Normal absorption and distribution of copper. Cu = copper, CP = ceruloplasmin, green = ATP7B carrying copper.
Copper is an essential trace element that is vital to the health of all living things (humans, plants, animals, and microorganisms). In humans, copper is essential to the proper functioning of organs and metabolic processes. The human body has complex homeostatic
mechanisms which attempt to ensure a constant supply of available
copper, while eliminating excess copper whenever this occurs. However,
like all essential elements and nutrients, too much or too little nutritional
ingestion of copper can result in a corresponding condition of copper
excess or deficiency in the body, each of which has its own unique set
of adverse health effects.
Daily dietary standards for copper have been set by various
health agencies around the world. Standards adopted by some nations
recommend different copper intake levels for adults, pregnant women,
infants, and children, corresponding to the varying need for copper
during different stages of life.
Copper deficiency and toxicity can be either of genetic or non-genetic origin. The study of copper's genetic diseases,
which are the focus of intense international research activity, has
shed insight into how human bodies use copper, and why it is important
as an essential micronutrient.
The studies have also resulted in successful treatments for genetic
copper excess conditions, enabling patients whose lives were once
jeopardized to live long and productive lives.
Researchers specializing in the fields of microbiology, toxicology, nutrition, and health risk assessments
are working together to define the precise copper levels that are
required for essentiality, while avoiding deficient or excess copper
intakes. Results from these studies are expected to be used to fine-tune
governmental dietary recommendation programs which are designed to help
protect public health.
Essentiality
Copper is an essential trace element (i.e., micronutrient) that is required for plant, animal, and human health.
It is also required for the normal functioning of aerobic (oxygen-requiring) microorganisms.
Copper is incorporated into a variety of proteins and metalloenzymes
which perform essential metabolic functions; the micronutrient is
necessary for the proper growth, development, and maintenance of bone, connective tissue, brain, heart, and many other body organs. Copper is involved in the formation of red blood cells, the absorption and utilization of iron, the metabolism of cholesterol and glucose, and the synthesis and release of life-sustaining proteins and enzymes. These enzymes in turn produce cellular energy and regulate nerve transmission, blood clotting, and oxygen transport.
Copper stimulates the immune system to fight infections, to repair injured tissues, and to promote healing. Copper also helps to neutralize "free-radicals", which can cause severe damage to cells.
Copper's essentiality was first discovered in 1928, when it was
demonstrated that rats fed a copper-deficient milk diet were unable to
produce sufficient red blood cells. The anemia was corrected by the addition of copper-containing ash from vegetable or animal sources.
As an essential trace element, daily dietary requirements for
copper have been recommended by a number of governmental health agencies
around the world.
Fetuses, infants, and children
Copper is essential for the normal growth and development of human fetuses, infants, and children. The human fetus accumulates copper rapidly in its liver
during the third trimester of pregnancy. At birth, a healthy infant has
four times the concentration of copper than a full-grown adult. Human milk is relatively low in copper, and the neonate's liver stores falls rapidly after birth, supplying copper to the fast-growing body during the breast feeding period. These supplies are necessary to carry out such metabolic functions as cellular respiration, melanin pigment and connective tissue synthesis, iron metabolism, free radical defense, gene expression, and the normal functioning of the heart and immune systems in infants.
Infants have special biochemical mechanisms for adequately
managing copper in their bodies while permanent lifelong mechanisms
develop and mature.
Severe deficiency of copper in pregnant mothers increases the
risk of health problems in their fetuses and infants. Health effects
noted include low birth weights, muscle weaknesses, and neurologic problems. However, copper deficiencies in pregnant women can be avoided with a balanced diet.
Since copper availability in the body is hindered by an excess of iron and zinc intake, pregnant women prescribed iron supplements to treat anemia
or zinc supplements to treat colds should consult physicians to be sure
that the prenatal supplements they may be taking also have
nutritionally-significant amounts of copper.
When newborn babies are breastfed, the babies' livers and the
mothers' breast milk provide sufficient quantities of copper for the
first 4–6 months of life. When babies are weaned, a balanced diet should provide adequate sources of copper.
Cow's milk and some older infant formulas are depleted in copper. Most formulas are now fortified with copper to prevent depletion.
Most well-nourished children have adequate intakes of copper. Health-compromised children, including those who are premature, malnourished, have low birth weights, suffer from infections, and who experience rapid catch-up growth
spurts, are at elevated risk for copper deficiencies. Fortunately,
diagnosis of copper deficiency in children is clear and reliable once
the condition is suspected. Supplements under a physician's supervision
usually facilitate a full recovery.
Homeostasis
Copper is absorbed, transported, distributed, stored, and excreted in the body according to complex homeostatic processes which ensure a constant and sufficient supply of the micronutrient while simultaneously avoiding excess levels.
If an insufficient amount of copper is ingested for a short period of
time, copper stores in the liver will be depleted. Should this depletion
continue, a copper health deficiency condition may develop. If too much
copper is ingested, an excess condition can result. Both of these
conditions, deficiency and excess, can lead to tissue injury and
disease. However, due to homeostatic regulation, the human body is
capable of balancing a wide range of copper intakes for the needs of
healthy individuals.
