A
figure comparing the three types of enzyme inhibitors and how they work
in regards to substrate binding sites and inhibitors binding sites.
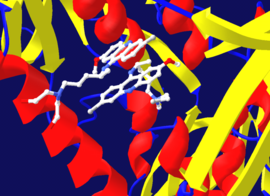
An enzyme binding site that would normally bind substrate can alternatively bind a competitive inhibitor, preventing substrate access. Dihydrofolate reductase is inhibited by methotrexate which prevents binding of its substrate, folic acid. Binding site in blue, inhibitor in green, and substrate in black. (PDB: 4QI9)
An enzyme inhibitor is a molecule that binds to an enzyme and decreases its activity.
By binding to enzymes' active sites, inhibitors reduce the
compatibility of substrate and enzyme and this leads to the inhibition
of Enzyme-Substrate complexes' formation, preventing the catalyzation of
reactions and decreasing (at times to zero) the amount of product
produced by a reaction. It can be said that as the concentration of
enzyme inhibitors increases, the rate of enzyme activity decreases, and
thus, the amount of product produced is inversely proportional to the
concentration of inhibitor molecules.
Since blocking an enzyme's activity can kill a pathogen or correct a metabolic imbalance, many drugs are enzyme inhibitors. They are also used in pesticides. Not all molecules that bind to enzymes are inhibitors; enzyme activators bind to enzymes and increase their enzymatic activity, while enzyme substrates bind and are converted to products in the normal catalytic cycle of the enzyme.
The binding of an inhibitor can stop a substrate from entering the enzyme's active site and/or hinder the enzyme from catalyzing its reaction. Inhibitor binding is either reversible or irreversible. Irreversible inhibitors usually react with the enzyme and change it chemically (e.g. via covalent bond formation). These inhibitors modify key amino acid residues needed for enzymatic activity. In contrast, reversible inhibitors bind non-covalently and different types of inhibition are produced depending on whether these inhibitors bind to the enzyme, the enzyme-substrate complex, or both.
Many drug molecules are enzyme inhibitors, so their discovery and improvement is an active area of research in biochemistry and pharmacology. A medicinal enzyme inhibitor is often judged by its specificity (its lack of binding to other proteins) and its potency (its dissociation constant,
which indicates the concentration needed to inhibit the enzyme). A high
specificity and potency ensure that a drug will have few side effects and thus low toxicity.
Enzyme inhibitors also occur naturally and are involved in the regulation of metabolism. For example, enzymes in a metabolic pathway can be inhibited by downstream products. This type of negative feedback slows the production line when products begin to build up and is an important way to maintain homeostasis in a cell. Other cellular enzyme inhibitors are proteins that specifically bind to and inhibit an enzyme target. This can help control enzymes that may be damaging to a cell, like proteases or nucleases. A well-characterised example of this is the ribonuclease inhibitor, which binds to ribonucleases in one of the tightest known protein–protein interactions. Natural enzyme inhibitors can also be poisons and are used as defenses against predators or as ways of killing prey.
Reversible inhibitors
Types of reversible inhibitors
Reversible inhibitors attach to enzymes with non-covalent interactions such as hydrogen bonds, hydrophobic interactions and ionic bonds. Multiple weak bonds between the inhibitor and the active site combine to produce strong and specific binding. In contrast to substrates
and irreversible inhibitors, reversible inhibitors generally do not
undergo chemical reactions when bound to the enzyme and can be easily
removed by dilution or dialysis.
There are four kinds of reversible enzyme inhibitors. They are
classified according to the effect of varying the concentration of the
enzyme's substrate on the inhibitor.
Types of inhibition. This classification was introduced by W.W. Cleland.
- In competitive inhibition, the substrate and inhibitor cannot bind to the enzyme at the same time, as shown in the figure on the right. This usually results from the inhibitor having an affinity for the active site of an enzyme where the substrate also binds; the substrate and inhibitor compete for access to the enzyme's active site. This type of inhibition can be overcome by sufficiently high concentrations of substrate (Vmax remains constant), i.e., by out-competing the inhibitor. However, the apparent Km will increase as it takes a higher concentration of the substrate to reach the Km point, or half the Vmax. Competitive inhibitors are often similar in structure to the real substrate (see examples below).
