|
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
2-Chlorobuta-1,3-diene
| |||
| Other names
Chloroprene, 2-chloro-1,3-butadiene, Chlorobutadiene, β-Chloroprene
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.004.381 | ||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
CompTox Dashboard (EPA)
|
|||
| Properties | |||
| C4H5Cl | |||
| Molar mass | 88.5365 g/mol | ||
| Appearance | Colorless liquid | ||
| Odor | pungent, ether-like | ||
| Density | 0.9598 g/cm3 | ||
| Melting point | −130 °C (−202 °F; 143 K) | ||
| Boiling point | 59.4 °C (138.9 °F; 332.5 K) | ||
| 0.026 g/100 mL | |||
| Solubility | soluble in alcohol, diethyl ether miscible in ethyl ether, acetone, benzene | ||
| Vapor pressure | 188 mmHg (20 °C) | ||
Refractive index (nD)
|
1.4583 | ||
| Hazards | |||
| Main hazards | Highly flammable, irritant, toxic. | ||
| R-phrases (outdated) | R45, R11, R20/22, R36/37/38, R48/20 | ||
| S-phrases (outdated) | S53, S45 | ||
| NFPA 704 (fire diamond) | |||
| Flash point | −15.6 °C (3.9 °F; 257.5 K) | ||
| Explosive limits | 1.9%–11.3% | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
450 mg/kg (rat, oral) | ||
LC50 (median concentration)
|
3207 ppm (rat, 4 hr) | ||
LCLo (lowest published)
|
1052 ppm (rabbit, 8 hr) 350 ppm (cat, 8 hr) | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible)
|
TWA 25 ppm (90 mg/m3) [skin] | ||
REL (Recommended)
|
Ca C 1 ppm (3.6 mg/m3) [15-minute] | ||
IDLH (Immediate danger)
|
300 ppm | ||
| Related compounds | |||
Related Dienes
|
Butadiene Isoprene | ||
Related compounds
|
Vinyl chloride | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
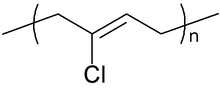
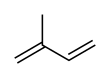
Chloroprene is the common name for 2-chlorobuta-1,3-diene (IUPAC name) with the chemical formula CH2=CCl−CH=CH2. Chloroprene is a colorless volatile liquid, almost exclusively used as a monomer for the production of the polymer polychloroprene, a type of synthetic rubber. Polychloroprene is better known as Neoprene, the trade name given by DuPont.
History
Although
it may have been discovered earlier, the chemistry of chloroprene was
largely developed by DuPont during the early 1930s, specifically with
the formation of neoprene in mind. The chemists Elmer K. Bolton, Wallace Carothers, Arnold Collins and Ira Williams are generally accredited with its development and commercialisation although the work was based upon that of Julius Arthur Nieuwland, with whom they collaborated.
Production
Chloroprene is produced in three steps from 1,3-butadiene: (i) chlorination, (ii) isomerization of part of the product stream, and (iii) dehydrochlorination of 3,4-dichlorobut-1-ene.
Chlorine
adds to 1,3-butadiene to afford a mixture of 3,4-dichlorobut-1-ene and
1,4-dichlorobut-2-ene. The 1,4-dichloro isomer is subsequently
isomerized to 3,4 isomer, which in turn is treated with base to induce
dehydrochlorination to 2-chlorobuta-1,3-diene. This dehydrohalogenation
entails loss of a hydrogen atom
in the 3 position and the chlorine atom in the 4 position thereby
forming a double bond between carbons 3 and 4. In 1983, approximately
2,000,000 kg was produced in this manner. The chief impurity in chloroprene prepared in this way is
1-chlorobuta-1,3-diene, which is usually separated by distillation.
Acetylene process
Until
the 1960s, chloroprene production was dominated by the "acetylene
process," which was modeled after the original synthesis of vinylacetylene. In this process, acetylene is dimerized to give vinyl acetylene, which is then combined with hydrogen chloride to afford 4-chloro-1,2-butadiene (an allene derivative), which in the presence of copper(I) chloride, rearranges to the targeted 2-chlorobuta-1,3-diene.
This process is energy-intensive and has high investment costs.
Furthermore, the intermediate vinyl acetylene is unstable. This
"acetylene process" has been replaced by a process, which adds Cl2 to one of the double bonds in 1,3-butadiene, and subsequent elimination produces HCl instead, as well as chloroprene.
