From Wikipedia, the free encyclopedia
Management of depression is the treatment of depression that may involve a number of different therapies: medications, behavior therapy, psychotherapy, and medical devices.
Depression is a symptom of some physical diseases; a side effect of some drugs and medical treatments; and a symptom of some mood disorders such as major depressive disorder or dysthymia. Physical causes are ruled out with a clinical assessment of depression that measures vitamins, minerals, electrolytes, and hormones.
Though psychiatric medication is the most frequently prescribed therapy for major depression, psychotherapy may be effective, either alone or in combination with medication.
Combining psychotherapy and antidepressants may provide a "slight
advantage", but antidepressants alone or psychotherapy alone are not
significantly different from other treatments, like "active intervention
controls". ( e.g., sham acupuncture)
Given an accurate diagnosis of major depressive disorder, in general
the type of treatment (psychotherapy and/or antidepressants, alternate
or other treatments, or active intervention) is "less important than
getting depressed patients involved in an active therapeutic program."
Psychotherapy is the treatment of choice in those under the age
of 18, with medication offered only in conjunction with the former and
generally not as a first line agent. The possibility of depression,
substance misuse or other mental health problems in the parents should
be considered and, if present and if it may help the child, the parent
should be treated in parallel with the child.
Psychotherapy and behavior therapy
There are a number of different psychotherapies for depression which
are provided to individuals or groups by psychotherapists,
psychiatrists, psychologists, clinical social workers,
counselors or psychiatric nurses. With more chronic forms of
depression, the most effective treatment is often considered to be a
combination of medication and psychotherapy. Psychotherapy is the treatment of choice in people under 18.
A meta-analysis examined the effectiveness of psychotherapy for
depression across ages from younger than 13 years to older than 75
years. It summarizes results from 366 trials included 36,702 patients.
It found that the best results were for young adults, with an average effect size of g=.98 (95% CI, 0.79-1.16). The effects were smallest for young children (<13 years), g = .35 (95% CI, 0.15-0.55), and second largest in the oldest group, g = .97 (95% CI,
0.42-1.52). The study was not able to compare the different types of
therapy to each other. Most of the studies with children used therapies
originally developed with adults, which may have reduced the
effectiveness. The greater benefits with young adults might be due to a
large number of studies including college students, who might have an
easier time learning therapy skills and techniques.
Most of the studies in children were done in the US, whereas in older
age groups, more balanced numbers of studies came from Europe and other
parts of the world as well.
As the most studied form of psychotherapy for depression, cognitive behavioral therapy
(CBT) is thought to work by teaching clients to learn a set of
cognitive and behavioral skills, which they can employ on their own.
Earlier research suggested that cognitive behavioral therapy was not as
effective as antidepressant medication in the treatment of depression;
however, more recent research suggests that it can perform as well as
antidepressants in treating patients with moderate to severe depression. Beck's treatment manual, Cognitive therapy of depression, has undergone the most research and accumulated the most evidence for its use. However, a number of other CBT manuals also have evidence to support their effectiveness with depression.
The effect of psychotherapy on patient and clinician rated
improvement as well as on revision rates have declined steadily from the
1970s.
A systematic review of data comparing low-intensity CBT (such as
guided self-help by means of written materials and limited professional
support, and website-based interventions) with usual care found that
patients who initially had more severe depression benefited from
low-intensity interventions at least as much as less-depressed patients.
For the treatment of adolescent depression, one published study found that CBT without medication performed no better than a placebo, and significantly worse than the antidepressant fluoxetine. However, the same article reported that CBT and fluoxetine outperformed treatment with only fluoxetine. Combining fluoxetine with CBT appeared to bring no additional benefit in two different studies or, at the most, only marginal benefit, in a fourth study.
Behavior therapy for depression is sometimes referred to as behavioral activation. In addition, behavioral activation appears to take less time and lead to longer lasting change. Two well-researched treatment manuals include Social skills training for depression and Behavioral activation treatment for depression.
Emotionally focused therapy,
founded by Sue Johnson and Les Greenberg in 1985, treats depression by
identifying and processing underlying emotions. The treatment manual, Facilitating emotional change, outlines treatment techniques.