Many aspects of copper homeostasis are known at the molecular level. Copper's essentiality is due to its ability to act as an electron donor or acceptor as its oxidation state fluxes between Cu1+(cuprous) and Cu2+ (cupric). As a component of about a dozen cuproenzymes, copper is involved in key redox (i.e., oxidation-reduction) reactions in essential metabolic processes such as mitochondrial respiration, synthesis of melanin, and cross-linking of collagen.
Copper is an integral part of the antioxidant enzyme, copper-zinc
superoxide dismutase (Cu,Zn-SOD), and has a role in iron homeostasis as a
cofactor in ceruloplasmin. A list of some key copper-containing enzymes and their functions is summarized below:
| Enzymes | Function |
|---|---|
| Amine oxidases | Group of enzymes oxidizing primary amines (e.g., tyramine, histidine and polylamines) |
| Ceruloplasmin (ferroxidase I) | Multi-copper oxidase in plasma, essential for iron transport |
| Cytochrome c oxidase | Terminal oxidase enzyme in mitochondrial respiratory chain, involved in electron transport |
| Dopamine β-hydroxylase | Involved in catecholamine metabolism, catalyzes conversion of dopamine to norepinephrine |
| Hephaestin | Multi-copper ferroxidase, involved in iron transport across intestinal mucosa into portal circulation |
| Lysyl oxidase | Cross-linking of collagen and elastin |
| Peptidylglycine alpha-amidating mono-oxygenase (PAM) |
Multifunction enzyme involved in maturation and modification of key neuropeptides (e.g., neurotransmitters, neuroendocrine peptides) |
| Superoxide dismutase (Cu, Zn) | Intracellular and extracellular enzyme involved in defense against reactive oxygen species (e.g., destruction of superoxide radicals) |
| Tyrosinase | Enzyme catalyzing melanin and other pigment production |
The transport and metabolism of copper in living organisms is
currently the subject of much active research. Copper transport at the
cellular level involves the movement of extracellular copper across the cell membrane and into the cell by specialized transporters. In the bloodstream, copper is carried throughout the body by albumin, ceruloplasmin,
and other proteins. The majority of blood copper (or serum copper) is
bound to ceruloplasmin. The proportion of ceruloplasmin-bound copper can
range from 70-95% and differs between individuals, depending, for
example, on hormonal cycle, season, and copper status. Intracellular
copper is routed to sites of synthesis of copper-requiring enzymes and to organelles by specialized proteins called metallochaperones. Another set of these transporters carries copper into subcellular compartments.
Certain mechanisms exist to release copper from the cell. Specialized
transporters return excess unstored copper to the liver for additional
storage and/or biliary excretion.
These mechanisms ensure that free unbound toxic ionic copper is
unlikely to exist in the majority of the population (i.e., those without
genetic copper metabolism defects).
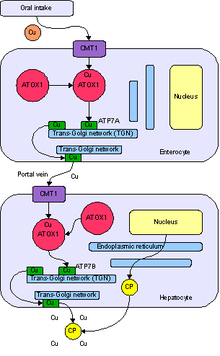
Copper is imported into cells through the cell wall by the plasma membrane
transport protein known as Copper Transporter 1, or Ctr1. Ctr1 rapidly
binds to intracellular copper chaperone proteins. Atox1 delivers copper
to the secretory pathway and docks with either copper-transporting
ATPase ATP7B in the liver or ATP7A in other cells. ATP7B directs copper
to plasma ceruloplasmin or to biliary excretion in concert with a newly
discovered chaperone, Murr1, the protein missing in canine copper
toxicosis. ATP7A directs copper within the trans-Golgi network to the proteins dopamine beta-monooxygenase, peptidylglycine alpha-amidating monooxygenase, lysyl oxidase, and tyrosinase, depending on the cell type. CCS
is the copper chaperone for the Cu/Zn-superoxide dismutase that
protects cells against reactive oxygen species; it delivers copper in
the cytoplasm and intermitochondrial space. Cox17 delivers copper to mitochondria to cytochrome c oxidase via the chaperones Cox11, Sco1, and Sco2. Other copper chaperones may exist and might include metallothionein and amyloid precursor protein (APP). Genetic and nutritional studies have illustrated the essential nature of these copper-binding proteins.
Absorption
In
mammals copper is absorbed in the stomach and small intestine, although
there appear to be differences among species with respect to the site
of maximal absorption. Copper is absorbed from the stomach and duodenum in rats and from the lower small intestine in hamsters.
The site of maximal copper absorption is not known for humans, but is
assumed to be the stomach and upper intestine because of the rapid
appearance of Cu64 in the plasma after oral administration.
Absorption of copper ranges from 15–97%, depending on copper content, form of the copper, and composition of the diet.
Various factors influence copper absorption. For example, copper absorption is enhanced by ingestion of animal protein, citrate, and phosphate. Copper salts, including copper gluconate, copper acetate, or copper sulfate, are more easily absorbed than copper oxides. Elevated levels of dietary zinc, as well as cadmium, high intakes of phytate and simple sugars (fructose, sucrose) inhibit dietary absorption of copper. Furthermore, low levels of dietary copper inhibit iron absorption.
Some forms of copper are not soluble in stomach acids and cannot
be absorbed from the stomach or small intestine. Also, some foods may
contain indigestible fiber that binds with copper. High intakes of zinc can significantly decrease copper absorption. Extreme intakes of Vitamin C or iron
can also affect copper absorption, reminding us of the fact that
micronutrients need to be consumed as a balanced mixture. This is one
reason why extreme intakes of any one single micronutrient are not
advised.