Regulations
Transportation
Transportation of uninhibited chloroprene has been banned in the United States by the US Department of Transportation. Stabilized chloroprene is in hazard class 3 (flammable liquid). Its UN number is 1991 and is in packing group 1.
Occupational health and safety
Hazards
GHS hazard pictograms
that apply to chloroprene. From left: flammability; carcinogenicity,
mutagenicity, reproductive toxicity, respiratory sensitization, target
organ toxicity, or aspiration toxicity; irritant (skin and eye), skin
sensitizer, respiratory tract irritant, hazardous to ozone layer, may
have narcotic effects; aquatic toxicity; and acute toxicity (fatal or
toxic).
As a way to visually communicate hazards associated with chloroprene exposure, the United Nations Globally Harmonized System of Classification and Labeling of Chemicals
(GHS) has designated the following hazards for exposure to chloroprene:
flammable, toxic, dangerous to the environment, health hazard and
irritant. Chloroprene poses fire hazard (flash point -4 °F).
OSHA identifies chloroprene as a category 2 flammable liquid and
emphasizes that at least one portable fire extinguisher should be within
10 and no more than 25 feet away from the flammable liquid storage
area.
OSHA provides resources on addressing flammable liquids at industrial
plants which is where the likely exposure to chloroprene exists (see
external resources). As a vapor, chloroprene is heavier than air.
According to the National Fire Protection Association's
rating system, chloroprene is designated with a category 2 health
hazard (temporary incapacitation or residual injury), a category 3 fire
hazard (ignition under the presence of moderate heat), and a category 1
reactivity (unstable at high temperatures and pressures).
Chronic exposure to chloroprene may have the following symptoms:
liver function abnormalities, disorders of the cardiovascular system,
and depression of the immune system.
The Environmental Protection Agency(EPA)
designated chloroprene as likely to be carcinogenic to humans based on
evidence from studies that showed a statistically significant
association between occupational chloroprene exposure and the risk of
lung cancer.
As early as 1975, NIOSH had identified the potential health hazards of
chloroprene in their bulletin primarily citing two Russian cohort
studies from those working with chloroprene in an occupational setting.
Hazard determination
OSHA
defines hazard determination as "the process of evaluating available
scientific evidence in order to determine if a chemical is hazardous
pursuant to the HCS." While chemical manufacturers and importers are
required to conduct a hazard determination, other companies may
voluntarily conduct a hazard determination to ensure worker health and
safety. Under the hazard determination framework, any chemical that has a
physical or health hazard is considered a hazardous chemical. Physical
hazards include fire hazards, reactive hazards, and explosion hazards.
Heath hazards include systemic effects and target organ effects.
Chloroprene is on OSHA's list for substances that are regulated as toxic
and hazardous.
In the European Union, the hazard-determination-equivalent is the
Registration, Evaluation, Authorization, and Restriction of Chemicals
(REACH) regulation enacted on June 1, 2007 by the European Chemicals
Agency (ECHA). The goal of REACH is to "improve the protection of human
health and the environment from the risks that can be posed by
chemicals, while enhancing the competitiveness of the EU chemicals
industry." If risks of chemicals are unmanageable, ECHA may ban its use.
Hazard controls
Several epidemiological studies and toxicological reports provide
evidence of chloroprene's capability to inflict occupational health and
safety concerns. However, varying reviews of the degree to which
chloroprene should be held responsible for health concerns highlight the
criticality of sound scientific research.
Nonetheless, health and safety practices should always be implemented
in the workplace. Some of these occupational concerns include: cleaning
equipment or unclogging pipes coated with chloroprene, inhaling
chloroprene off-gas, chloroprene spontaneously reacting with other
chemicals and chloroprene inducing a workplace fire. Upon the clogging
of equipment associated with occupational chloroprene use, employers
should ensure that their employees are wearing the proper PPE and set up
administrative controls so that skin exposure to and inhalation of
chloroprene is avoided.
Only one fatality as a result of chloroprene intoxication has been
recorded which was a result of cleaning a container used for
chloroprene.
NIOSH Hierarchy of Controls
The primary occupational concern for chloroprene is limited to the
facilities producing chloroprene and using chloroprene to produce the
synthetic rubber, polychloroprene.
NIOSH developed a list of actions to address specific workplace
hazards. These actions are represented in their diagram of the "Hierarchy of Controls" shown below with the most effective steps at the top and the least effective at the bottom.
