A Medley of Potpourri is just what it says; various thoughts, opinions, ruminations, and contemplations on a variety of subjects.
Search This Blog
Mystical or religious experience
From Wikipedia, the free encyclopedia https://en.wikipedia.org/wiki/Mystical_or_religious_experience A mysti...
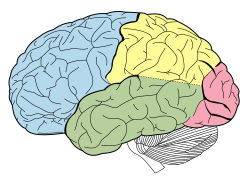
-
From Wikipedia, the free encyclopedia Ward Cunningham , inventor of the wiki A wiki is a website on whi...
-
From Wikipedia, the free encyclopedia https://en.wikipedia.org/wiki/Human_cloning Diagram of ...
-
From Wikipedia, the free encyclopedia A reproduction of the palm -leaf manuscript in Siddham script ...