- In uncompetitive inhibition, the inhibitor binds only to the substrate-enzyme complex. This type of inhibition causes Vmax to decrease (maximum velocity decreases as a result of removing activated complex) and Km to decrease (due to better binding efficiency as a result of Le Chatelier's principle and the effective elimination of the ES complex thus decreasing the Km which indicates a higher binding affinity).
- In non-competitive inhibition, the binding of the inhibitor to the enzyme reduces its activity but does not affect the binding of substrate. As a result, the extent of inhibition depends only on the concentration of the inhibitor. Vmax will decrease due to the inability for the reaction to proceed as efficiently, but Km will remain the same as the actual binding of the substrate, by definition, will still function properly.
- In mixed inhibition, the inhibitor can bind to the enzyme at the same time as the enzyme's substrate. However, the binding of the inhibitor affects the binding of the substrate, and vice versa. This type of inhibition can be reduced, but not overcome by increasing concentrations of substrate. Although it is possible for mixed-type inhibitors to bind in the active site, this type of inhibition generally results from an allosteric effect where the inhibitor binds to a different site on an enzyme. Inhibitor binding to this allosteric site changes the conformation (i.e., tertiary structure or three-dimensional shape) of the enzyme so that the affinity of the substrate for the active site is reduced.
Quantitative description of reversible inhibition
Reversible inhibition can be described quantitatively in terms of the inhibitor's binding to the enzyme and to the enzyme-substrate complex, and its effects on the kinetic constants of the enzyme. In the classic Michaelis-Menten scheme
below, an enzyme (E) binds to its substrate (S) to form the
enzyme–substrate complex ES. Upon catalysis, this complex breaks down to
release product P and free enzyme. The inhibitor (I) can bind to either
E or ES with the dissociation constants Ki or Ki', respectively.
|
Kinetic scheme for reversible enzyme inhibitors
|
When an enzyme has multiple substrates, inhibitors can show different
types of inhibition depending on which substrate is considered. This
results from the active site containing two different binding sites
within the active site, one for each substrate. For example, an
inhibitor might compete with substrate A for the first binding site, but
be a non-competitive inhibitor with respect to substrate B in the
second binding site.
Measuring the dissociation constants of a reversible inhibitor
As noted above, an enzyme inhibitor is characterised by its two dissociation constants, Ki and Ki', to the enzyme and to the enzyme-substrate complex, respectively. The enzyme-inhibitor constant Ki can be measured directly by various methods; one extremely accurate method is isothermal titration calorimetry, in which the inhibitor is titrated into a solution of enzyme and the heat released or absorbed is measured. However, the other dissociation constant Ki'
is difficult to measure directly, since the enzyme-substrate complex is
short-lived and undergoing a chemical reaction to form the product.
Hence, Ki' is usually measured indirectly, by observing the enzyme activity under various substrate and inhibitor concentrations, and fitting the data to a modified Michaelis–Menten equation
where the modifying factors α and α' are defined by the inhibitor concentration and its two dissociation constants
Thus, in the presence of the inhibitor, the enzyme's effective Km and Vmax become (α/α')Km and (1/α')Vmax,
respectively. However, the modified Michaelis-Menten equation assumes
that binding of the inhibitor to the enzyme has reached equilibrium,
which may be a very slow process for inhibitors with sub-nanomolar
dissociation constants. In these cases, it is usually more practical to
treat the tight-binding inhibitor as an irreversible inhibitor (see
below); however, it can still be possible to estimate Ki' kinetically if Ki is measured independently.
The effects of different types of reversible enzyme inhibitors on
enzymatic activity can be visualized using graphical representations of
the Michaelis–Menten equation, such as Lineweaver–Burk and Eadie-Hofstee plots. For example, in the Lineweaver–Burk plots at the right, the competitive inhibition lines intersect on the y-axis, illustrating that such inhibitors do not affect Vmax. Similarly, the non-competitive inhibition lines intersect on the x-axis, showing these inhibitors do not affect Km. However, it can be difficult to estimate Ki and Ki' accurately from such plots, so it is advisable to estimate these constants using more reliable nonlinear regression methods, as described above.
Reversible inhibitors
Traditionally
reversible enzyme inhibitors have been classified as competitive,
uncompetitive, or non-competitive, according to their effects on Km and Vmax.