The high vaporization potential and flammability of chloroprene
has significant implications for handling and storage operations in the
occupational setting. Chloroprene should be stored in closed containers
in a cool, well-ventilated area with the temperature no higher than 50
degrees Fahrenheit. In addition, chloroprene has a high reactivity and
should be stored away from oxidizing agents such as perchlorate,
peroxides, permanganates, chlorates, nitrates, chlorine, bromine, and
fluorine. All activities inducing a potential fire hazard should be
avoided. For instance, smoking, having open flames or using sparking
tools to open or close storage containers should be prohibited. It is
also advised that grounded and bonded metal containers are used for the
transport of chloroprene.
Occupational exposure limits
The
official legal body that develops and enforces occupational exposure
limits (OEL) in order to ensure workplace safety and health regulations
is the Occupational Health and Safety Administration (OSHA) that works
under the U.S. Department of Labor.
OSHA's permissible exposure limits (PELs), a guideline for occupational
exposures, were adopted from the 1968 threshold limit values (TLVs) of
the American Conference of Governmental Industrial Hygienists (ACGIH). Each year, the ACGIH publish their TLV and BEI booklet
that provides updated information on "occupational exposure guidelines
for more than 700 chemical substances and physical agents."
The scientific literature on certain chemical and physical exposures
has evolved since 1968, therefore OSHA recognizes that their PELs may
not guarantee worker health and safety. The National Institute for Occupational Health and Safety (NIOSH) under the U.S. Department of Health and Human Services
compensates for the rigidity of the PEL by researching "all medical,
biological, engineering, chemical, and trade information relevant to the
hazard" and publishing recommended exposure limits (RELs) based on
their research.
Therefore, as a way to ensure worker safety and health, the following
sections on safety guidelines and hazard control will consider the most
recent occupational exposure limits from ACGIH's 2018 TLV and BEI booklet and NIOSH's REL.
A table of occupational exposure limits (OELs) from various jurisdictions follows. In general, the OELs range from 0.55 ppm to 25 ppm.
| Occupational Exposure Limits for Chloroprene |
|---|
In the ACGIH's 2018 TLV and BEI booklet, chloroprene was
designated with a skin and an A2 notation. The skin notation designation
is based on animal and human research that have shown chloroprene's
ability to be absorbed by the skin. An A2 designation by the ACGIH means that the substance is a suspected human carcinogen
with support from human data that are accepted as adequate in quality
but may not be enough to declare an A1 (known human carcinogen)
designation. Additionally, the TLV basis for these designations are due
to scientific studies that show an association between chloroprene
exposure and lung cancer, upper respiratory tract (URT) and eye
irritation.
Public health implications
Since
chloroprene usage is limited to those facilities producing Neoprene,
the occupational health risks are isolated to those facilities. However,
insufficient control of chloroprene emissions may extend the health and
safety concerns of chloroprene beyond the facility and into the
surrounding areas. Chloroprene release is predominately as an air
pollutant, but other feasible fates and transport of chloroprene in the
environment are discussed below.
In the fourteenth edition of the National Institute of Health
report on carcinogens, the half-life time differences between
chloroprene in air, water and soil were highlighted. In the air,
chloroprene “reacts with photo-chemically generated hydroxyl radicals”
and has a half-life of 18 hours. The smaller amounts that are removed by
reaction with ozone have a half-life of 10 days. In streams,
chloroprene is stated to volatilize quickly with a half-life of 3 hours.
However, in bigger bodies of water such as a lake, the half-life of
chloroprene is 4 days. Similar to its reaction with water, chloroprene
on soil was cited to volatilize from the surface. However, the report
remarked that chloroprene holds the potential to leach into groundwater
supplies.
Due to its volatility and extreme reactivity, the threat of chloroprene
exists predominantly as an air pollutant and is not expected to
bioaccumulate or persist in the environment according to the U.S EPA
Toxicological Review of Chloroprene.
However, the Centers for Disease Control and Prevention (CDC) states
that chloroprene does, in fact, have the potential to persist in the
environment. Nonetheless, the primary route of exposure for animals and
humans is inhalation, but can be absorbed through the skin or
indigestion.
In December 2015, the EPA released its 2011 National Air Toxic
Assessment to help state and local agencies prioritize the required
steps in identifying and mitigating sources of air pollution.
In this report, it was measured that chloroprene was being released
from Denka Performance Elastomer's Pontchartrain facility located in LaPlace, Louisiana.
EPA worked with the Louisiana Department of Environmental Quality,
DuPont and the nonprofit organization Louisiana Environmental Action
Network to institute monitoring of chloroprene pollution near the
facility and in the surrounding neighborhood. Air monitoring is ongoing.