Individuals with chronic digestive problems may be unable to absorb
sufficient amounts of copper, even though the foods they eat are
copper-rich.
Several copper transporters have been identified that can move copper across cell membranes.
Other intestinal copper transporters may exist. Intestinal copper
uptake may be catalyzed by Ctr1. Ctr1 is expressed in all cell types so
far investigated, including enterocytes, and it catalyzes the transport
of Cu+1 across the cell membrane.
Excess copper (as well as other heavy metal ions like zinc or
cadmium) may be bound by metallothionein and sequestered within
intracellular vesicles of enterocytes (i.e., predominant cells in the small intestinal mucosa).
Distribution
Copper released from intestinal cells moves to the serosal (i.e., thin membrane lining) capillaries where it binds to albumin, glutathione, and amino acids in the portal blood. There is also evidence for a small protein, transcuprein, with a specific role in plasma copper transport
Several or all of these copper-binding molecules may participate in
serum copper transport. Copper from portal circulation is primarily
taken up by the liver. Once in the liver, copper is either incorporated
into copper-requiring proteins, which are subsequently secreted into the
blood. Most of the copper (70 – 95%) excreted by the liver is
incorporated into ceruloplasmin, the main copper carrier in blood. Copper is transported to extra-hepatic tissues by ceruloplasmin, albumin and amino acids, or excreted into the bile. By regulating copper release, the liver exerts homeostatic control over extrahepatic copper.
Excretion
Bile is the major pathway for the excretion of copper and is vitally important in the control of liver copper levels.
Most fecal copper results from biliary excretion; the remainder is
derived from unabsorbed copper and copper from desquamated mucosal
cells.
| Dose range | Approximate daily intakes | Health outcomes |
|---|---|---|
| Death | ||
| Gross dysfunction and disturbance of metabolism of other nutrients; hepatic
"detoxification" and homeostasis overwhelmed
| ||
| Toxic | >5.0 mg/kg body weight | Gastrointestinal metallothionein induced (possible differing effects of acute and chronic
exposure)
|
| 100 μg/kg body weight | Plateau of absorption maintained; homeostatic mechanisms regulate absorption of copper | |
| Adequate | 34 μg/kg body weight | Hepatic uptake, sequestration and excretion effect homeostasis; glutathione-dependent uptake of copper; binding to metallothionein; and lysosomal excretion of copper |
| 11 μg/kg body weight | Biliary excretion and gastrointestinal uptake normal | |
| 9 μg/kg body weight | Hepatic deposit(s) reduced; conservation of endogenous copper; gastrointestinal
absorption increased
| |
| Deficient | 8.5 μg/kg body weight | Negative copper balance |
| 5.2 μg/kg body weight | Functional defects, such as lysyl oxidase and superoxide dismutase activities reduced; impaired substrate metabolism | |
| 2 μg/kg body weight | Peripheral pools disrupted; gross dysfunction and disturbance of metabolism of other
nutrients; death
|
Dietary recommendations
Various
national and international organizations concerned with nutrition and
health have standards for copper intake at levels judged to be adequate
for maintaining good health. These standards are periodically changed
and updated as new scientific data become available. The standards
sometimes differ among countries and organizations.
Adults
The World Health Organization recommends a minimal acceptable intake of approximately 1.3 mg/day.
These values are considered to be adequate and safe for most of the
general population. In North America, the U.S. Institute of Medicine
(IOM) set the Recommended Dietary Allowance (RDA) for copper for healthy
adult men and women at 0.9 mg/day. As for safety, the IOM also sets Tolerable upper intake levels (ULs) for vitamins and minerals when evidence is sufficient. In the case of copper the UL is set at 10 mg/day. The European Food Safety Authority reviewed the same safety question and set its UL at 5 mg/day.
Adolescents, children, and infants
The
World Health Organization has not developed minimum daily intakes for
these age groups. In North America the RDAs are as follows: 0.34 mg/day
for children of 1–3 years; 0.44 mg/day for 4–8 years; 0.7 mg/day for
9–13 years; and 0.89 mg/day for 14–18 years. The ULs are: 1 mg/day for
children of 1–3 years; 3 mg/day for 4–8 years; 5 mg/day for 9–13 years;
and 8 mg/day for 14–18 years.
Full-term and premature infants are more sensitive to copper
deficiency than adults. Since the fetus accumulates copper during the
last 3 months of pregnancy, infants that are born prematurely have not
had sufficient time to store adequate reserves of copper in their livers
and therefore require more copper at birth than full-term infants.
For full-term infants, the North American recommended safe and
adequate intake is approximately 0.2 mg/day. For premature babies, it is
considerably higher: 1 mg/day. The World Health Organization has
recommended similar minimum adequate intakes and advises that premature
infants be given formula supplemented with extra copper to prevent the
development of copper deficiency.
Pregnant and lactating women
In North America, the IOM has set the RDA for pregnancy at 1.0 mg/day and for lactation at 1.3 mg/day. The European Food Safety Authority
(EFSA) refers to the collective set of information as Dietary Reference
Values, with Population Reference Intake (PRI) instead of RDA. PRI for
pregnancy is 1.6 mg/day, for lactation 1.6 mg/day - higher than the U.S.
RDAs.
Food sources
Foods rich in copper
Copper is an essential trace mineral that cannot be formed by the human body. It must be ingested from dietary sources.