Acceptance and commitment therapy
(ACT), a mindfulness form of CBT, which has its roots in behavior
analysis, also demonstrates that it is effective in treating depression,
and can be more helpful than traditional CBT, especially where
depression is accompanied by anxiety and where it is resistant to
traditional CBT.
A review of four studies on the effectiveness of mindfulness-based cognitive therapy
(MBCT), a recently developed class-based program designed to prevent
relapse, suggests that MBCT may have an additive effect when provided
with the usual care in patients who have had three or more depressive
episodes, although the usual care did not include antidepressant
treatment or any psychotherapy, and the improvement observed may have
reflected non-specific or placebo effects. Of note, although Mindfulness-based cognitive therapy for depression
prevented relapse of future depressive episodes, there is no research
on whether it can cause the remission of a current depressive episode.
Interpersonal psychotherapy
(IPT) focuses on the social and interpersonal triggers that may cause
depression. There is evidence that it is an effective treatment for
depression.
Here, the therapy takes a fairly structured course (often 12 sessions,
as in the original research versions) as in the case with CBT; however,
the focus is on relationships with others. Unlike family therapy, IPT is
an individual format, so it is possible to work on interpersonal themes
even if other family members do not come to the session. Therapy can be
used to help a person develop or improve interpersonal skills in order to allow him or her to communicate more effectively and reduce stress. In a meta-analysis of 16 studies and 4,356 patients, the average improvement in depressive symptoms was an effect size of d = 0.63 (95% CI, 0.36 to 0.90).
IPT combined with pharmacotherapy was more effective in preventing
relapse than pharmacotherapy alone, number needed to treat = 7.63.
Psychoanalysis, a school of thought founded by Sigmund Freud that emphasizes the resolution of unconscious mental conflicts, is used by its practitioners to treat clients presenting with major depression. A more widely practiced technique, called psychodynamic psychotherapy, is loosely based on psychoanalysis and has an additional social and interpersonal focus.
In a meta-analysis of three controlled trials, psychodynamic
psychotherapy was found to be as effective as medication for mild to
moderate depression.
Shared care
Shared
decision making is an approach whereby patients and clinicians freely
share important evidence when tasked with decision making and where
patients are guided to consider the best available options to make an
informed decision.
The principles are well documented, but there is a gap in that it's
hard to apply them in routine clinical practice. The steps have been
simplified into five steps. The first step is seeking patient
participation in that the health practitioner is tasked with
communicating existing choices and therefore inviting them to the
decision-making process. The next step involves assisting the patient to
explore and compare the treatment options by a critical analysis of the
risks and benefits. The third step involves the assessment of the
patient's values and what they prefer taking to account what is of
paramount urgency to the patient. Step 4 involves decision making where
the patient and the practitioner make a conclusive decision on the best
option and arrange for subsequent follow up meetings. Finally, the fifth
step involves the analysis of the patient's decision'. Five steps for
you and your patients to work together to make the best possible health
care decisions. The step involves monitoring of the degree of
implementation, overcoming of barriers of decision implantation
consequently the decisions need to be revisited and optimized thus
ensuring the decision has a positive impact on health outcomes. Its
success relies on the ability of the health practitioner to create a
good interpersonal relationship with the patient. (Stone, 2017)
Depression is a major problem globally, affecting an estimated
4.4 percent of the world population in 2017, roughly equivalent to 300
million people.
The depression is multifactorial and has been on the increase due to
societal pressure, genetic association and increase in use of drugs
(Zhang et al. 2016).
incorporation of nursing in management of depression may seem important
in that nursing hold a pivotal role in health care delivery where they
are they are the health practitioners that have been trained to be
versatile from clinical to psychological care Their incorporation shared
decision making in treating depression may be important as nurses are
known to have the best interpersonal relationship with the patients thus
a better collaborative model can be achieved due to this fact (Williams
et al. 2016).
With this in mind, the nurses may serve to administer drugs in
management, prepare and maintain the patient's records, interaction with
other care staff to achieve optimum care, and organizing therapy
sessions (Lu et al. 2019).
Kathleen Walsh, 2017, recognizes that Dr. Velligan
stated that SDM is of importance in demonstrating patient preferences
in decision making when there is no clear approach to treatment. In
addition, numerous tools can be used to make the decision making the
process easier these include the Controlled Preferences Scale that
informs clinicians on how to actively involve patients. She further
gives the suggestion that providers need to embrace shared decision
making by making sure the patients participate actively in their
management thus enabling the success of the model.
