| Orthomyxovirus | |
|---|---|
| Virus classification | |
| (unranked): | Virus |
| Realm: | Riboviria |
| Phylum: | Negarnaviricota |
| Class: | Insthoviricetes |
| Order: | Articulavirales |
| Family: | Orthomyxoviridae |
| Genera | |
Orthomyxoviridae (ὀρθός, orthós, Greek for "straight"; μύξα, mýxa, Greek for "mucus") is a family of RNA viruses. It includes seven genera: Influenzavirus A, Influenzavirus B, Influenzavirus C, Influenzavirus D, Isavirus, Thogotovirus, and Quaranjavirus. The first four genera contain viruses that cause influenza in vertebrates, including birds, humans, and other mammals. Isaviruses infect salmon; the thogotoviruses are arboviruses, infecting vertebrates and invertebrates, such as ticks and mosquitoes.
The four genera of Influenza virus that infect vertebrates, which are identified by antigenic differences in their nucleoprotein and matrix protein, are as follows:
- Influenzavirus A infects humans, other mammals, and birds, and causes all flu pandemics
- Influenzavirus B infects humans and seals
- Influenzavirus C infects humans, pigs, and dogs.
- Influenzavirus D infects pigs and cattle
Classification
Influenza virus
In a phylogenetic-based taxonomy, the category "RNA virus" includes the category "negative-sense ssRNA virus", which includes the Order "Mononegavirales", and the Family "Orthomyxovirus" (among others). The genera-associated species and serotypes of Orthomyxovirus are shown in the following table.
| Genus | Species (* indicates type species) | Serotypes or Subtypes | Hosts |
|---|---|---|---|
| Influenza virus A | Influenza A virus* | H1N1, H1N2, H2N2, H3N1, H3N2, H3N8, H5N1, H5N2, H5N3, H5N8, H5N9, H7N1, H7N2, H7N3, H7N4, H7N7, H7N9, H9N2, H10N7 | Human, pig, bird, horse, bat |
| Influenza virus B | Influenza B virus* | Victoria, Yamagata[5] | Human, seal |
| Influenza virus C | Influenza C virus* |
|
Human, pig, dog |
| Influenza virus D | Influenza D virus* |
|
Pig, cattle |
| Isavirus | Infectious salmon anemia virus* |
|
Atlantic salmon |
| Thogotovirus | Thogotovirus* |
|
Tick, mosquito, mammal (including human) |
| Dhori virus | Batken virus, Bourbon virus, Jos virus | ||
| Quaranjavirus[6] | |||
| Quaranfil virus,* Johnston Atoll virus |
|
|
Types
There
are four genera of influenza virus, each containing only a single
species, or type. Influenza A and C infect a variety of species, while
influenza B almost exclusively infects humans, and influenza D infects
cattle and pigs.
Influenza A
Influenza A viruses are further classified, based on the viral surface proteins hemagglutinin (HA or H) and neuraminidase (NA or N). Sixteen H subtypes (or serotypes) and nine N subtypes of influenza A virus have been identified.
Diagram of influenza nomenclature
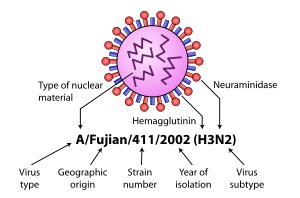
Further variation exists; thus, specific influenza strain isolates
are identified by a standard nomenclature specifying virus type,
geographical location where first isolated, sequential number of
isolation, year of isolation, and HA and NA subtype.
Examples of the nomenclature are:
- A/Brisbane/59/2007 (H1N1)
- A/Moscow/10/99 (H3N2).
The type A viruses are the most virulent human pathogens among the
three influenza types and cause the most severe disease. The serotypes
that have been confirmed in humans, ordered by the number of known human pandemic deaths, are:
- H1N1 caused "Spanish flu" in 1918 and "Swine flu" in 2009.
- H2N2 caused "Asian Flu".
- H3N2 caused "Hong Kong Flu".
- H5N1, "avian" or "bird flu".
- H7N7 has unusual zoonotic potential.
- H1N2 is endemic in humans and pigs.
- H9N2, H7N2, H7N3, H10N7.
| Name of pandemic | Date | Deaths | Case fatality rate | Subtype involved | Pandemic Severity Index |
|---|---|---|---|---|---|
| 1889–1890 flu pandemic (Asiatic or Russian Flu) |
1889–1890 | 1 million | 0.15% | possibly H3N8 or H2N2 |
NA |
| 1918 flu pandemic (Spanish flu) |
1918–1920 | 20 to 100 million | 2% | H1N1 | 5 |
| Asian Flu | 1957–1958 | 1 to 1.5 million | 0.13% | H2N2 | 2 |
| Hong Kong Flu | 1968–1969 | 0.75 to 1 million | <0 .1="" font=""> | H3N2 | 2 |
| Russian flu | 1977–1978 | no accurate count | N/A | H1N1 | N/A |
| 2009 flu pandemic | 2009–2010 | 105,700–395,600 | 0.03% | H1N1 | NA |
Influenza B
Influenza B virus is almost exclusively a human pathogen, and is less
common than influenza A. The only other animal known to be susceptible
to influenza B infection is the seal. This type of influenza mutates at a rate 2–3 times lower than type A and consequently is less genetically diverse, with only one influenza B serotype. As a result of this lack of antigenic
diversity, a degree of immunity to influenza B is usually acquired at
an early age. However, influenza B mutates enough that lasting immunity
is not possible. This reduced rate of antigenic change, combined with its limited host range (inhibiting cross species antigenic shift), ensures that pandemics of influenza B do not occur.