These different effects result from the inhibitor binding to the enzyme
E, to the enzyme–substrate complex ES, or to both, respectively. The
division of these classes arises from a problem in their derivation and
results in the need to use two different binding constants for one
binding event. The binding of an inhibitor and its effect on the
enzymatic activity are two distinctly different things, another problem
the traditional equations fail to acknowledge. In noncompetitive
inhibition the binding of the inhibitor results in 100% inhibition of
the enzyme only, and fails to consider the possibility of anything in
between.
The common form of the inhibitory term also obscures the relationship
between the inhibitor binding to the enzyme and its relationship to any
other binding term be it the Michaelis–Menten equation or a dose
response curve associated with ligand receptor binding. To demonstrate
the relationship the following rearrangement can be made:
This rearrangement demonstrates that similar to the Michaelis–Menten
equation, the maximal rate of reaction depends on the proportion of the
enzyme population interacting with its substrate.
Fraction of the enzyme population bound by substrate:
Fraction of the enzyme population bound by inhibitor:
the effect of the inhibitor is a result of the percent of the enzyme
population interacting with inhibitor. The only problem with this
equation in its present form is that it assumes absolute inhibition of
the enzyme with inhibitor binding, when in fact there can be a wide
range of effects anywhere from 100% inhibition of substrate turn over to
just >0%. To account for this the equation can be easily modified
to allow for different degrees of inhibition by including a delta Vmax term.
or
This term can then define the residual enzymatic activity present
when the inhibitor is interacting with individual enzymes in the
population. However the inclusion of this term has the added value of
allowing for the possibility of activation if the secondary Vmax
term turns out to be higher than the initial term. To account for the
possibly of activation as well the notation can then be rewritten
replacing the inhibitor "I" with a modifier term denoted here as "X".
While this terminology results in a simplified way of dealing with
kinetic effects relating to the maximum velocity of the Michaelis–Menten
equation, it highlights potential problems with the term used to
describe effects relating to the Km. The Km
relating to the affinity of the enzyme for the substrate should in most
cases relate to potential changes in the binding site of the enzyme
which would directly result from enzyme inhibitor interactions. As such
a term similar to the one proposed above to modulate Vmax should be appropriate in most situations:
Special cases
- The mechanism of partially competitive inhibition is similar to that of non-competitive, except that the EIS complex has catalytic activity, which may be lower or even higher (partially competitive activation) than that of the enzyme–substrate (ES) complex. This inhibition typically displays a lower Vmax, but an unaffected Km value.
- Uncompetitive inhibition occurs when the inhibitor binds only to the enzyme–substrate complex, not to the free enzyme; the EIS complex is catalytically inactive. This mode of inhibition is rare and causes a decrease in both Vmax and the Km value.
- Substrate and product inhibition is where either the substrate or product of an enzyme reaction inhibit the enzyme's activity. This inhibition may follow the competitive, uncompetitive or mixed patterns. In substrate inhibition there is a progressive decrease in activity at high substrate concentrations. This may indicate the existence of two substrate-binding sites in the enzyme. At low substrate, the high-affinity site is occupied and normal kinetics are followed. However, at higher concentrations, the second inhibitory site becomes occupied, inhibiting the enzyme. Product inhibition is often a regulatory feature in metabolism and can be a form of negative feedback.
- Slow-tight inhibition occurs when the initial enzyme–inhibitor complex EI undergoes isomerisation to a second more tightly held complex, EI*, but the overall inhibition process is reversible. This manifests itself as slowly increasing enzyme inhibition. Under these conditions, traditional Michaelis–Menten kinetics give a false value for Ki, which is time–dependent. The true value of Ki can be obtained through more complex analysis of the on (kon) and off (koff) rate constants for inhibitor association. See irreversible inhibition below for more information.
- Bi-substrate analog inhibitors are high affinity and selectivity inhibitors that can be prepared for enzymes that catalyze bi-molecular reactions by capturing the binding energy of each substrate into one molecule. For example, in the formyl transfer reactions of purine biosynthesis, a potent multi-substrate adduct inhibitor (MAI) to GAR TFase was prepared synthetically by linking analogs of the glycinamide ribonucleotide (GAR) substrate and the N-10-formyl tetrahydrofolate cofactor together to produce thioglycinamide ribonucleotide dideazafolate (TGDDF), or enzymatically from the natural GAR substrate to yield GDDF. Here the subnanomolar dissociation constant (KD) of TGDDF was greater than predicted presumably due to entropic advantages gained and/or positive interactions acquired through the atoms linking the components. MAIs have also been observed to be produced in cells by reactions of pro-drugs such as isoniazid or enzyme inhibitor ligands (e.g., PTC124) with cellular cofactors such as NADH and ATP respectively.