Medication
To find the most effective pharmaceutical drug
treatment, the dosages of medications must often be adjusted, different
combinations of antidepressants tried, or antidepressants changed.
Selective serotonin reuptake inhibitors
Selective serotonin reuptake inhibitors (SSRIs), such as sertraline (Zoloft, Lustral), escitalopram (Lexapro, Cipralex), fluoxetine (Prozac), paroxetine (Seroxat), and citalopram,
are the primary medications considered, due to their relatively mild
side effects and broad effect on the symptoms of depression and anxiety,
as well as reduced risk in overdose, compared to their older tricyclic
alternatives. Those who do not respond to the first SSRI tried can be
switched to another. If sexual dysfunction is present prior to the onset
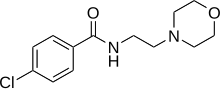
of depression, SSRIs should be avoided. Another popular option is to switch to the atypical antidepressant bupropion (Wellbutrin) or to add bupropion to the existing therapy; this strategy is possibly more effective. It is not uncommon for SSRIs to cause or worsen insomnia; the sedating noradrenergic and specific serotonergic antidepressant (NaSSA) antidepressant mirtazapine (Zispin, Remeron) can be used in such cases.
For children and adolescents with moderate-to-severe depressive
disorder, fluoxetine seems to be the best treatment (either with or
without cognitive behavioural therapy) but more research is needed to be certain. Sertraline, escitalopram, duloxetine might also help in reducing symptoms.
In the UK fluoxetine and escitalopram are the only antidepressants
recommended for people under the age of 18, though, if a child or
adolescent patient is intolerant to fluoxetine, another SSRI may be
considered.
Evidence of effectiveness of SSRIs in those with depression complicated by dementia is lacking.
Norepinephrine dopamine reuptake inhibitor
Some norepinephrine–dopamine reuptake inhibitors can be used as antidepressants.
Norepinephrine reuptake inhibitor
Norepinephrine reuptake inhibitors (NRIs) can be used as antidepressants.
Serotonin norepinephrine reuptake inhibitor
Venlafaxine (Effexor) from the SNRI class may be moderately more effective than SSRIs; however, it is not recommended as a first-line treatment because of the higher rate of side effects, and its use is specifically discouraged in children and adolescents.
Tricyclic antidepressant
Tricyclic antidepressants
(TCAs) have more side effects than SSRIs and are usually reserved for
the treatment of inpatients, for whom the tricyclic antidepressant amitriptyline, in particular, appears to be more effective.
Monoamine oxidase inhibitor
Monoamine oxidase inhibitors,
have historically been plagued by questionable efficacy (although early
studies used dosages now considered too low) and life-threatening
adverse effects. They are still used only rarely, although newer agents
of this class (RIMA), with a better side effect profile, have been developed.
In older patients TCAs and SSRIs are of the same efficacy.
However, there are differences between TCA related antidepressants and
classical TCAs in terms of side effect profiles and withdrawal when
compared to SSRIs.
There is evidence a prominent side-effect of antidepressants,
emotional blunting, is confused with a symptom of depression itself. The
cited study, according to Professor Linda Gask was:
'funded by a pharmaceutical company
(Servier) and two of its authors are employees of that company', which
may bias the results. The study authors' note: "emotional blunting is
reported by nearly half of depressed patients on antidepressants and
that it appears to be common to all monoaminergic antidepressants not
only SSRIs". Additionally, they note: "The OQuESA scores are highly
correlated with the HAD depression score; emotional blunting cannot be
described simply as a side-effect of antidepressant, but also as a
symptom of depression. More emotional blunting is associated with a
poorer quality of remission.
NMDA antagonists
Ketamine
Research on the antidepressant effects of ketamine
infusions at subanaesthetic doses has consistently shown rapid (4 to 72
hours) responses from single doses, with substantial improvement in
mood in the majority of patients and remission
in some. However, these effects are often short-lived, and attempts to
prolong the antidepressant effect with repeated doses and extended
("maintenance") treatment have resulted in only modest success. A nasal spray formulation of esketamine,
sold under the brand name Spravato, gained FDA approval in 2019 for the
treatment of treatment-resistant depression when combined with an oral
antidepressant.
