ray tubes range from roughly 20 kV to 150 kV and thus the
highest energies of the X-ray photons range from roughly 20 keV to
150 keV.
Both of these X-ray production processes are inefficient, with
only about one percent of the electrical energy used by the tube
converted into X-rays, and thus most of the electric power
consumed by the tube is released as waste heat. When producing a usable
flux of X-rays, the X-ray tube must be designed to dissipate the excess
heat.
A specialized source of X-rays which is becoming widely used in research is synchrotron radiation, which is generated by particle accelerators.
Its unique features are X-ray outputs many orders of magnitude greater
than those of X-ray tubes, wide X-ray spectra, excellent collimation, and linear polarization.
Short nanosecond bursts of X-rays peaking at 15 keV in energy may
be reliably produced by peeling pressure-sensitive adhesive tape from
its backing in a moderate vacuum. This is likely to be the result of
recombination of electrical charges produced by triboelectric charging. The intensity of X-ray triboluminescence is sufficient for it to be used as a source for X-ray imaging.
Production by fast positive ions
X-rays can also be produced by fast protons or other positive ions. The proton-induced X-ray emission or particle-induced X-ray emission is widely used as an analytical procedure. For high energies, the production cross section is proportional to Z12Z2−4, where Z1 refers to the atomic number of the ion, Z2 refers to that of the target atom. An overview of these cross sections is given in the same reference.
Production in lightning and laboratory discharges
X-rays are also produced in lightning accompanying terrestrial gamma-ray flashes.
The underlying mechanism is the acceleration of electrons in lightning
related electric fields and the subsequent production of photons through
Bremsstrahlung. This produces photons with energies of some few keV and several tens of MeV.
In laboratory discharges with a gap size of approximately 1 meter
length and a peak voltage of 1 MV, X-rays with a characteristic energy
of 160 keV are observed. A possible explanation is the encounter of two streamers and the production of high-energy run-away electrons;
however, microscopic simulations have shown that the duration of
electric field enhancement between two streamers is too short to produce
a significant number of run-away electrons.
Recently, it has been proposed that air perturbations in the vicinity
of streamers can facilitate the production of run-away electrons and
hence of X-rays from discharges.
Detectors
X-ray detectors vary in shape and function depending on their purpose. Imaging detectors such as those used for radiography were originally based on photographic plates and later photographic film, but are now mostly replaced by various digital detector types such as image plates and flat panel detectors. For radiation protection direct exposure hazard is often evaluated using ionization chambers, while dosimeters are used to measure the radiation dose a person has been exposed to. X-ray spectra can be measured either by energy dispersive or wavelength dispersive spectrometers. For X-ray diffraction applications, such as X-ray crystallography, hybrid photon counting detectors are widely used.
Medical uses
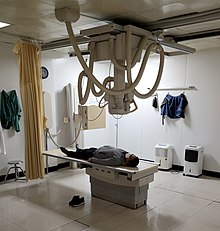
Patient undergoing an x-ray exam in a hospital radiology room.
Since Röntgen's discovery that X-rays can identify bone structures, X-rays have been used for medical imaging. The first medical use was less than a month after his paper on the subject. Up to 2010, five billion medical imaging examinations had been conducted worldwide. Radiation exposure from medical imaging in 2006 made up about 50% of total ionizing radiation exposure in the United States.
Projectional radiographs
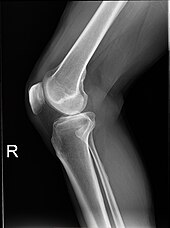
Plain radiograph of the right knee
Projectional radiography is the practice of producing two-dimensional images using X-ray radiation. Bones contain a high concentration of calcium, which, due to its relatively high atomic number,
absorbs X-rays efficiently. This reduces the amount of X-rays reaching
the detector in the shadow of the bones, making them clearly visible on
the radiograph. The lungs and trapped gas also show up clearly because
of lower absorption compared to tissue, while differences between tissue
types are harder to see.
