 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Strattera, others |
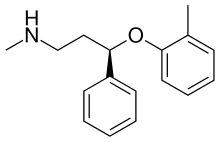
| Other names | (R)-N-Methyl-3-phenyl-3-(o-tolyloxy)propan-1-amine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a603013 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 63 to 94% |
| Protein binding | 98% |
| Metabolism | Liver, via CYP2D6 |
| Elimination half-life | 4.5-19 hours |
| Excretion | Kidney (80%) and faecal (17%) |
| Identifiers | |
| CAS Number | |
|---|---|
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.120.306 |
| Chemical and physical data | |
| Formula | C17H21NO |
| Molar mass | 255.361 g·mol−1 |
| 3D model (JSmol) | |
Atomoxetine, sold under the brand name Strattera, among others, is a medication used to treat attention deficit hyperactivity disorder (ADHD). It may be used alone or along with psychostimulants. Use of atomoxetine is only recommended for those who are at least six years old. It is taken by mouth. It was approved for medical use in the United States in 2002. In 2018, it was the 162nd most commonly prescribed medication in the United States, with more than 3 million prescriptions.
Common side effects of atomoxetine include abdominal pain, loss of appetite, nausea, feeling tired, and dizziness. Serious side effects may include angioedema, liver problems, stroke, psychosis, heart problems, suicide, and aggression. There is a lack of data regarding its safety during pregnancy; as of 2019, its safety during pregnancy and for use during breastfeeding is not certain.
Atomoxetine is a norepinephrine reuptake inhibitor and is believed to work by increasing norepinephrine and dopamine levels in the brain.
Medical uses
Attention deficit hyperactivity disorder
Atomoxetine is approved for use in children, adolescents, and adults. However, its efficacy has not been studied in children under six years old. Its primary advantage over the standard stimulant treatments for ADHD is that it has little known abuse potential. While it has been shown to significantly reduce inattentive and hyperactive symptoms, the responses were lower than the response to stimulants. Additionally, 40% of participants who were treated with atomoxetine experienced residual ADHD symptoms.
While its efficacy may be less than that of stimulant medications, there is some evidence that it may be used in combination with stimulants. Doctors may prescribe non-stimulants including atomoxetine when a person has bothersome side effects from stimulants; when a stimulant was not effective; in combination with a stimulant to increase effectiveness; or when there is concern about the abuse potential of psychostimulants in a patient with a history of drug use disorder.
Unlike α2 adrenoceptor agonists such as guanfacine and clonidine, atomoxetine's use can be abruptly stopped without significant discontinuation effects being seen.
The initial therapeutic effects of atomoxetine usually take 2–4 weeks to become apparent. A further 2–4 weeks may be required for the full therapeutic effects to be seen. The maximum recommended total daily dose in children and adolescents over 70 kg and adults is 100 mg.
Other
Atomoxetine may be used in those with ADHD and bipolar disorder although such use has not been well studied. Some benefit has also been seen in people with ADHD and autism.
Contraindications
Contraindications include:
- Hypersensitivity to atomoxetine or any of the inactive ingredients in the product
- Symptomatic cardiovascular disease including:
- -moderate to severe hypertension
- -atrial fibrillation
- -atrial flutter
- -ventricular tachycardia
- -ventricular fibrillation
- -ventricular flutter
- -advanced arteriosclerosis
- Severe cardiovascular disorders
- Pheochromocytoma
- Concomitant treatment with monoamine oxidase inhibitors
- Narrow angle glaucoma
- Poor metabolizers (due to the metabolism of atomoxetine by CYP2D6)
Adverse effects
Common side effects include abdominal pain, loss of appetite, nausea, feeling tired, and dizziness. Serious side effects may include angioedema, liver problems, stroke, psychosis, heart problems, suicide, and aggression. A 2020 meta-analysis found that atomoxetine was associated with anorexia, weight loss, and hypertension, rating it as a "potentially least preferred agent based on safety" for treating ADHD. As of 2019, safety in pregnancy and breastfeeding is not clear; a 2018 review stated that, "[b]ecause of lack of data, the treating physician should consider stopping atomoxetine treatment in women with ADHD during pregnancy."
The U.S. Food and Drug Administration (FDA) has issued a black box warning for suicidal behavior/ideation. Similar warnings have been issued in Australia. Unlike stimulant medications, atomoxetine does not have abuse liability or the potential to cause withdrawal effects on abrupt discontinuation.