Examples of reversible inhibitors
As
enzymes have evolved to bind their substrates tightly, and most
reversible inhibitors bind in the active site of enzymes, it is
unsurprising that some of these inhibitors are strikingly similar in
structure to the substrates of their targets. Inhibitors of DHFR are
prominent examples. Other example of these substrate mimics are the protease inhibitors, a very successful class of antiretroviral drugs used to treat HIV. The structure of ritonavir, a protease inhibitor based on a peptide and containing three peptide bonds,
is shown on the right. As this drug resembles the protein that is the
substrate of the HIV protease, it competes with this substrate in the
enzyme's active site.
Enzyme inhibitors are often designed to mimic the transition state
or intermediate of an enzyme-catalyzed reaction. This ensures that the
inhibitor exploits the transition state stabilising effect of the
enzyme, resulting in a better binding affinity (lower Ki) than substrate-based designs. An example of such a transition state inhibitor is the antiviral drug oseltamivir; this drug mimics the planar nature of the ring oxonium ion in the reaction of the viral enzyme neuraminidase.
However, not all inhibitors are based on the structures of
substrates. For example, the structure of another HIV protease inhibitor
tipranavir
is shown on the left. This molecule is not based on a peptide and has
no obvious structural similarity to a protein substrate. These
non-peptide inhibitors can be more stable than inhibitors containing
peptide bonds, because they will not be substrates for peptidases and are less likely to be degraded.
In drug design it is important to consider the concentrations of
substrates to which the target enzymes are exposed. For example, some protein kinase inhibitors have chemical structures that are similar to adenosine triphosphate,
one of the substrates of these enzymes. However, drugs that are simple
competitive inhibitors will have to compete with the high concentrations
of ATP in the cell. Protein kinases can also be inhibited by
competition at the binding sites where the kinases interact with their
substrate proteins, and most proteins are present inside cells at
concentrations much lower than the concentration of ATP. As a
consequence, if two protein kinase inhibitors both bind in the active
site with similar affinity, but only one has to compete with ATP, then
the competitive inhibitor at the protein-binding site will inhibit the
enzyme more effectively.
Irreversible inhibitors
Types of irreversible inhibition (covalent inactivation)
Reaction of the irreversible inhibitor diisopropylfluorophosphate (DFP) with a serine protease
Irreversible inhibitors usually covalently
modify an enzyme, and inhibition can therefore not be reversed.
Irreversible inhibitors often contain reactive functional groups such as
nitrogen mustards, aldehydes, haloalkanes, alkenes, Michael acceptors, phenyl sulfonates, or fluorophosphonates. These nucleophilic groups react with amino acid side chains to form covalent adducts. The residues modified are those with side chains containing nucleophiles such as hydroxyl or sulfhydryl groups; these include the amino acids serine (as in DFP, right), cysteine, threonine, or tyrosine.
Irreversible inhibition is different from irreversible enzyme
inactivation. Irreversible inhibitors are generally specific for one
class of enzyme and do not inactivate all proteins; they do not function
by destroying protein structure but by specifically altering the active site of their target. For example, extremes of pH or temperature usually cause denaturation of all protein structure,
but this is a non-specific effect. Similarly, some non-specific
chemical treatments destroy protein structure: for example, heating in
concentrated hydrochloric acid will hydrolyse the peptide bonds holding proteins together, releasing free amino acids.
Irreversible inhibitors display time-dependent inhibition and their potency therefore cannot be characterised by an IC50
value. This is because the amount of active enzyme at a given
concentration of irreversible inhibitor will be different depending on
how long the inhibitor is pre-incubated with the enzyme. Instead, kobs/[I] values are used, where kobs is the observed pseudo-first order rate of inactivation (obtained by plotting the log of % activity vs. time) and [I] is the concentration of inhibitor. The kobs/[I] parameter is valid as long as the inhibitor does not saturate binding with the enzyme (in which case kobs = kinact).