Foods contribute virtually all of the copper consumed by humans. The best dietary sources include seafood (especially shellfish), organ meats (e.g., liver), whole grains, legumes (e.g., beans and lentils) and chocolate. Nuts, including peanuts and pecans, are especially rich in copper, as are grains such as wheat and rye, and several fruits including lemons and raisins. Other food sources that contain copper include cereals, potatoes, peas, red meat, mushrooms, some dark green leafy vegetables (such as kale), and fruits (coconuts, papaya and apples). Tea, rice and chicken are relatively low in copper, but can provide a reasonable amount of copper when they are consumed in significant amounts.
Eating a balanced diet with a range of foods from different food
groups is the best way to avoid copper deficiency. In both developed and
developing countries, adults, young children, and adolescents who
consume diets of grain, millet, tuber,
or rice along with legumes (beans) or small amounts of fish or meat,
some fruits and vegetables, and some vegetable oil are likely to obtain
adequate copper if their total food consumption is adequate in calories.
In developed countries where consumption of red meat is high, copper
intake is also likely to be adequate.
As a natural element in the earth's crust, copper exists in most
of the world's surface water and groundwater, although the actual
concentration of copper in natural waters varies geographically.
Drinking water can comprise 20-25% of dietary copper.
In many regions of the world, copper tubing that conveys drinking
water can be a source of dietary copper. Copper tube can leach a small
amount of copper, particularly in its first year or two of service.
Afterwards, a protective surface usually forms on the inside of copper
tubes that retards leaching.
In France and some other countries, copper bowls are traditionally used for whipping egg white,
as the copper helps stabilise bonds in the white as it is beaten and
whipped. Small amounts of copper may leach from the bowl during the
process and enter the egg white.
Supplementation
Copper
supplements can prevent copper deficiency, but supplements should be
taken only under a doctor's supervision. Different forms of copper
supplementation have different absorption rates. For example, the
absorption of copper from cupric oxide supplements is lower than that from copper gluconate, sulfate, or carbonate.
Supplementation is generally not recommended for healthy adults
who consume a well-balanced diet which includes a wide range of foods.
However, supplementation under the care of a physician may be necessary
for premature infants or those with low birth weights, infants fed
unfortified formula or cow's milk during the first year of life, and
malnourished young children. Physicians may consider copper
supplementation for 1) illnesses that reduce digestion (e.g., children
with frequent diarrhea or infections; alcoholics), 2) insufficient food consumption (e.g., the elderly, the infirm, those with eating disorders or on diets), 3) patients taking medications that block the body's use of copper, 4) anemia patients who are treated with iron supplements, 5) anyone taking zinc supplements, and 6) those suffering from osteoporosis.
Many popular vitamin supplements include copper as small
inorganic molecules such as cupric oxide. These supplements can result
in excess free copper in the brain as the copper can cross the
blood-brain barrier directly. Normally, organic copper in food is first
processed by the liver which keeps free copper levels under control.
Copper deficiency and excess health conditions (non-genetic)
If insufficient quantities of copper are ingested, copper reserves in
the liver will become depleted and a copper deficiency leading to
disease or tissue injury (and in extreme cases, death). Toxicity from
copper deficiency can be treated with a balanced diet or supplementation
under the supervision of a doctor. On the contrary, like all
substances, excess copper intake at levels far above World Health Organization limits can become toxic.
Acute copper toxicity is generally associated with accidental
ingestion. These symptoms abate when the high copper food source is no
longer ingested.
In 1996, the International Program on Chemical Safety, a World
Health Organization-associated agency, stated "there is greater risk of
health effects from deficiency of copper intake than from excess copper
intake." This conclusion was confirmed in recent multi-route exposure
surveys.
The health conditions of non-genetic copper deficiency and copper excess are described below.
Copper deficiency
There
are conflicting reports on the extent of deficiency in the U.S. One
review indicates approximately 25% of adolescents, adults, and people
over 65, do not meet the Recommended Dietary Allowance for copper.
Another source states less common: a federal survey of food consumption
determined that for women and men over the age of 19, average
consumption from foods and beverages was 1.11 and 1.54 mg/day,
respectively. For women, 10% consumed less than the Estimated Average
Requirement, for men fewer than 3%.
Acquired copper deficiency has recently been implicated in adult-onset progressive myeloneuropathy and in the development of severe blood disorders including myelodysplastic syndrome. Fortunately, copper deficiency can be confirmed by very low serum metal and ceruloplasmin concentrations in the blood.
Other conditions linked to copper deficiency include osteoporosis, osteoarthritis, rheumatoid arthritis,
cardiovascular disease, colon cancer, and chronic conditions involving
bone, connective tissue, heart and blood vessels. nervous system and
immune system. Copper deficiency alters the role of other cellular constituents involved in antioxidant activities, such as iron, selenium, and glutathione, and therefore plays an important role in diseases in which oxidant stress
is elevated. A marginal, i.e., 'mild' copper deficiency, believed to be
more widespread than previously thought, can impair human health in
subtle ways.