Incidence of adverse effects:
Very common (>10% incidence) adverse effects include:
- Nausea (26%)
- Xerostomia (Dry mouth) (20%)
- Appetite loss (16%)
- Insomnia (15%)
- Fatigue (10%)
- Headache
- Cough
- Vomiting (in children and adolescents)
Common (1–10% incidence) adverse effects include:
- Constipation (8%)
- Dizziness (8%)
- Erectile dysfunction (8%)
- Somnolence (sleepiness) (8%)
- Abdominal pain (7%)
- Urinary hesitation (6%)
- Tachycardia (high heart rate) (5–10%)
- Hypertension (high blood pressure) (5–10%)
- Irritability (5%)
- Abnormal dreams (4%)
- Dyspepsia (4%)
- Ejaculation disorder (4%)
- Hyperhidrosis (abnormally increased sweating) (4%)
- Vomiting (4%)
- Hot flashes (3%)
- Paraesthesia (sensation of tingling, tickling, etc.) (3%)
- Menstrual disorder (3%)
- Weight loss (2%)
- Depression
- Sinus headache
- Dermatitis
- Mood swings
Uncommon (0.1–1% incidence) adverse effects include:
- Suicide-related events
- Hostility
- Emotional lability
- Aggression
- Psychosis
- Syncope (fainting)
- Tremor
- Migraine
- Hypoaesthesia
- Seizure
- Palpitations
- Sinus tachycardia
- QT interval prolongation
- Increased blood bilirubin
- Allergic reactions
Rare (0.01–0.1% incidence) adverse effects including:
- Raynaud's phenomenon
- Abnormal/increased liver function tests
- Jaundice
- Hepatitis
- Liver injury
- Acute liver failure
- Urinary retention
- Priapism
- Male genital pain
Overdose
Atomoxetine is relatively non-toxic in overdose. Single-drug overdoses involving over 1500 mg of atomoxetine have not resulted in death. The most common symptoms of overdose include:
- Gastrointestinal symptoms
- Somnolence
- Dizziness
- Tremor
- Abnormal behaviour
- Hyperactivity
- Agitation
- Dry mouth
- Tachycardia
- Hypertension
- Mydriasis
Less common symptoms:
- Seizures
- QTc interval prolongation
The recommended treatment for atomoxetine overdose includes use of activated charcoal to prevent further absorption of the drug.
Interactions
Atomoxetine is a substrate for CYP2D6. Concurrent treatment with a CYP2D6 inhibitor such as bupropion, fluoxetine, or paroxetine has been shown to increase plasma atomoxetine by 100% or more, as well as increase N-desmethylatomoxetine levels and decrease plasma 4-hydroxyatomoxetine levels by a similar degree.
Atomoxetine has been found to directly inhibit hERG potassium currents with an IC50 of 6.3 μM, which has the potential to cause arrhythmia. QT prolongation has been reported with atomoxetine at therapeutic doses and in overdose; it is suggested that atomoxetine not be used with other medications that may prolong the QT interval, concomitantly with CYP2D6 inhibitors, and caution to be used in poor metabolizers.