Analysis of irreversible inhibition
Kinetic scheme for irreversible inhibitors
As shown in the figure to the right, irreversible inhibitors have a
short instance where they form a reversible non-covalent complex with
the enzyme (EI or ESI) and this then reacts to produce the covalently
modified "dead-end complex" EI* (an irreversible covalent complex). The
rate at which EI* is formed is called the inactivation rate or kinact.
Since formation of EI may compete with ES, binding of irreversible
inhibitors can be prevented by competition either with substrate or with
a second, reversible inhibitor. This protection effect is good evidence
of a specific reaction of the irreversible inhibitor with the active
site.
The binding and inactivation steps of this reaction are
investigated by incubating the enzyme with inhibitor and assaying the
amount of activity remaining over time. The activity will be decreased
in a time-dependent manner, usually following exponential decay. Fitting these data to a rate equation
gives the rate of inactivation at this concentration of inhibitor. This
is done at several different concentrations of inhibitor. If a
reversible EI complex is involved the inactivation rate will be
saturable and fitting this curve will give kinact and Ki.
Another method that is widely used in these analyses is mass spectrometry.
Here, accurate measurement of the mass of the unmodified native enzyme
and the inactivated enzyme gives the increase in mass caused by reaction
with the inhibitor and shows the stoichiometry of the reaction. This is usually done using a MALDI-TOF mass spectrometer. In a complementary technique, peptide mass fingerprinting involves digestion of the native and modified protein with a protease such as trypsin. This will produce a set of peptides
that can be analysed using a mass spectrometer. The peptide that
changes in mass after reaction with the inhibitor will be the one that
contains the site of modification.
Special cases
Chemical
mechanism for irreversible inhibition of ornithine decarboxylase by
DFMO. Pyridoxal 5'-phosphate (Py) and enzyme (E) are not shown.
Not all irreversible inhibitors form covalent adducts with their
enzyme targets. Some reversible inhibitors bind so tightly to their
target enzyme that they are essentially irreversible. These
tight-binding inhibitors may show kinetics similar to covalent
irreversible inhibitors. In these cases, some of these inhibitors
rapidly bind to the enzyme in a low-affinity EI complex and this then
undergoes a slower rearrangement to a very tightly bound EI* complex
(see figure above). This kinetic behaviour is called slow-binding. This slow rearrangement after binding often involves a conformational change
as the enzyme "clamps down" around the inhibitor molecule. Examples of
slow-binding inhibitors include some important drugs, such methotrexate, allopurinol, and the activated form of acyclovir.
Examples of irreversible inhibitors
Trypanothione reductase with the lower molecule of an inhibitor bound irreversibly and the upper one reversibly. Created from PDB 1GXF.
Diisopropylfluorophosphate (DFP) is shown as an example of an irreversible protease inhibitor in the figure above right. The enzyme hydrolyses the phosphorus–fluorine bond, but the phosphate residue remains bound to the serine in the active site, deactivating it. Similarly, DFP also reacts with the active site of acetylcholine esterase in the synapses of neurons, and consequently is a potent neurotoxin, with a lethal dose of less than 100 mg.
Suicide inhibition
is an unusual type of irreversible inhibition where the enzyme converts
the inhibitor into a reactive form in its active site. An example is
the inhibitor of polyamine biosynthesis, α-difluoromethylornithine or DFMO, which is an analogue of the amino acid ornithine, and is used to treat African trypanosomiasis (sleeping sickness). Ornithine decarboxylase
can catalyse the decarboxylation of DFMO instead of ornithine, as shown
above. However, this decarboxylation reaction is followed by the
elimination of a fluorine atom, which converts this catalytic
intermediate into a conjugated imine,
a highly electrophilic species. This reactive form of DFMO then reacts
with either a cysteine or lysine residue in the active site to
irreversibly inactivate the enzyme.
Since irreversible inhibition often involves the initial
formation of a non-covalent EI complex, it is sometimes possible for an
inhibitor to bind to an enzyme in more than one way. For example, in the
figure showing trypanothione reductase from the human protozoan parasite Trypanosoma cruzi, two molecules of an inhibitor called quinacrine mustard
are bound in its active site. The top molecule is bound reversibly, but
the lower one is bound covalently as it has reacted with an amino acid
residue through its nitrogen mustard group.