Populations susceptible to copper deficiency include those with genetic defects for Menkes disease,
low-birth-weight infants, infants fed cow's milk instead of breast milk
or fortified formula, pregnant and lactating mothers, patients
receiving total parenteral nutrition, individuals with "malabsorption syndrome" (impaired dietary absorption), diabetics,
individuals with chronic diseases that result in low food intake, such
as alcoholics, and persons with eating disorders. The elderly and athletes may also be at higher risk for copper deficiency due to special needs that increase the daily requirements. Vegetarians may have decreased copper intake due to the consumption of plant foods in which copper bioavailability is low.
Fetuses and infants of severely copper deficient women have increased
risk of low birth weights, muscle weaknesses, and neurological problems.
Copper deficiencies in these populations may result in anemia, bone
abnormalities, impaired growth, weight gain, frequent infections (colds,
flu, pneumonia), poor motor coordination, and low energy.
Copper excess
Copper excess is a subject of much current research. Distinctions
have emerged from studies that copper excess factors are different in
normal populations versus those with increased susceptibility to adverse
effects and those with rare genetic diseases.
This has led to statements from health organizations that could be
confusing to the uninformed. For example, according to a U.S. Institute
of Medicine report,
the intake levels of copper for a significant percentage of the
population are lower than recommended levels. On the other hand, the
U.S. National Research Council
concluded in its report Copper in Drinking Water that there is concern
for copper toxicity in susceptible populations and recommended that
additional research be conducted to identify and characterize
copper-sensitive populations.
Excess copper intake causes stomach upset, nausea, and diarrhea and can lead to tissue injury and disease.
The oxidation potential
of copper may be responsible for some of its toxicity in excess
ingestion cases. At high concentrations copper is known to produce oxidative damage to biological systems, including peroxidation of lipids or other macromolecules.
While the cause and progression of Alzheimer's disease are not well understood, research indicates that, among several other key observations, iron, aluminum, and copper
accumulate in the brains of Alzheimer's patients. However, it is not
yet known whether this accumulation is a cause or a consequence of the
disease.
Research has been ongoing over the past two decades to determine
whether copper is a causative or a preventive agent of Alzheimer's
disease. For example, as a possible causative agent or an expression of a metal homeostasis
disturbance, studies indicate that copper may play a role in increasing
the growth of protein clumps in Alzheimer's disease brains, possibly by damaging a molecule that removes the toxic buildup of amyloid beta (Aβ) in the brain. There is an association between a diet rich in copper and iron together with saturated fat and Alzheimer's disease.
On the other hand, studies also demonstrate potential beneficial roles
of copper in treating rather than causing Alzheimer's disease. For example, copper has been shown to 1) promote the non-amyloidogenic processing of amyloid beta precursor protein (APP), thereby lowering amyloid beta (Aβ) production in cell culture systems 2) increase lifetime and decrease soluble amyloid production in APP transgenic mice, and 3) lower Aβ levels in cerebral spinal fluid in Alzheimer's disease patients.
Furthermore, long-term copper treatment (oral intake of 8 mg
copper (Cu-(II)-orotate-dihydrate)) was excluded as a risk factor for
Alzheimer's disease in a noted clinical trial on humans
and a potentially beneficial role of copper in Alzheimer's disease has
been demonstrated on cerebral spinal fluid levels of Aβ42, a toxic
peptide and biomarker of the disease.
More research is needed to understand metal homeostasis disturbances in
Alzheimer's disease patients and how to address these disturbances
therapeutically. Since this experiment used Cu-(II)-orotate-dihydrate,
it does not relate to the effects of cupric oxide in supplements.
Copper toxicity from excess exposures
In
humans, the liver is the primary organ of copper-induced toxicity.
Other target organs include bone and the central nervous and immune
systems.
Excess copper intake also induces toxicity indirectly by interacting
with other nutrients. For example, excess copper intake produces anemia
by interfering with iron transport and/or metabolism.
The identification of genetic disorders of copper metabolism leading to severe copper toxicity (i.e., Wilson disease)
has spurred research into the molecular genetics and biology of copper
homeostasis (for further information, refer to the following section on
copper genetic diseases). Much attention has focused on the potential
consequences of copper toxicity in normal and potentially susceptible
populations. Potentially susceptible subpopulations include hemodialysis
patients and individuals with chronic liver disease. Recently, concern
was expressed about the potential sensitivity to liver disease of
individuals who are heterozygote
carriers of Wilson disease genetic defects (i.e., those having one
normal and one mutated Wilson copper ATPase gene) but who do not have
the disease (which requires defects in both relevant genes). However, to date, no data are available that either support or refute this hypothesis.
Acute exposures
In
case reports of humans intentionally or accidentally ingesting high
concentrations of copper salts (doses usually not known but reported to
be 20–70 grams of copper), a progression of symptoms was observed
including abdominal pain, headache, nausea, dizziness, vomiting and
diarrhea, tachycardia, respiratory difficulty, hemolytic anemia, hematuria, massive gastrointestinal bleeding, liver and kidney failure, and death.
Episodes of acute gastrointestinal upset following single or
repeated ingestion of drinking water containing elevated levels of
copper (generally above 3–6 mg/L) are characterized by nausea, vomiting,
and stomach irritation. These symptoms resolve when copper in the
drinking water source is reduced.
Three experimental studies were conducted that demonstrate a
threshold for acute gastrointestinal upset of approximately 4–5 mg/L in
healthy adults, although it is not clear from these findings whether
symptoms are due to acutely irritant effects of copper and/or to
metallic, bitter, salty taste.