Other notable drug interactions include:
- Antihypertensive agents, due to atomoxetine acting as an indirect sympathomimetic
- Indirect-acting sympathomimetics, such as pseudoephedrine, norepinephrine reuptake inhibitors, or MAOIs
- Direct-acting sympathomimetics, such as phenylephrine or other α1 adrenoceptor agonists, including pressors such as dobutamine or isoprenaline and β2 adrenoceptor agonists
- Highly plasma protein-bound drugs: atomoxetine has the potential to displace these drugs from plasma proteins which may potentiate their adverse or toxic effects. In vitro, atomoxetine does not affect the plasma protein binding of aspirin, desipramine, diazepam, paroxetine, phenytoin, or warfarin
Pharmacology
Pharmacodynamics
| Site | ATX | 4-OH-ATX | N-DM-ATX | |
|---|---|---|---|---|
| SERT | 77 | 43 | ND | |
| NET | 5 | 3 | 92 | |
| DAT | 1,451 | ND | ND | |
| 5-HT1A | >1,000 | ND | ND | |
| 5-HT1B | >1,000 | ND | ND | |
| 5-HT1D | >1,000 | ND | ND | |
| 5-HT2 | 2,000 | 1,000 | 1,700 | |
| 5-HT6 | >1,000 | ND | ND | |
| 5-HT7 | >1,000 | ND | ND | |
| α1 | 11,400 | 20,000 | 19,600 | |
| α2A | 29,800 | >30,000 | >10,000 | |
| β1 | 18,000 | 56,100 | 32,100 | |
| M1 | >100,000 | >100,000 | >100,000 | |
| M2 | >100,000 | >100,000 | >100,000 | |
| D1 | >10,000 | >10,000 | >10,000 | |
| D2 | >10,000 | >10,000 | >10,000 | |
| H1 | 12,100 | >100,000 | >100,000 | |
| MOR | ND | 422 | ND | |
| DOR | ND | 300 | ND | |
| KOR | ND | 95 | ND | |
| σ1 | >1,000 | ND | ND | |
| GABAA | 200 | >30,000 | >10,000 | |
| NMDA | 3,470a | ND | ND | |
| Kir3.1/3.2 | 10,900b | ND | ND | |
| Kir3.2 | 12,400b | ND | ND | |
| Kir3.1/3.4 | 6,500b | ND | ND | |
| hERG | 6,300 | 20,000 | 5,710 | |
| Values are Ki (nM). The smaller the value, the more strongly the drug binds to the site. All values are for human receptors unless otherwise specified. arat cortex. bXenopus oocytes. Additional sources: | ||||
Atomoxetine inhibits the presynaptic norepinephrine transporter (NET), preventing the reuptake of norepinephrine throughout the brain along with inhibiting the reuptake of dopamine in specific brain regions such as the prefrontal cortex, where dopamine transporter (DAT) expression is minimal. In rats, atomoxetine increased prefrontal cortex catecholamine concentrations without altering dopamine levels in the striatum or nucleus accumbens; in contrast, methylphenidate, a dopamine reuptake inhibitor, was found to increase prefrontal, striatal, and accumbal dopamine levels to the same degree. In mice, atomoxetine was also found to increase prefrontal catecholamine levels without affecting striatal or accumbal levels.
Atomoxetine's status as a serotonin transporter (SERT) inhibitor at clinical doses in humans is uncertain. A PET imaging study on rhesus monkeys found that atomoxetine occupied >90% and >85% of neural NET and SERT, respectively. However, both mouse and rat microdialysis studies have failed to find an increase in extracellular serotonin in the prefrontal cortex following acute or chronic atomoxetine treatment. Supporting atomoxetine's selectivity, a human study found no effects on platelet serotonin uptake (a marker of SERT inhibition) and inhibition of the pressor effects of tyramine (a marker of NET inhibition).
Atomoxetine has been found to act as an NMDA receptor antagonist in rat cortical neurons at therapeutic concentrations. It causes a use-dependent open-channel block and its binding site overlaps with the Mg2+ binding site. Atomoxetine's ability to increase prefrontal cortex firing rate in anesthetized rats could not be blocked by D1 or α2-adrenergic receptor antagonists, but could be potentiated by NMDA or an α1-adrenergic receptor antagonist, suggesting a glutaminergic mechanism. In Sprague Dawley rats, atomoxetine reduces NR2B protein content without altering transcript levels. Aberrant glutamate and NMDA receptor function have been implicated in the etiology of ADHD.
Atomoxetine also reversibly inhibits GIRK currents in Xenopus oocytes in a concentration-dependent, voltage-independent, and time-independent manner. Kir3.1/3.2 ion channels are opened downstream of M2, α2, D2, and A1 stimulation, as well as other Gi-coupled receptors. Therapeutic concentrations of atomoxetine are within range of interacting with GIRKs, especially in CYP2D6 poor metabolizers. It is not known whether this contributes to the therapeutic effects of atomoxetine in ADHD.
4-Hydroxyatomoxetine, the major active metabolite of atomoxetine in CYP2D6 extensive metabolizers, has been found to have sub-micromolar affinity for opioid receptors, acting as an antagonist at μ-opioid receptors and a partial agonist at κ-opioid receptors. It is not known whether this action at the kappa-opioid receptor leads to CNS-related adverse effects.