Discovery and design of inhibitors
Robots used for the high-throughput screening of chemical libraries to discover new enzyme inhibitors
New drugs are the products of a long drug development
process, the first step of which is often the discovery of a new enzyme
inhibitor. In the past the only way to discover these new inhibitors
was by trial and error: screening huge libraries of compounds against a
target enzyme and hoping that some useful leads would emerge. This brute
force approach is still successful and has even been extended by combinatorial chemistry approaches that quickly produce large numbers of novel compounds and high-throughput screening technology to rapidly screen these huge chemical libraries for useful inhibitors.
More recently, an alternative approach has been applied: rational drug design uses the three-dimensional structure of an enzyme's active site to predict which molecules might be inhibitors.
These predictions are then tested and one of these tested compounds may
be a novel inhibitor. This new inhibitor is then used to try to obtain a
structure of the enzyme in an inhibitor/enzyme complex to show how the
molecule is binding to the active site, allowing changes to be made to
the inhibitor to try to optimise binding. This test and improve cycle is
then repeated until a sufficiently potent inhibitor is produced. Computer-based methods of predicting the affinity of an inhibitor for an enzyme are also being developed, such as molecular docking and molecular mechanics.
Uses of inhibitors
Enzyme inhibitors are found in nature and are also designed and produced as part of pharmacology and biochemistry. Natural poisons are often enzyme inhibitors that have evolved to defend a plant or animal against predators.
These natural toxins include some of the most poisonous compounds
known. Artificial inhibitors are often used as drugs, but can also be insecticides such as malathion, herbicides such as glyphosate, or disinfectants such as triclosan. Other artificial enzyme inhibitors block acetylcholinesterase, an enzyme which breaks down acetylcholine, and are used as nerve agents in chemical warfare.
Chemotherapy
The structure of sildenafil (Viagra)
|
The coenzyme folic acid (left) compared to the anti-cancer drug methotrexate (right)
|
The structure of a complex between penicillin G and the Streptomyces transpeptidase. Generated from PDB 1PWC.
|
The most common uses for enzyme inhibitors are as drugs to treat
disease. Many of these inhibitors target a human enzyme and aim to
correct a pathological condition. However, not all drugs are enzyme
inhibitors. Some, such as anti-epileptic drugs, alter enzyme activity by causing more or less of the enzyme to be produced. These effects are called enzyme induction and inhibition and are alterations in gene expression,
which is unrelated to the type of enzyme inhibition discussed here.
Other drugs interact with cellular targets that are not enzymes, such as
ion channels or membrane receptors.
An example of a medicinal enzyme inhibitor is sildenafil (Viagra), a common treatment for male erectile dysfunction. This compound is a potent inhibitor of cGMP specific phosphodiesterase type 5, the enzyme that degrades the signalling molecule cyclic guanosine monophosphate. This signalling molecule triggers smooth muscle relaxation and allows blood flow into the corpus cavernosum,
which causes an erection. Since the drug decreases the activity of the
enzyme that halts the signal, it makes this signal last for a longer
period of time.
Another example of the structural similarity of some inhibitors
to the substrates of the enzymes they target is seen in the figure
comparing the drug methotrexate to folic acid. Folic acid is a substrate of dihydrofolate reductase, an enzyme involved in making nucleotides
that is potently inhibited by methotrexate. Methotrexate blocks the
action of dihydrofolate reductase and thereby halts the production of
nucleotides. This block of nucleotide biosynthesis is more toxic to
rapidly growing cells than non-dividing cells, since a rapidly growing
cell has to carry out DNA replication, therefore methotrexate is often used in cancer chemotherapy.
Antibiotics
Drugs also are used to inhibit enzymes needed for the survival of pathogens. For example, bacteria are surrounded by a thick cell wall made of a net-like polymer called peptidoglycan. Many antibiotics such as penicillin and vancomycin inhibit the enzymes that produce and then cross-link the strands of this polymer together.
This causes the cell wall to lose strength and the bacteria to burst.
In the figure, a molecule of penicillin (shown in a ball-and-stick form)
is shown bound to its target, the transpeptidase from the bacteria Streptomyces R61 (the protein is shown as a ribbon-diagram).
Antibiotic drug design
is facilitated when an enzyme that is essential to the pathogen's
survival is absent or very different in humans. In the example above,
humans do not make peptidoglycan, therefore inhibitors of this process
are selectively toxic to bacteria. Selective toxicity is also produced
in antibiotics by exploiting differences in the structure of the ribosomes in bacteria, or how they make fatty acids.