In an experimental study with healthy adults, the average taste
threshold for copper sulfate and chloride in tap water, deionized water,
or mineral water was 2.5–3.5 mg/L. This is just below the experimental threshold for acute gastrointestinal upset.
Chronic exposures
The
long-term toxicity of copper has not been well studied in humans, but
it is infrequent in normal populations that do not have a hereditary
defect in copper homeostasis.
There is little evidence to indicate that chronic human exposure to copper results in systemic effects other than liver injury.
Chronic copper poisoning leading to liver failure was reported in a
young adult male with no known genetic susceptibility who consumed
30–60 mg/d of copper as a mineral supplement for 3 years.
Individuals residing in U.S. households supplied with tap water
containing >3 mg/L of copper exhibited no adverse health effects.
No effects of copper supplementation on serum liver enzymes,
biomarkers of oxidative stress, and other biochemical endpoints have
been observed in healthy young human volunteers given daily doses of 6
to 10 mg/d of copper for up to 12 weeks.
Infants aged 3–12 months who consumed water containing 2 mg Cu/L for 9
months did not differ from a concurrent control group in
gastrointestinal tract (GIT) symptoms, growth rate, morbidity, serum
liver enzyme and bilirubin levels, and other biochemical endpoints.)
Serum ceruloplasmin was transiently elevated in the exposed infant
group at 9 months and similar to controls at 12 months, suggesting
homeostatic adaptation and/or maturation of the homeostatic response.
Dermal exposure has not been associated with systemic toxicity
but anecdotal reports of allergic responses may be a sensitization to
nickel and cross-reaction with copper or a skin irritation from copper.
Workers exposed to high air levels of copper (resulting in an estimated
intake of 200 mg Cu/d) developed signs suggesting copper toxicity
(e.g., elevated serum copper levels, hepatomegaly). However, other
co-occurring exposures to pesticidal agents or in mining and smelting
may contribute to these effects.
Effects of copper inhalation are being thoroughly investigated by an
industry-sponsored program on workplace air and worker safety. This
multi-year research effort is expected to be finalized in 2011.
Measurements of elevated copper status
Although
a number of indicators are useful in diagnosing copper deficiency,
there are no reliable biomarkers of copper excess resulting from dietary
intake. The most reliable indicator of excess copper status is liver
copper concentration. However, measurement of this endpoint in humans is
intrusive and not generally conducted except in cases of suspected
copper poisoning. Increased serum copper or ceruolplasmin levels are not
reliably associated with copper toxicity as elevations in
concentrations can be induced by inflammation, infection, disease,
malignancies, pregnancy, and other biological stressors. Levels of
copper-containing enzymes, such as cytochrome c oxidase, superoxide
dismutase, and diaminase oxidase, vary not only in response to copper
state but also in response to a variety of other physiological and
biochemical factors and therefore are inconsistent markers of excess
copper status.
A new candidate biomarker for copper excess as well as deficiency
has emerged in recent years. This potential marker is a chaperone
protein, which delivers copper to the antioxidant protein SOD1 (copper,
zinc superoxide dismutase). It is called "copper chaperone for SOD1"
(CCS), and excellent animal data supports its use as a marker in
accessible cells (e.g., erythrocytes) for copper deficiency as well as excess. CCS is currently being tested as a biomarker in humans.
Hereditary copper metabolic diseases
Several rare genetic diseases (Wilson disease, Menkes disease, idiopathic copper toxicosis, Indian childhood cirrhosis) are associated with the improper utilization of copper in the body. All of these diseases involve mutations of genes containing the genetic codes
for the production of specific proteins involved in the absorption and
distribution of copper. When these proteins are dysfunctional, copper
either builds up in the liver or the body fails to absorb copper.
These diseases are inherited and cannot be acquired. Adjusting
copper levels in the diet or drinking water will not cure these
conditions (although therapies are available to manage symptoms of
genetic copper excess disease).
The study of genetic copper metabolism diseases and their
associated proteins are enabling scientists to understand how human
bodies use copper and why it is important as an essential micronutrient.
The diseases arise from defects in two similar copper pumps, the Menkes and the Wilson Cu-ATPases.
The Menkes ATPase is expressed in tissues like skin-building
fibroblasts, kidneys, placenta, brain, gut and vascular system, while
the Wilson ATPase is expressed mainly in the liver, but also in mammary
glands and possibly in other specialized tissues. This knowledge is leading scientists towards possible cures for genetic copper diseases.
Menkes disease
Menkes disease,
a genetic condition of copper deficiency, was first described by John
Menkes in 1962. It is a rare X-linked disorder that affects
approximately 1/200,000 live births, primarily boys.
Livers of Menkes disease patients cannot absorb essential copper needed
for patients to survive. Death usually occurs in early childhood: most
affected individuals die before the age of 10 years, although several
patients have survived into their teens and early 20s.
The protein produced by the Menkes gene is responsible for transporting copper across the gastrointestinal tract (GIT) mucosa and the blood–brain barrier.
Mutational defects in the gene encoding the copper ATPase cause copper
to remain trapped in the lining of the small intestine. Hence, copper
cannot be pumped out of the intestinal cells and into the blood for
transport to the liver and consequently to rest of the body. The disease therefore resembles a severe nutritional copper deficiency despite adequate ingestion of copper.
Symptoms of the disease include coarse, brittle, depigmented hair
and other neonatal problems, including the inability to control body
temperature, mental retardation, skeletal defects, and abnormal
connective tissue growth.