Pharmacokinetics
Orally administered atomoxetine is rapidly and completely absorbed. First-pass metabolism by the liver is dependent on CYP2D6 activity, resulting in an absolute bioavailability of 63% for extensive metabolizers and 94% for poor metabolizers. Maximum plasma concentration is reached in 1–2 hours. If taken with food, the maximum plasma concentration decreases by 10-40% and delays the tmax by 1 hour. Drugs affecting gastric pH have no effect on oral bioavailability.
Atomoxetine has a volume of distribution of 0.85 L/kg, with limited partitioning into red blood cells. It is highly bound to plasma proteins (98.7%), mainly albumin, along with α1-acid glycoprotein (77%) and IgG (15%). Its metabolite N-desmethylatomoxetine is 99.1% bound to plasma proteins, while 4-hydroxyatomoxetine is only 66.6% bound.
The half-life of atomoxetine varies widely between individuals, with an average range of 4.5 to 19 hours. As atomoxetine is metabolized by CYP2D6, exposure may be increased 10-fold in CYP2D6 poor metabolizers.
Atomoxetine, N-desmethylatomoxetine, and 4-hydroxyatomoxetine produce minimal to no inhibition of CYP1A2 and CYP2C9, but inhibit CYP2D6 in human liver microsomes at concentrations between 3.6-17 μmol/L. Plasma concentrations of 4-hydroxyatomoxetine and N-desmethylatomoxetine at steady state are 1.0% and 5% that of atomoxetine in CYP2D6 extensive metabolizers, and are 5% and 45% that of atomoxetine in CYP2D6 poor metabolizers.
Atomoxetine is excreted unchanged in urine at <3% in both extensive and poor CYP2D6 metabolizers, with >96% and 80% of a total dose being excreted in urine, respectively. The fractions excreted in urine as 4-hydroxyatomoxetine and its glucuronide account for 86% of a given dose in extensive metabolizers, but only 40% in poor metabolizers. CYP2D6 poor metabolizers excrete greater amounts of minor metabolites, namely N-desmethylatomoxetine and 2-hydroxymethylatomoxetine and their conjugates.
Pharmacogenomics
Chinese adults homozygous for the hypoactive CYP2D6*10 allele have been found to exhibit two-fold higher AUCs and 1.5-fold higher maximum plasma concentrations compared to extensive metabolizers.
Japanese men homozygous for CYP2D6*10 have similarly been found to experience two-fold higher AUCs compared to extensive metabolizers.
Chemistry
Atomoxetine, or (−)-methyl[(3R)-3-(2-methylphenoxy)-3-phenylpropylamine, is a white, granular powder that is highly soluble in water.
Synthesis
Detection in biological fluids
Atomoxetine may be quantitated in plasma, serum or whole blood in order to distinguish extensive versus poor metabolizers in those receiving the drug therapeutically, to confirm the diagnosis in potential poisoning victims or to assist in the forensic investigation in a case of fatal overdosage.
History
Atomoxetine is manufactured, marketed, and sold in the United States as the hydrochloride salt (atomoxetine HCl) under the brand name Strattera by Eli Lilly and Company, the original patent-filing company and current U.S. patent owner. Atomoxetine was initially intended to be developed as an antidepressant, but it was found to be insufficiently efficacious for treating depression. It was, however, found to be effective for ADHD and was approved by the FDA in 2002, for the treatment of ADHD. Its patent expired in May 2017. On 12 August 2010, Lilly lost a lawsuit that challenged its patent on Strattera, increasing the likelihood of an earlier entry of a generic into the US market. On 1 September 2010, Sun Pharmaceuticals announced it would begin manufacturing a generic in the United States. In a 29 July 2011 conference call, however, Sun Pharmaceutical's Chairman stated "Lilly won that litigation on appeal so I think [generic Strattera]'s deferred."
In 2017 the FDA approved the generic production of atomoxetine by four pharmaceutical companies.
Society and culture
Brand names
In India, atomoxetine is sold under brand names including Axetra, Axepta, Attera, Tomoxetin, and Attentin. In Australia, Portugal, Italy and Romania, atomoxetine is sold under the brand name Strattera. In Iran, atomoxetine is sold under brand names including Stramox. In 2017, a generic version was approved in the United States.
Research
There has been some suggestion that atomoxetine might be a helpful adjunct in people with major depression, particularly in cases with concomitant ADHD.