Metabolic control
Enzyme inhibitors are also important in metabolic control. Many metabolic pathways in the cell are inhibited by metabolites that control enzyme activity through allosteric regulation or substrate inhibition. A good example is the allosteric regulation of the glycolytic pathway. This catabolic pathway consumes glucose and produces ATP, NADH and pyruvate. A key step for the regulation of glycolysis is an early reaction in the pathway catalysed by phosphofructokinase-1
(PFK1). When ATP levels rise, ATP binds an allosteric site in PFK1 to
decrease the rate of the enzyme reaction; glycolysis is inhibited and
ATP production falls. This negative feedback
control helps maintain a steady concentration of ATP in the cell.
However, metabolic pathways are not just regulated through inhibition
since enzyme activation is equally important. With respect to PFK1, fructose 2,6-bisphosphate and ADP are examples of metabolites that are allosteric activators.
Physiological enzyme inhibition can also be produced by specific protein inhibitors. This mechanism occurs in the pancreas, which synthesises many digestive precursor enzymes known as zymogens. Many of these are activated by the trypsin
protease, so it is important to inhibit the activity of trypsin in the
pancreas to prevent the organ from digesting itself. One way in which
the activity of trypsin is controlled is the production of a specific
and potent trypsin inhibitor
protein in the pancreas. This inhibitor binds tightly to trypsin,
preventing the trypsin activity that would otherwise be detrimental to
the organ.
Although the trypsin inhibitor is a protein, it avoids being hydrolysed
as a substrate by the protease by excluding water from trypsin's active
site and destabilising the transition state. Other examples of physiological enzyme inhibitor proteins include the barstar inhibitor of the bacterial ribonuclease barnase.
Pesticides
Many pesticides are enzyme inhibitors. Acetylcholinesterase
(AChE) is an enzyme found in animals, from insects to humans. It is
essential to nerve cell function through its mechanism of breaking down
the neurotransmitter acetylcholine into its constituents, acetate and choline. This is somewhat unusual among neurotransmitters as most, including serotonin, dopamine, and norepinephrine, are absorbed from the synaptic cleft
rather than cleaved. A large number of AChE inhibitors are used in
both medicine and agriculture. Reversible competitive inhibitors, such
as edrophonium, physostigmine, and neostigmine, are used in the treatment of myasthenia gravis and in anaesthesia. The carbamate pesticides are also examples of reversible AChE inhibitors. The organophosphate pesticides such as malathion, parathion, and chlorpyrifos irreversibly inhibit acetylcholinesterase.
The herbicide glyphosate is an inhibitor of 3-phosphoshikimate 1-carboxyvinyltransferase, other herbicides, such as the sulfonylureas inhibit the enzyme acetolactate synthase. Both these enzymes are needed for plants to make branched-chain amino acids. Many other enzymes are inhibited by herbicides, including enzymes needed for the biosynthesis of lipids and carotenoids and the processes of photosynthesis and oxidative phosphorylation.
Natural poisons
Animals and plants have evolved to synthesise a vast array of poisonous products including secondary metabolites, peptides and proteins that can act as inhibitors. Natural toxins are usually small organic molecules and are so diverse that there are probably natural inhibitors for most metabolic processes.
The metabolic processes targeted by natural poisons encompass more than
enzymes in metabolic pathways and can also include the inhibition of
receptor, channel and structural protein functions in a cell. For
example, paclitaxel (taxol), an organic molecule found in the Pacific yew tree, binds tightly to tubulin dimers and inhibits their assembly into microtubules in the cytoskeleton.
Many natural poisons act as neurotoxins that can cause paralysis
leading to death and have functions for defence against predators or in
hunting and capturing prey. Some of these natural inhibitors, despite
their toxic attributes, are valuable for therapeutic uses at lower
doses. An example of a neurotoxin are the glycoalkaloids, from the plant species in the family Solanaceae (includes potato, tomato and eggplant), that are acetylcholinesterase
inhibitors. Inhibition of this enzyme causes an uncontrolled increase
in the acetylcholine neurotransmitter, muscular paralysis and then
death. Neurotoxicity can also result from the inhibition of receptors;
for example, atropine from deadly nightshade (Atropa belladonna) that functions as a competitive antagonist of the muscarinic acetylcholine receptors.