Zinc
A 2012 cross-sectional study found an association between zinc deficiency and depressive symptoms among women, but not men,
and a 2013 meta-analysis of 17 observational studies found that blood
zinc concentrations were lower in depressed subjects than in control
subjects.
A 2012 meta-analysis found that zinc supplementation as an adjunct to
antidepressant drug treatment significantly lowered depressive symptom
scores of depressed patients.
The potential mechanisms underlying the association between low serum
zinc and depression remain unclear, but may involve the regulation of
neurotransmitter, endocrine and neurogenesis pathways. Zinc supplementation has been reported to improve symptoms of ADHD and depression. A 2013 review found that zinc supplementation may be an effective treatment in major depression.
Acetyl-l-carnitine
Acetylcarnitine levels were lower in depressed patients than controls and in rats it causes rapid antidepressant effects through epigenetic mechanisms.
A systematic review and meta-analysis of 12 randomized controlled
trials found "supplementation significantly decreases depressive
symptoms compared with placebo/no intervention, while offering a
comparable effect with that of established antidepressant agents with
fewer adverse effects."
Augmentation
Physicians
often add a medication with a different mode of action to bolster the
effect of an antidepressant in cases of treatment resistance; a 2002
large community study of 244,859 depressed Veterans Administration
patients found that 22% had received a second agent, most commonly a
second antidepressant. Stephen M. Stahl, renowned academician in psychopharmacology, has stated resorting to a dynamic psychostimulant, in particular, d-amphetamine is the "classical augmentation strategy for treatment-refractory depression". However, the use of stimulants in cases of treatment-resistant depression is relatively controversial.
It is also possible to use a benzodiazepine as to improve sleep
without impairing the antidepressant response specially in patients
presenting symptoms of insomnia and disturbed sleep. A randomized
controlled trial found that the use of eszopiclone with fluoxetine
resulted in a better remission rate.
Addition of atypical antipsychotics
when the patient has not responded to an antidepressant is also known
to increase the effectiveness of antidepressant drugs, albeit at the
cost of more frequent and potentially serious side effects.
Lithium
Lithium has been used to augment antidepressant therapy in those who have failed to respond to antidepressants alone. Furthermore, lithium dramatically decreases the suicide risk in recurrent depression.
According to the results of the STAR-D experiment, the remission rate of
lithium for treatment-resistant depression is about 15.9%.
Thyroid hormones
There is some evidence for the addition of a thyroid hormone, triiodothyronine, in patients with normal thyroid function.
For TRD patients, T3 has been studied in the STAR-D study with
having a remission rate of 24.7%. T4 is also being studied for this
purpose and found remission rates of 21.5% - 64.7% for TRD patients.
Efficacy of medication and psychotherapy
Antidepressants are statistically superior to placebo but their overall effect is low-to-moderate. In that respect they often did not exceed the National Institute for Health and Clinical Excellence
criteria for a "clinically significant" effect. In particular, the
effect size was very small for moderate depression but increased with
severity, reaching "clinical significance" for very severe depression.
These results were consistent with the earlier clinical studies in
which only patients with severe depression benefited from either
psychotherapy or treatment with an antidepressant, imipramine, more than from the placebo treatment. Despite obtaining similar results, the authors argued about their
interpretation. One author concluded that there "seems little evidence
to support the prescription of antidepressant medication to any but the
most severely depressed patients, unless alternative treatments have
failed to provide benefit."
The other author agreed that "antidepressant 'glass' is far from full"
but disagreed "that it is completely empty". He pointed out that the
first-line alternative to medication is psychotherapy, which does not
have superior efficacy.
Antidepressants in general are as effective as psychotherapy for
major depression, and this conclusion holds true for both severe and
mild forms of MDD. In contrast, medication gives better results for dysthymia.
The subgroup of SSRIs may be slightly more efficacious than
psychotherapy. On the other hand, significantly more patients drop off
from the antidepressant treatment than from psychotherapy, likely
because of the side effects of antidepressants.
Successful psychotherapy appears to prevent the recurrence of
depression even after it has been terminated or replaced by occasional
"booster" sessions. The same degree of prevention can be achieved by
continuing antidepressant treatment.