Influenza C
The influenza C virus infects humans and pigs, and can cause severe illness and local epidemics. However, influenza C is less common than the other types and usually causes mild disease in children.
Influenza D
This is a genus that was classified in 2016, the members of which were first isolated in 2011. This genus appears to be most closely related to Influenza C, from which it diverged several hundred years ago. There are at least two strains of this genus in extant. The main hosts appear to be cattle, but this virus has seen to infect pigs as well.
Morphology
Structure of the influenza virion. The hemagglutinin (HA) and neuraminidase
(NA) proteins are shown on the surface of the particle. The viral RNAs
that make up the genome are shown as red coils inside the particle and
bound to Ribonuclear Proteins (RNPs).
The virion
is pleomorphic; the envelope can occur in spherical and filamentous
forms. In general, the virus's morphology is ellipsoidal with particles
80 to 120 nm in diameter, or filamentous virions 80–120 nm in diameter and up to 20 µm long.
There are some 500 distinct spike-like surface projections of the
envelope each projecting 10 to 14 nm from the surface with varying
surface densities.
The major glycoprotein (HA) is interposed irregularly by clusters of neuraminidase (NA), with a ratio of HA to NA of about 4–5 to 1.
Cholesterol-laden membranes with protruding glycoproteins enclose the nucleocapsids;
nucleoproteins of different size classes with a loop at each end; the
arrangement within the virion is uncertain. The ribonuclear proteins are
filamentous and fall in the range of 50 to 130 nm long and 9 to 15 nm
in diameter. They have a helical symmetry.
Genome
Viruses of this family contain 6 to 8 segments of linear negative-sense single stranded RNA.
The total genome length is 12000–15000 nucleotides (nt). The size of each segment is as follows:
| segment | protein | size (nt) | protein size (aa) |
|---|---|---|---|
| PB1 | polymerase | 2300–2500 | 757+87 (F2) |
| PB2 | polymerase | 2300–2500 | 759 |
| PA | polymerase | 2200–2300 | 716 |
| HA | Hemagglutinin | 1700–1800 | 550 |
| NP | nucleoprotein | 1500–1600 | 498 |
| NA | Neuraminidase | 1400–1500 | 454 |
| M | Membrane protein(s) | 1000–1100 | 252+97 |
| NS | non-structural protein(s) | 800–900 | 230+121 |
The Genome sequence has terminal repeated sequences; repeated at both
ends. Terminal repeats at the 5'-end 12–13 nucleotides long. Nucleotide
sequences of 3'-terminus identical; the same in genera of same family;
most on RNA (segments), or on all RNA species. Terminal repeats at the
3'-end 9–11 nucleotides long. Encapsidated nucleic acid is solely
genomic. Each virion may contain defective interfering copies. In
Influenza A (H1N1) PB1-F2 is produced from an alternative reading frame in PB1. The M and NS genes produce 2 different genes via alternative splicing.
Structure
The following applies for Influenza A viruses, although other influenza strains are very similar in structure:
The influenza A virus particle or virion is 80–120 nm in diameter, usually producing both ellipsoidal, baciliform, and filamentous particles. Unusually for a virus, the influenza A genome is not a single piece of nucleic acid; instead, it contains eight pieces of segmented negative-sense RNA (13.5 kilobases total), which encode 11 proteins (HA, NA, NP, M1, M2, NS1, NEP, PA, PB1, PB1-F2, PB2). The best-characterised of these viral proteins are hemagglutinin and neuraminidase, two large glycoproteins found on the outside of the viral particles. Neuraminidase is an enzyme involved in the release of progeny virus from infected cells, by cleaving sugars that bind the mature viral particles. By contrast, hemagglutinin is a lectin that mediates binding of the virus to target cells and entry of the viral genome into the target cell. The hemagglutinin (H) and neuraminidase (N) proteins are targets for antiviral drugs. These proteins are also recognised by antibodies, i.e. they are antigens. The responses of antibodies to these proteins are used to classify the different serotypes of influenza A viruses, hence the H and N in H5N1.
Replication cycle
Invasion and replication of the influenza virus. The steps in this process are discussed in the text.
Typically, influenza is transmitted from infected mammals through the air by coughs or sneezes, creating aerosols containing the virus, and from infected birds through their droppings. Influenza can also be transmitted by saliva, nasal secretions, feces and blood.