Although many natural toxins are secondary metabolites, these
poisons also include peptides and proteins. An example of a toxic
peptide is alpha-amanitin, which is found in relatives of the death cap mushroom. This is a potent enzyme inhibitor, in this case preventing the RNA polymerase II enzyme from transcribing DNA. The algal toxin microcystin is also a peptide and is an inhibitor of protein phosphatases. This toxin can contaminate water supplies after algal blooms and is a known carcinogen that can also cause acute liver hemorrhage and death at higher doses.
Proteins can also be natural poisons or antinutrients, such as the trypsin inhibitors (discussed above) that are found in some legumes,
as shown in the figure above. A less common class of toxins are toxic
enzymes: these act as irreversible inhibitors of their target enzymes
and work by chemically modifying their substrate enzymes. An example is ricin, an extremely potent protein toxin found in castor oil beans. This enzyme is a glycosidase
that inactivates ribosomes. Since ricin is a catalytic irreversible
inhibitor, this allows just a single molecule of ricin to kill a cell.



![V={\frac {V_{max}[S]}{\alpha K_{m}+\alpha ^{\prime }[S]}}={\frac {(1/\alpha ^{\prime })V_{max}[S]}{(\alpha /\alpha ^{\prime })K_{m}+[S]}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/4a8f0a9dda1d308de7f090f99c2833f944f11a09)
![\alpha =1+{\frac {[I]}{K_{i}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/57fcf54938a9784f9313437681b220079ff43ee5)
![\alpha ^{\prime }=1+{\frac {[I]}{K_{i}^{\prime }}}.](https://wikimedia.org/api/rest_v1/media/math/render/svg/65bf16742482cae7b0743781f47c327ddcf537e3)
![{\displaystyle {\begin{aligned}{\cfrac {V_{\max }}{1+{\cfrac {\ce {[I]}}{K_{i}}}}}&={V_{\max }}\left({\cfrac {K_{i}}{K_{i}+[{\ce {I}}]}}\right)&&{\text{multiply by }}{\cfrac {K_{i}}{K_{i}}}=1\\&={V_{\max }}\left({\cfrac {K_{i}+[{\ce {I}}]-[{\ce {I}}]}{K_{i}+[{\ce {I}}]}}\right)&&{\text{add }}[{\ce {I}}]-[{\ce {I}}]=0{\text{ to numerator}}\\&={V_{\max }}\left(1-{\cfrac {[{\ce {I}}]}{K_{i}+[{\ce {I}}]}}\right)&&{\text{simplify }}{\cfrac {K_{i}+[{\ce {I}}]}{K_{i}+[{\ce {I}}]}}=1\\&=V_{\max }-V_{\max }{\cfrac {\ce {[I]}}{K_{i}+[{\ce {I}}]}}&&{\text{multiply out by }}V_{\max }\end{aligned}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/37eda4dec307f8acfca89b2d8f4811474ea764ec)
![{\displaystyle {\cfrac {\ce {[S]}}{[{\ce {S}}]+K_{m}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/eb08dd139085a394e6e7370f47ebfa255f1ad685)
![{\displaystyle {\cfrac {\ce {[I]}}{[{\ce {I}}]+K_{i}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/9ed50a1f7a5f2c52f406b52263916ab48b268e07)
![{\displaystyle V_{\max }-\Delta V_{\max }{\cfrac {\ce {[I]}}{[{\ce {I}}]+K_{i}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/7dff424ec79284c3a1cea14f0f82b0eaace53c69)
![{\displaystyle V_{\max 1}-(V_{\max 1}-V_{\max 2}){\cfrac {\ce {[I]}}{[{\ce {I}}]+K_{i}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/f3874623edd9524ba2741fe448927bf5cf0ab257)
![{\displaystyle V_{\max 1}-(V_{\max 1}-V_{\max 2}){\cfrac {\ce {[X]}}{[{\ce {X}}]+K_{x}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/306d44733a89308883053e3b8372a8cf9ce0239b)
![{\displaystyle K_{m1}-(K_{m1}-K_{m2}){\cfrac {\ce {[X]}}{[{\ce {X}}]+K_{x}}}}](https://wikimedia.org/api/rest_v1/media/math/render/svg/cb4e0de216e1e625bb803ee725bf85c9989a15f5)