Menkes patients exhibit severe neurological abnormalities,
apparently due to the lack of several copper-dependent enzymes required
for brain development, including reduced cytochrome c oxidase activity. The brittle, kinky hypopigmented hair of steely appearance is due to a deficiency in an unidentified cuproenzyme. Reduced lysyl oxidase activity results in defective collagen and elastin polymerization and corresponding connective-tissue abnormalities including aortic aneurisms, loose skin, and fragile bones.
With early diagnosis and treatment consisting of daily injections of copper histidine intraperitoneally and intrathecally
to the central nervous system, some of the severe neurological problems
may be avoided and survival prolonged. However, Menkes disease patients
retain abnormal bone and connective-tissue disorders and show mild to
severe mental retardation. Even with early diagnosis and treatment, Menkes disease is usually fatal.
Ongoing research into Menkes disease is leading to a greater understanding of copper homeostasis, the biochemical mechanisms involved in the disease, and possible ways to treat it.
Investigations into the transport of copper across the blood/brain
barrier, which are based on studies of genetically altered mice, are
designed to help researchers understand the root cause of copper
deficiency in Menkes disease. The genetic makeup of "transgenic mice"
is altered in ways that help researchers garner new perspectives about
copper deficiency. The research to date has been valuable: genes can be
'turned off' gradually to explore varying degrees of deficiency.
Researchers have also demonstrated in test tubes that damaged DNA
in the cells of a Menkes patient can be repaired. In time, the
procedures needed to repair damaged genes in the human body may be
found.
Wilson's disease
Wilson's disease is a rare autosomal (chromosome 13) recessive genetic disorder of copper transport that causes an excess of copper to build up in the liver. This results in liver toxicity, among other symptoms. The disease is now treatable.
Wilson's disease is produced by mutational defects of a protein that transports copper from the liver to the bile for excretion.
The disease involves poor incorporation of copper into ceruloplasmin
and impaired biliary copper excretion and is usually induced by
mutations impairing the function of the Wilson copper ATPase. These
genetic mutations produce copper toxicosis due to excess copper
accumulation, predominantly in the liver and brain and, to a lesser
extent, in kidneys, eyes, and other organs.
The disease, which affects about 1/30,000 infants of both genders,
may become clinically evident at any time from infancy through early
adulthood. The age of onset of Wilson's disease ranges from 3 to 50
years of age. Initial symptoms include hepatic, neurologic, or psychiatric disorders and, rarely, renal, skeletal, or endocrine symptomatology. The disease progresses with deepening jaundice and the development of encephalopathy, severe clotting abnormalities, occasionally associated with intravascular coagulation,
and terminal renal insufficiency. A peculiar type of tremor in the
upper extremities, slowness of movement, and changes in temperament
become apparent. Kayser-Fleischer rings,
a rusty brown discoloration at the outer rims of the iris due to copper
deposition noted in 90% of patients, become evident as copper begins to
accumulate and affect the nervous system.
Almost always, death occurs if the disease is untreated.
Fortunately, identification of the mutations in the Wilson ATPase gene
underlying most cases of Wilson's disease has made DNA testing for
diagnosis possible.
If diagnosed and treated early enough, patients with Wilson's disease may live long and productive lives. Wilson's disease is managed by copper chelation therapy with D-penicillamine
(which picks up and binds copper and enables patients to excrete excess
copper accumulated in the liver), therapy with zinc sulfate or zinc
acetate, and restrictive dietary metal intake, such as the elimination
of chocolate, oysters, and mushrooms.
Zinc therapy is now the treatment of choice. Zinc produces a mucosal
block by inducing metallothionein, which binds copper in mucosal cells
until they slough off and are eliminated in the feces. and it competes with copper for absorption in the intestine by DMT1 (Divalent Metal transporter 1). More recently, experimental treatments with tetrathiomolybdate
showed promising results. Tetrathiomolybdate appears to be an excellent
form of initial treatment in patients who have neurologic symptoms. In
contrast to penicillamine therapy, initial treatment with
tetrathiomolybdate rarely allows further, often irreversible, neurologic
deterioration.
Over 100 different genetic defects leading to Wilson's disease have been described and are available on the Internet at . Some of the mutations have geographic clustering.
Many Wilson's patients carry different mutations on each chromosome 13 (i.e., they are
compound heterozygotes). Even in individuals who are homozygous for a mutation, onset and severity of the disease may vary. Individuals homozygous
for severe mutations (e.g., those truncating the protein) have earlier
disease onset. Disease severity may also be a function of environmental
factors, including the amount of copper in the diet or variability in
the function of other proteins that influence copper homeostasis.
It has been suggested that heterozygote carriers of the Wilson's
disease gene mutation may be potentially more susceptible to elevated
copper intake than the general population. A heterozygotic frequency of 1/90 people has been estimated in the overall population. However, there is no evidence to support this speculation.
Further, a review of the data on single-allelic autosomal recessive
diseases in humans does not suggest that heterozygote carriers are
likely to be adversely affected by their altered genetic status.
Other diseases in which abnormalities in copper metabolism appear to be involved include Indian childhood cirrhosis (ICC), endemic Tyrolean copper toxicosis (ETIC), and idiopathic
copper toxicosis (ICT), also known as non-Indian childhood cirrhosis.