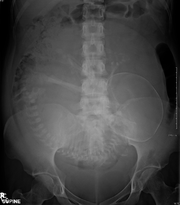
Projectional radiographs are useful in the detection of pathology of the skeletal system as well as for detecting some disease processes in soft tissue. Some notable examples are the very common chest X-ray, which can be used to identify lung diseases such as pneumonia, lung cancer, or pulmonary edema, and the abdominal x-ray, which can detect bowel (or intestinal) obstruction, free air (from visceral perforations), and free fluid (in ascites). X-rays may also be used to detect pathology such as gallstones (which are rarely radiopaque) or kidney stones which are often (but not always) visible. Traditional plain X-rays are less useful in the imaging of soft tissues such as the brain or muscle. One area where projectional radiographs are used extensively is in evaluating how an orthopedic implant,
such as a knee, hip or shoulder replacement, is situated in the body
with respect to the surrounding bone. This can be assessed in two
dimensions from plain radiographs, or it can be assessed in three
dimensions if a technique called '2D to 3D registration' is used. This
technique purportedly negates projection errors associated with
evaluating implant position from plain radiographs.
Dental radiography is commonly used in the diagnoses of common oral problems, such as cavities.
In medical diagnostic applications, the low energy (soft) X-rays
are unwanted, since they are totally absorbed by the body, increasing
the radiation dose without contributing to the image. Hence, a thin
metal sheet, often of aluminium, called an X-ray filter, is usually placed over the window of the X-ray tube, absorbing the low energy part in the spectrum. This is called hardening the beam since it shifts the center of the spectrum towards higher energy (or harder) X-rays.
To generate an image of the cardiovascular system, including the arteries and veins (angiography)
an initial image is taken of the anatomical region of interest. A
second image is then taken of the same region after an iodinated contrast agent
has been injected into the blood vessels within this area. These two
images are then digitally subtracted, leaving an image of only the
iodinated contrast outlining the blood vessels. The radiologist or surgeon
then compares the image obtained to normal anatomical images to
determine whether there is any damage or blockage of the vessel.
Computed tomography
Computed tomography (CT scanning) is a medical imaging modality where tomographic images
or slices of specific areas of the body are obtained from a large
series of two-dimensional X-ray images taken in different directions. These cross-sectional images can be combined into a three-dimensional image of the inside of the body and used for diagnostic and therapeutic purposes in various medical disciplines.
Fluoroscopy
Fluoroscopy is an imaging technique commonly used by physicians or radiation therapists
to obtain real-time moving images of the internal structures of a
patient through the use of a fluoroscope. In its simplest form, a
fluoroscope consists of an X-ray source and a fluorescent screen,
between which a patient is placed. However, modern fluoroscopes couple
the screen to an X-ray image intensifier and CCD video camera
allowing the images to be recorded and played on a monitor. This method
may use a contrast material. Examples include cardiac catheterization
(to examine for coronary artery blockages) and barium swallow (to examine for esophageal disorders and swallowing disorders).
Radiotherapy
The use of X-rays as a treatment is known as radiation therapy and is largely used for the management (including palliation) of cancer;
it requires higher radiation doses than those received for imaging
alone. X-rays beams are used for treating skin cancers using lower
energy X-ray beams while higher energy beams are used for treating
cancers within the body such as brain, lung, prostate, and breast.
Adverse effects
Abdominal radiograph of a pregnant woman, a procedure that should be performed only after proper assessment of benefit versus risk
Diagnostic X-rays (primarily from CT scans due to the large dose used) increase the risk of developmental problems and cancer in those exposed. X-rays are classified as a carcinogen by both the World Health Organization's International Agency for Research on Cancer and the U.S. government. It is estimated that 0.4% of current cancers in the United States are due to computed tomography (CT scans) performed in the past and that this may increase to as high as 1.5–2% with 2007 rates of CT usage.
Experimental and epidemiological data currently do not support the proposition that there is a threshold dose of radiation below which there is no increased risk of cancer. However, this is under increasing doubt.