Two studies suggest that the combination of psychotherapy and
medication is the most effective way to treat depression in adolescents.
Both TADS (Treatment of Adolescents with Depression Study) and TORDIA
(Treatment of Resistant Depression in Adolescents) showed very similar
results. TADS resulted in 71% of their teen subjects having "much" or
"very much" improvement in mood over the 61% with medication alone and
43% with CBT alone. Similarly, TORDIA showed a 55% improvement with CBT and drugs versus a 41% with drug therapy alone.
However, a more recent meta-analysis of 34 trials of 14 drugs used with
children and adolescents found that only fluoxetine produced
significant benefit compared to placebo, with a medium-sized effect
(standardize mean difference = .5).
Treatment resistance
The risk factors for treatment resistant depression are: the duration of the episode of
depression, severity of the episode, if bipolar, lack of improvement in
symptoms within the first couple of treatment weeks, anxious or avoidant
and borderline comorbidity and old age. Treatment resistant depression
is best handled with a combination of conventional antidepressant
together with atypical antipsychotics. Another approach is to try
different antidepressants. It's inconclusive which approach is superior.
Treatment resistant depression can be misdiagnosed if subtherapeutic
doses of antidepressants is the case, patient nonadherence, intolerable
adverse effects or their thyroid disease or other conditions is
misdiagnosed as depression.
Experimental treatments
Ayahuasca
Research into ayahuasca has been recommended, given there is limited early evidence of potential antidepressant and anxiolytic effects.
Chromium
Clinical and experimental studies have reported antidepressant activity of chromium particularly in atypical depression, characterized by increased appetite and carbohydrate craving.
Creatine
The amino acid creatine, commonly used as a supplement to improve the performance of bodybuilders,
has been studied for its potential antidepressant properties. A review
found that creatine "has the potential to improve these disruptions [of
brain metabolism] in some patients, and early clinical trials indicate
that it may have efficacy as an antidepressant agent." Studies on mice have found that the antidepressant effects of creatine can be blocked by dopamine receptor antagonists such as haloperidol, suggesting that the drug acts on dopamine pathways.
Inositol
Inositol,
a sugar alcohol in fruits, beans, grains and nuts, was found to be
significantly better than placebo in treating depression in a
double-blind, controlled trial.
It was also reported to be reduced in human CSF in depression and found
to lead to “major improvement” in 9 of 11 depressed patients in an open
label trial.
Magnesium
A meta-analysis has found an association between magnesium intake and depression. Magnesium was lower in serum of depressed patients than controls.
A 2018 review found that Mg2+ supplementation (range 225–4000 mg) and number of weeks of treatment (range 1–12) were not related to changes in mood disorder.
Essential Fatty Acids
There is insufficient evidence to determine that omega-3 fatty acid has any effect on depression. A 2016 review found that if trials with formulations containing mostly eicosapentaenoic acid (EPA) are separated from trials using formulations containing docosahexaenoic acid (DHA), it appeared that EPA may have an effect while DHA may not, but there was insufficient evidence to be sure.
A 2020 meta-analysis showed that a high dose of omega-3
polyunsaturated fatty acid (>2g/day) used as an adjuvent improved
depressive symptoms.
Dopamine receptor agonist
Some research suggests dopamine receptor agonists, most commonly pramipexole, may be effective in treating depression. Studies are few and results are preliminary, however.
N-Acetylcysteine
A systematic review and meta-analysis of 5 studies found that N-acetylcysteine reduces depressive symptoms more than placebo and has good tolerability. N-acetylecysteine may exert its benefits by replenishing the chief cellular antioxidant, glutathione, thus modulating glutamatergic, neurotropic and inflammatory pathways.
Psilocybin
Psilocybin has been shown in several studies to improve symptoms in people with treatment-resistant depression. In 2018 and 2019, the FDA designated psilocybin as a "breakthrough therapy" for drug-resistant depression and major depressive disorder.