Infections occur through contact with these bodily fluids or with
contaminated surfaces. Out of a host, flu viruses can remain infectious
for about one week at human body temperature, over 30 days at 0 °C
(32 °F), and indefinitely at very low temperatures (such as lakes in
northeast Siberia). They can be inactivated easily by disinfectants and detergents.
The viruses bind to a cell through interactions between its hemagglutinin glycoprotein and sialic acid sugars on the surfaces of epithelial cells in the lung and throat (Stage 1 in infection figure). The cell imports the virus by endocytosis. In the acidic endosome,
part of the haemagglutinin protein fuses the viral envelope with the
vacuole's membrane, releasing the viral RNA (vRNA) molecules, accessory
proteins and RNA-dependent RNA polymerase into the cytoplasm (Stage 2). These proteins and vRNA form a complex that is transported into the cell nucleus, where the RNA-dependent RNA polymerase begins transcribing complementary positive-sense cRNA (Steps 3a and b).
The cRNA is either exported into the cytoplasm and translated (step 4),
or remains in the nucleus. Newly synthesised viral proteins are either
secreted through the Golgi apparatus
onto the cell surface (in the case of neuraminidase and hemagglutinin,
step 5b) or transported back into the nucleus to bind vRNA and form new
viral genome particles (step 5a). Other viral proteins have multiple
actions in the host cell, including degrading cellular mRNA and using the released nucleotides for vRNA synthesis and also inhibiting translation of host-cell mRNAs.
Negative-sense vRNAs that form the genomes
of future viruses, RNA-dependent RNA transcriptase, and other viral
proteins are assembled into a virion. Hemagglutinin and neuraminidase
molecules cluster into a bulge in the cell membrane. The vRNA and viral
core proteins leave the nucleus and enter this membrane protrusion (step
6). The mature virus buds off from the cell in a sphere of host
phospholipid membrane, acquiring hemagglutinin and neuraminidase with
this membrane coat (step 7). As before, the viruses adhere to the cell through hemagglutinin; the mature viruses detach once their neuraminidase has cleaved sialic acid residues from the host cell. After the release of new influenza virus, the host cell dies.
Orthomyxoviridae viruses are one of two RNA viruses that replicate in the nucleus (the other being retroviridae).
This is because the machinery of orthomyxo viruses cannot make their
own mRNAs. They use cellular RNAs as primers for initiating the viral
mRNA synthesis in a process known as cap snatching.
Once in the nucleus, the RNA Polymerase Protein PB2 finds a cellular
pre-mRNA and binds to its 5' capped end. Then RNA Polymerase PA cleaves
off the cellular mRNA near the 5' end and uses this capped fragment as a
primer for transcribing the rest of the viral RNA genome in viral mRNA. This is due to the need of mRNA to have a 5' cap in order to be recognized by the cell's ribosome for translation.
Since RNA proofreading
enzymes are absent, the RNA-dependent RNA transcriptase makes a single
nucleotide insertion error roughly every 10 thousand nucleotides, which
is the approximate length of the influenza vRNA. Hence, nearly every
newly manufactured influenza virus will contain a mutation in its
genome. The separation of the genome into eight separate segments of vRNA allows mixing (reassortment) of the genes if more than one variety of influenza virus has infected the same cell (superinfection).
The resulting alteration in the genome segments packaged into viral
progeny confers new behavior, sometimes the ability to infect new host
species or to overcome protective immunity of host populations to its
old genome (in which case it is called an antigenic shift).
Viability and disinfection
Mammalian influenza viruses tend to be labile, but can survive several hours in mucus.
Avian influenza virus can survive for 100 days in distilled water at
room temperature, and 200 days at 17 °C (63 °F). The avian virus is
inactivated more quickly in manure, but can survive for up to 2 weeks in
feces on cages. Avian influenza viruses can survive indefinitely when
frozen.
Influenza viruses are susceptible to bleach, 70% ethanol, aldehydes,
oxidizing agents, and quaternary ammonium compounds. They are
inactivated by heat of 133 °F (56 °C) for minimum of 60 minutes, as well
as by low pH <2 .="" p="">
Vaccination and prophylaxis
Vaccines
and drugs are available for the prophylaxis and treatment of influenza
virus infections. Vaccines are composed of either inactivated or live
attenuated virions of the H1N1 and H3N2 human influenza A viruses, as
well as those of influenza B viruses. Because the antigenicities of the
wild viruses evolve, vaccines are reformulated annually by updating the
seed strains.
When the antigenicities of the seed strains and wild viruses do
not match, vaccines fail to protect the vaccinees. In addition, even
when they do match, escape mutants are often generated.
Drugs available for the treatment of influenza include Amantadine and Rimantadine, which inhibit the uncoating of virions by interfering with M2, and Oseltamivir (marketed under the brand name Tamiflu), Zanamivir, and Peramivir,
which inhibit the release of virions from infected cells by interfering
with NA. However, escape mutants are often generated for the former
drug and less frequently for the latter drug.