It is estimated that the additional radiation from diagnostic X-rays
will increase the average person's cumulative risk of getting cancer by
age 75 by 0.6–3.0%. The amount of absorbed radiation depends upon the type of X-ray test and the body part involved. CT and fluoroscopy entail higher doses of radiation than do plain X-rays.
To place the increased risk in perspective, a plain chest X-ray will expose a person to the same amount from background radiation
that people are exposed to (depending upon location) every day over 10
days, while exposure from a dental X-ray is approximately equivalent to 1
day of environmental background radiation.
Each such X-ray would add less than 1 per 1,000,000 to the lifetime
cancer risk. An abdominal or chest CT would be the equivalent to 2–3
years of background radiation to the whole body, or 4–5 years to the
abdomen or chest, increasing the lifetime cancer risk between 1 per
1,000 to 1 per 10,000. This is compared to the roughly 40% chance of a US citizen developing cancer during their lifetime.
For instance, the effective dose to the torso from a CT scan of the
chest is about 5 mSv, and the absorbed dose is about 14 mGy. A head CT scan (1.5 mSv, 64 mGy)
that is performed once with and once without contrast agent, would be
equivalent to 40 years of background radiation to the head. Accurate
estimation of effective doses due to CT is difficult with the estimation
uncertainty range of about ±19% to ±32% for adult head scans depending
upon the method used.
The risk of radiation is greater to a fetus, so in pregnant
patients, the benefits of the investigation (X-ray) should be balanced
with the potential hazards to the fetus. In the US, there are an estimated 62 million CT scans performed annually, including more than 4 million on children. Avoiding unnecessary X-rays (especially CT scans) reduces radiation dose and any associated cancer risk.
Medical X-rays are a significant source of human-made radiation
exposure. In 1987, they accounted for 58% of exposure from human-made
sources in the United States.
Since human-made sources accounted for only 18% of the total radiation
exposure, most of which came from natural sources (82%), medical X-rays
only accounted for 10% of total American radiation exposure; medical procedures as a whole (including nuclear medicine)
accounted for 14% of total radiation exposure. By 2006, however,
medical procedures in the United States were contributing much more
ionizing radiation than was the case in the early 1980s. In 2006,
medical exposure constituted nearly half of the total radiation exposure
of the U.S. population from all sources. The increase is traceable to
the growth in the use of medical imaging procedures, in particular computed tomography (CT), and to the growth in the use of nuclear medicine.
Dosage due to dental X-rays varies significantly depending on the
procedure and the technology (film or digital). Depending on the
procedure and the technology, a single dental X-ray of a human results
in an exposure of 0.5 to 4 mrem. A full mouth series of X-rays may
result in an exposure of up to 6 (digital) to 18 (film) mrem, for a
yearly average of up to 40 mrem.
Financial incentives have been shown to have a significant impact
on X-ray use with doctors who are paid a separate fee for each X-ray
providing more X-rays.
Early photon tomography or EPT (as of 2015) along with other techniques are being researched as potential alternatives to X-rays for imaging applications.
Other uses
Other notable uses of X-rays include:
Each
dot, called a reflection, in this diffraction pattern forms from the
constructive interference of scattered X-rays passing through a crystal.
The data can be used to determine the crystalline structure.
- X-ray crystallography in which the pattern produced by the diffraction
of X-rays through the closely spaced lattice of atoms in a crystal is
recorded and then analysed to reveal the nature of that lattice. A
related technique, fiber diffraction, was used by Rosalind Franklin to discover the double helical structure of DNA.
- X-ray astronomy, which is an observational branch of astronomy, which deals with the study of X-ray emission from celestial objects.
- X-ray microscopic analysis, which uses electromagnetic radiation in the soft X-ray band to produce images of very small objects.
- X-ray fluorescence,
a technique in which X-rays are generated within a specimen and
detected. The outgoing energy of the X-ray can be used to identify the
composition of the sample.
- Industrial radiography uses X-rays for inspection of industrial parts, particularly welds.