St John's wort
A 2008 Cochrane Collaboration meta-analysis concluded that "The available evidence suggests that the hypericum
extracts tested in the included trials a) are superior to placebo in
patients with major depression; b) are similarly effective as standard
antidepressants; c) and have fewer side effects
than standard antidepressants. The association of country of origin and
precision with effects sizes complicates the interpretation." The United States National Center for Complementary and Integrative Health
advice is that "St. John's wort may help some types of depression,
similar to treatment with standard prescription antidepressants, but the
evidence is not definitive." and warns that "Combining St. John's wort
with certain antidepressants can lead to a potentially life-threatening
increase of serotonin, a brain chemical targeted by antidepressants. St. John's wort can also limit the effectiveness of many prescription medicines."
Rhodiola rosea
A 2011 review reported Rhodiola rosea
"is an adaptogen plant that can be especially helpful in treating
asthenic or lethargic depression, and may be combined with conventional
antidepressants to alleviate some of their common side effects."
Saffron
A 2013 meta-analysis found that saffron
supplementation significantly reduced depression symptoms compared to
placebo, and both saffron supplementation and the antidepressant groups
were similarly effective in reducing depression symptoms.
A 2015 meta-analysis supported the "efficacy of saffron as compared to
placebo in improving the following conditions: depressive symptoms
(compared to anti-depressants and placebo), premenstrual symptoms, and
sexual dysfunction. In addition, saffron use was also effective in
reducing excessive snacking behavior." Therapeutic doses of saffron exhibits no significant toxicity in both clinical and experimental investigations.
SAMe
S-Adenosyl methionine (SAMe) is available as a prescription antidepressant in Europe and an over-the-counter dietary supplement
in the US. Evidence from 16 clinical trials with a small number of
subjects, reviewed in 1994 and 1996 suggested it to be more effective
than placebo and as effective as standard antidepressant medication for
the treatment of major depression.
Tryptophan and 5-HTP
The amino acid tryptophan is converted into 5-hydroxytryptophan (5-HTP) which is subsequently converted into the neurotransmitter serotonin.
Since serotonin deficiency has been recognized as a possible cause of
depression, it has been suggested that consumption of tryptophan or
5-HTP may therefore improve depression symptoms by increasing the level
of serotonin in the brain. 5-HTP and tryptophan are sold over the counter
in North America, but requires a prescription in Europe. The use of
5-HTP instead of tryptophan bypasses the conversion of tryptophan into
5-HTP by the enzyme tryptophan hydroxylase,
which is the rate-limiting step in the synthesis of serotonin, and
5-HTP easily crosses the blood–brain barrier unlike tryptophan, which
requires a transporter.
Small studies have been performed using 5-HTP and tryptophan as
adjunctive therapy in addition to standard treatment for depression.
While some studies had positive results, they were criticized for having
methodological flaws, and a more recent study did not find sustained
benefit from their use. The safety of these medications has not been well studied.
Due to the lack of high quality studies, preliminary nature of studies
showing effectiveness, the lack of adequate study on their safety, and
reports of Eosinophilia–myalgia syndrome from contaminated tryptophan in 1989 and 1990, the use of tryptophan and 5-HTP is not highly recommended or thought to be clinically useful.
Medical devices
A
variety of medical devices are in use or under consideration for
treatment of depression including devices that offer electroconvulsive
therapy, vagus nerve stimulation, repetitive transcranial magnetic stimulation, and cranial electrotherapy stimulation. The use of such devices in the United States requires approval by the U.S. Food and Drug Administration
(FDA) after field trials. In 2010 an FDA advisory panel considered the
question of how such field trials should be managed. Factors considered
were whether drugs had been effective, how many different drugs had been
tried, and what tolerance for suicides should be in field trials.
Electroconvulsive therapy
In 2004, a meta-analytic review paper found in terms of efficacy, "a
significant superiority of ECT in all comparisons: ECT versus simulated
ECT, ECT versus placebo, ECT versus antidepressants in general, ECT versus tricyclics and ECT versus monoamine oxidase inhibitors."
Electroconvulsive therapy (ECT) is a standard psychiatric treatment in which seizures are electrically induced in patients to provide relief from psychiatric illnesses. ECT is used with informed consent as a last line of intervention for major depressive disorder.
Among the elderly, who often experience depression, the efficacy of ECT
is difficult to determine due to the lack of trials comparing ECT to
other treatments.
A round of ECT is effective for about 50% of people with
treatment-resistant major depressive disorder, whether it is unipolar or
bipolar. Follow-up treatment is still poorly studied, but about half of people who respond relapse with twelve months.