ICT is a genetic disease recognized in the early twentieth century
primarily in the Tyrolean region of Austria and in the Pune region of India.
ICC, ICT, and ETIC are infancy syndromes that are similar in their apparent etiology and presentation. Both appear to have a genetic component and a contribution from elevated copper intake.
In cases of ICC, the elevated copper intake is due to heating
and/or storing milk in copper or brass vessels. ICT cases, on the other
hand, are due to elevated copper concentrations in water supplies.
Although exposures to elevated concentrations of copper are commonly
found in both diseases, some cases appear to develop in children who are
exclusively breastfed or who receive only low levels of copper in water
supplies.
The currently prevailing hypothesis is that ICT is due to a genetic
lesion resulting in impaired copper metabolism combined with high copper
intake. This hypothesis was supported by the frequency of occurrence of
parental consanguinity
in most of these cases, which is absent in areas with elevated copper
in drinking water and in which these syndromes do not occur.
ICT appears to be vanishing as a result of greater genetic
diversity within the affected populations in conjunction with
educational programs to ensure that tinned cooking utensils are used
instead of copper pots and pans being directly exposed to cooked foods.
The preponderance of cases of early childhood cirrhosis identified in Germany
over a period of 10 years were not associated with either external
sources of copper or with elevated hepatic metal concentrations Only occasional spontaneous cases of ICT arise today.
Cancer
The role of copper in angiogenesis associated with different types of cancers has been investigated. A copper chelator, tetrathiomolybdate, which depletes copper stores in the body, is under investigation as an anti-angiogenic agent in pilot and clinical trials. The drug may inhibit tumor angiogenesis in hepatocellular carcinoma, pleural mesothelioma, colorectal cancer, head and neck squamous cell carcinoma, breast cancer, and kidney cancer.
The copper complex of a synthetic salicylaldehyde pyrazole hydrazone
(SPH) derivative induced human umbilical endothelial cell (HUVEC)
apoptosis and showed anti-angiogenesis effect in vitro.
The trace element copper had been found promoting tumor growth.
Several evidence from animal models indicates that tumors concentrate
high levels of copper. Meanwhile, extra copper has been found in some
human cancers.
Recently, therapeutic strategies targeting copper in the tumor have
been proposed. Upon administration with a specific copper chelator,
copper complexes would be formed at a relatively high level in tumors.
Copper complexes are often toxic to cells, therefore tumor cells were
killed, while normal cells in the whole body remained alive for the
lower level of copper.
Some copper chelators get more effective or novel bioactivity
after forming copper-chelator complexes. It was found that Cu2+ was
critically needed for PDTC induced apoptosis in HL-60 cells.
The copper complex of salicylaldehyde benzoylhydrazone (SBH)
derivatives showed increased efficacy of growth inhibition in several
cancer cell lines, when compared with the metal-free SBHs.
SBHs can react with many kinds of transition metal cations and thereby forming a number of complexes.
Copper-SBH complexes were more cytotoxic than complexes of other
transitional metals (Cu > Ni > Zn = Mn > Fe = Cr > Co) in MOLT-4 cells,
an established human T-cell leukemia cell line. SBHs, especially their
copper complexes appeared to be potent inhibitors of DNA synthesis and
cell growth in several human cancer cell lines, and rodent cancer cell
lines.
Salicylaldehyde pyrazole hydrazone (SPH) derivatives were found to inhibit the growth of A549 lung carcinoma cells.
SPH has identical ligands for Cu2+ as SBH. The Cu-SPH complex was found
to induce apoptosis in A549, H322 and H1299 lung cancer cells.
Contraception with copper IUDs
A copper intrauterine device (IUD) is a type of long-acting reversible contraception that is considered to be one of the most effective forms of birth control.
It is also considered the most effective non-hormonal contraceptive device.
The copper IUD's primary mechanism of action is to prevent
fertilization. Active substances released from the IUD, together with
products derived from the inflammatory reaction present in the luminal
fluids of the genital tract, are toxic for spermatozoa and oocytes,
preventing the encounter of healthy gametes and the formation of viable
embryos.
Plant and animal health
In
addition to being an essential nutrient for humans, copper is vital for
the health of animals and plants and plays an important role in agriculture.
Plant health
Copper
concentrations in soil are not uniform around the world. In many areas,
soils have insufficient levels of copper. Soils that are naturally
deficient in copper often require copper supplements before agricultural
crops, such as cereals, can be grown.
Copper deficiencies in soil can lead to crop failure. Copper
deficiency is a major issue in global food production, resulting in
losses in yield and reduced quality of output. Nitrogen fertilizers can
worsen copper deficiency in agricultural soils.
The world's two most important food crops, rice and wheat, are highly susceptible to copper deficiency. So are several other important foods, including citrus, oats, spinach and carrots. On the other hand, some foods including coconuts, soybeans and asparagus, are not particularly sensitive to copper-deficient soils.
The most effective strategy to counter copper deficiency is to
supplement the soil with copper, usually in the form of copper sulfate. Sewage sludge is also used in some areas to replenish agricultural land with organics and trace metals, including copper.
Animal health
In livestock, cattle and sheep commonly show indications when they are copper deficient. Swayback, a sheep disease associated with copper deficiency, imposes enormous costs on farmers worldwide, particularly in Europe, North America, and many tropical countries. For pigs, copper has been shown to be an outstanding growth promoter.