- Radiography of cultural objects, most often x-rays of paintings to reveal underdrawing, pentimenti alterations in the course of painting or by later restorers, and sometimes previous paintings on the support. Many pigments such as lead white show well in radiographs.
- X-ray spectromicroscopy has been used to analyse the reactions of
pigments in paintings. For example, in analysing colour degradation in
the paintings of van Gogh.
Using
X-ray for inspection and quality control: the differences in the
structures of the die and bond wires reveal the left chip to be
counterfeit.
- Authentication and quality control of packaged items.
- Industrial CT
(computed tomography), a process that uses X-ray equipment to produce
three-dimensional representations of components both externally and
internally. This is accomplished through computer processing of
projection images of the scanned object in many directions.
- Airport security luggage scanners use X-rays for inspecting the interior of luggage for security threats before loading on aircraft.
- Border control truck scanners and domestic police departments use X-rays for inspecting the interior of trucks.
Visibility
While
generally considered invisible to the human eye, in special
circumstances X-rays can be visible. Brandes, in an experiment a short
time after Röntgen's
landmark 1895 paper, reported after dark adaptation and placing his eye
close to an X-ray tube, seeing a faint "blue-gray" glow which seemed to
originate within the eye itself.
Upon hearing this, Röntgen reviewed his record books and found he too
had seen the effect. When placing an X-ray tube on the opposite side of a
wooden door Röntgen had noted the same blue glow, seeming to emanate
from the eye itself, but thought his observations to be spurious because
he only saw the effect when he used one type of tube. Later he realized
that the tube which had created the effect was the only one powerful
enough to make the glow plainly visible and the experiment
was thereafter readily repeatable. The knowledge that X-rays are
actually faintly visible to the dark-adapted naked eye has largely been
forgotten today; this is probably due to the desire not to repeat what
would now be seen as a recklessly dangerous and potentially harmful
experiment with ionizing radiation.
It is not known what exact mechanism in the eye produces the
visibility: it could be due to conventional detection (excitation of rhodopsin
molecules in the retina), direct excitation of retinal nerve cells, or
secondary detection via, for instance, X-ray induction of phosphorescence in the eyeball with conventional retinal detection of the secondarily produced visible light.
Though X-rays are otherwise invisible, it is possible to see the ionization of the air molecules if the intensity of the X-ray beam is high enough. The beamline from the wiggler at the ID11 at the European Synchrotron Radiation Facility is one example of such high intensity.
Units of measure and exposure
The measure of X-rays ionizing ability is called the exposure:
- The coulomb per kilogram (C/kg) is the SI unit of ionizing radiation
exposure, and it is the amount of radiation required to create one
coulomb of charge of each polarity in one kilogram of matter.
- The roentgen (R) is an obsolete traditional unit of exposure, which represented the amount of radiation required to create one electrostatic unit of charge of each polarity in one cubic centimeter of dry air. 1 roentgen = 2.58×10−4 C/kg.
However, the effect of ionizing radiation on matter (especially living tissue) is more closely related to the amount of energy deposited into them rather than the charge generated. This measure of energy absorbed is called the absorbed dose:
- The gray (Gy), which has units of (joules/kilogram), is the SI unit of absorbed dose, and it is the amount of radiation required to deposit one joule of energy in one kilogram of any kind of matter.
- The rad is the (obsolete) corresponding traditional unit, equal to 10 millijoules of energy deposited per kilogram. 100 rad = 1 gray.
The equivalent dose is the measure of the biological effect of radiation on human tissue. For X-rays it is equal to the absorbed dose.
- The Roentgen equivalent man (rem) is the traditional unit of equivalent dose. For X-rays it is equal to the rad, or, in other words, 10 millijoules of energy deposited per kilogram. 100 rem = 1 Sv.
- The sievert (Sv) is the SI unit of equivalent dose, and also of effective dose. For X-rays the "equivalent dose" is numerically equal to a Gray (Gy). 1 Sv = 1 Gy. For the "effective dose" of X-rays, it is usually not equal to the Gray (Gy).