Aside from effects in the brain, the general physical risks of ECT are similar to those of brief general anesthesia. Immediately following treatment, the most common adverse effects are confusion and memory loss. ECT is considered one of the least harmful treatment options available for severely depressed pregnant women.
A usual course of ECT involves multiple administrations,
typically given two or three times per week until the patient no longer
has symptoms ECT is administered under anesthetic with a muscle
relaxant.
Electroconvulsive therapy can differ in its application in three ways:
electrode placement, frequency of treatments, and the electrical
waveform of the stimulus. These three forms of application have
significant differences in both adverse side effects and symptom
remission. After treatment, drug therapy is usually continued, and some
patients receive maintenance ECT.
ECT appears to work in the short term via an anticonvulsant effect mostly in the frontal lobes, and longer term via neurotrophic effects primarily in the medial temporal lobe.
Deep brain stimulation
The support for the use of deep brain stimulation in treatment-resistant depression comes from a handful of case studies, and this treatment is still in a very early investigational stage. In this technique electrodes are implanted in a specific region of the brain, which is then continuously stimulated.
A March 2010 systematic review found that "about half the patients did
show dramatic improvement" and that adverse events were "generally
trivial" given the younger psychiatric patient population than with
movements disorders.
Deep brain stimulation is available on an experimental basis only in
the United States; no systems are approved by the FDA for this use.
Repetitive transcranial magnetic stimulation
Transcranial magnetic stimulation (TMS) or deep transcranial magnetic stimulation
is a noninvasive method used to stimulate small regions of the brain.
During a TMS procedure, a magnetic field generator, or "coil" is placed
near the head of the person receiving the treatment. The coil produces small electric currents in the region of the brain just under the coil via electromagnetic induction. The coil is connected to a pulse generator, or stimulator, that delivers electric current to the coil.
TMS was approved by the FDA for treatment-resistant major depressive disorder in 2008 and as of 2014 clinical evidence supports this use. The American Psychiatric Association, the Canadian Network for Mood and Anxiety Disorders, and the Royal Australia and New Zealand College of Psychiatrists have endorsed rTMS for trMDD.
The response rate is about 29% for TRD patients. Remission rate is about 20%.
Vagus nerve stimulation
Vagus nerve stimulation
(VNS) uses an implanted electrode and generator to deliver electrical
pulses to the vagus nerve, one of the primary nerves emanating from the
brain. It is an approved therapy for treatment-resistant depression in
the EU and US and is sometimes used as an adjunct to existing
antidepressant treatment. The support for this method comes mainly from
open-label trials, which indicate that several months may be required to
see a benefit.
The only large double-blind trial conducted lasted only 10 weeks and
yielded inconclusive results; VNS failed to show superiority over a sham
treatment on the primary efficacy outcome, but the results were more
favorable for one of the secondary outcomes. The authors concluded "This
study did not yield definitive evidence of short-term efficacy for
adjunctive VNS in treatment-resistant depression."
Cranial electrotherapy stimulation
A 2014 Cochrane review found insufficient evidence to determine whether or not Cranial electrotherapy stimulation with alternating current is safe and effective for treating depression.
Transcranial direct current stimulation
A 2016 meta-analysis of transcranial direct current stimulation
(tDCS) reported some efficacy of tDCS in the treatment of acute
depressive disorder with moderate effect size, and low efficacy in
treatment-resistant depression, and that use of 2 mA current strength
over 20 min per day over a short time span can be considered safe.
Other treatments
Herbal remedies
Due
to safety concerns and side effects of many antidepressant medications,
herbal psychopharmacology research has increased, and herbal remedies
are becoming increasingly popular as alternatives to prescribed
medications for the treatment of major depressive disorder. Findings
from clinical trials conducted to date indicate that saffron
supplementation can improve symptoms of depression in adults. Overall
results showed that saffron possessed better efficacy in the improvement
of depressive symptoms when compared with placebo, whereas saffron was
as effective as synthetic antidepressants.
Bright light therapy
Bright light therapy is sometimes used to treat depression, especially in its
seasonal form.
A meta-analysis of bright light therapy commissioned by the American Psychiatric Association
found a significant reduction in depression symptom severity associated
with bright light treatment. Benefit was found for both seasonal affective disorder
and for nonseasonal depression, with effect sizes similar to those for
conventional antidepressants. For non-seasonal depression, adding light
therapy to the standard antidepressant treatment was not effective.
A meta-analysis of light therapy for non-seasonal depression conducted
by Cochrane Collaboration, studied a different set of trials, where
light was used mostly in combination with antidepressants or wake therapy.
A moderate statistically significant effect of light therapy was found,
with response significantly better than control treatment in
high-quality studies, in studies that applied morning light treatment,
and with patients who respond to total or partial sleep deprivation.
Both analyses noted poor quality of most studies and their small size,
and urged caution in the interpretation of their results. The short 1–2
weeks duration of most trials makes it unclear whether the effect of
light therapy could be sustained in the longer term.
Exercise
Physical exercise is one recommended way to manage mild depression, such as by playing
soccer.
The 2013 Cochrane Collaboration review on physical exercise
for depression noted that, based upon limited evidence, it is
moderately more effective than a control intervention and comparable to
psychological or antidepressant drug therapies. Smaller effects were
seen in more methodologically rigorous studies.
Three subsequent 2014 systematic reviews that included the Cochrane
review in their analysis concluded with similar findings: one indicated
that physical exercise is effective as an adjunct treatment with antidepressant medication;
the other two indicated that physical exercise has marked
antidepressant effects and recommended the inclusion of physical
activity as an adjunct treatment for mild–moderate depression and mental illness in general. These studies also found smaller effect sizes in more methodologically rigorous studies.
All four systematic reviews called for more research in order to
determine the efficacy or optimal exercise intensity, duration, and
modality. The evidence for brain-derived neurotrophic factor (BDNF) in mediating some of the neurobiological effects of physical exercise was noted in one review which hypothesized that increased BDNF signaling is responsible for the antidepressant effect. A meta-analysis
of 15 studies published in 2022 suggested a curvilinear dose-response
relationship between exercise and depression risk, with low levels of
exercise showing the best dose-response.
Meditation
Mindfulness meditation
programs may help improve symptoms of depression, but they are no
better than active treatments such as medication, exercise, and other
behavioral therapies.
Music therapy
A 2009 review found that 3 to 10 sessions of music therapy
(when added to standard care) resulted in a noticeable improvement in
depressive symptoms, with still greater improvement after 16 to 51
sessions.
A 2017 cochrane systematic review found that music therapy added
to the usual treatment of depression gives better outcome than the usual
treatment alone: "The effect size translates to a difference of 9.8
points on the HAM-D". It also found that there is no significant
difference between active and receptive music therapy comparing
depression outcome. It is also important to note that music therapy is
not associated with more or fewer adverse events than treatment as
usual.
Sleep
Depression is sometimes associated with insomnia
- (difficulty in falling asleep, early waking, or waking in the middle
of the night). The combination of these two results, depression and
insomnia, will only worsen the situation. Hence, good sleep hygiene is important to help break this vicious circle. It would include measures such as regular sleep routines, avoidance of stimulants such as caffeine and management of sleeping disorders such as sleep apnea.
Smoking cessation
Quitting smoking cigarettes is associated with reduced depression and anxiety, with the effect "equal or larger than" those of antidepressant treatments.
Total/partial sleep deprivation
Sleep deprivation
(skipping a night's sleep) has been found to improve symptoms of
depression in 40% - 60% of patients. Partial sleep deprivation in the
second half of the night may be as effective as an all night sleep
deprivation session. Improvement may last for weeks, though the
majority (50%-80%) relapse after recovery sleep. Shifting or reduction
of sleep time, light therapy, antidepressant drugs, and lithium have been found to potentially stabilize sleep deprivation treatment effects.
Shared Care
Shared
care, when primary and specialty physicians have joint management of an
individual's health care, has been shown to alleviate depression
outcomes.
Research
Trials are investigating whether botulinum toxin, when used to make a person appear to frown less, stops negative feedback from the face and affects depression.
Psilocybin may have a beneficial role in the treatment of depression.
A 2019 meta-analysis found that hypnotherapy may be an effective way of alleviating the symptoms of depression.
No model of depression in animals that fully explains the mechanism of depression has been found as of 2019.